Advanced Apalutamide Intermediate Synthesis: Scalable Green Chemistry for Commercial Production
Advanced Apalutamide Intermediate Synthesis: Scalable Green Chemistry for Commercial Production
The pharmaceutical industry continuously seeks robust synthetic pathways for next-generation oncology treatments, and the recent disclosure in patent CN110818631A offers a transformative approach to producing Apalutamide intermediates. This technology introduces a novel pyridine thiourea derivative synthesis that fundamentally alters the risk profile and economic feasibility of manufacturing this critical androgen receptor antagonist. By shifting away from hazardous gaseous reagents toward stable solid-state precursors, the process addresses long-standing safety concerns while maintaining high chemical fidelity. For global supply chain leaders, this represents a pivotal opportunity to secure a more resilient source of high-purity pharmaceutical intermediates. The strategic implementation of this chemistry ensures compliance with increasingly stringent environmental regulations without compromising on the rigorous purity standards required for clinical-grade materials.
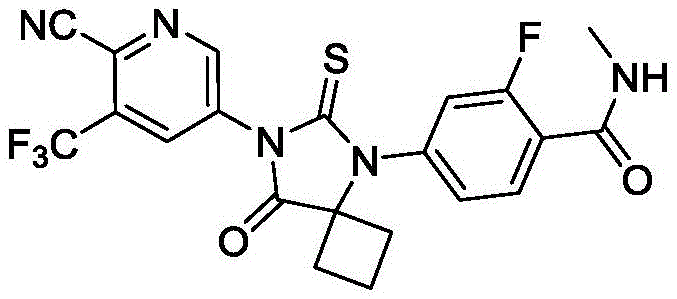
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Apalutamide has relied heavily on routes involving thiophosgene, a reagent notorious for its extreme toxicity and complex handling requirements. As illustrated in prior art such as WO2007126765A2, traditional methods necessitate the conversion of aminopyridine precursors into isothiocyanates using this hazardous gas, creating significant operational bottlenecks. The requirement for specialized containment equipment and rigorous scrubbing systems drastically inflates capital expenditure and operational overheads for manufacturing facilities. Furthermore, these legacy processes often involve microwave-assisted cyclization steps that are notoriously difficult to translate from laboratory benchtop to multi-ton reactor scales. The reliance on sodium cyanide in earlier steps further compounds the safety liabilities, demanding extensive waste treatment protocols that delay production timelines and increase the overall carbon footprint of the manufacturing campaign.
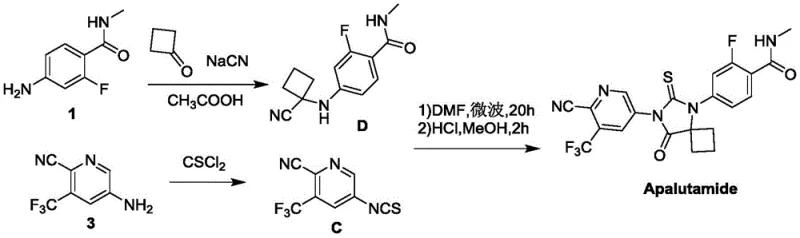
The Novel Approach
In stark contrast, the methodology outlined in CN110818631A circumvents these critical vulnerabilities by employing ammonium thiocyanate and aromatic acyl chlorides to construct the thiourea backbone. This strategic substitution eliminates the need for gaseous thiophosgene entirely, allowing the reaction to proceed under much milder and safer conditions using standard glass-lined reactors. The process utilizes readily available starting materials like 2-cyano-3-trifluoromethyl-5-aminopyridine, ensuring a stable supply chain不受 geopolitical fluctuations affecting specialty gases. By avoiding column chromatography purification in favor of simple crystallization and filtration, the new route significantly reduces solvent consumption and processing time. This streamlined workflow not only enhances the safety profile for plant operators but also delivers a substantial improvement in throughput efficiency, making it an ideal candidate for continuous manufacturing environments.
Mechanistic Insights into Thiourea-Mediated Cyclization
The core innovation lies in the formation and subsequent transformation of the pyridine thiourea derivative, which serves as a versatile linchpin for the entire synthesis. The reaction initiates with the nucleophilic attack of the aminopyridine on an in-situ generated acyl isothiocyanate species, forming the stable intermediate A with high regioselectivity. This intermediate is then subjected to controlled alkaline hydrolysis, cleaving the acyl group to reveal the free thiourea functionality in compound B without degrading the sensitive nitrile or trifluoromethyl groups. The precise control of pH and temperature during this hydrolysis step is critical, as demonstrated by the patent's optimization of base concentrations between 0.5 to 1.0 mol/L. This mechanistic pathway ensures that the reactive thiourea moiety is generated only when needed, minimizing side reactions and polymerization issues that often plague batch processes involving unstable isothiocyanates.
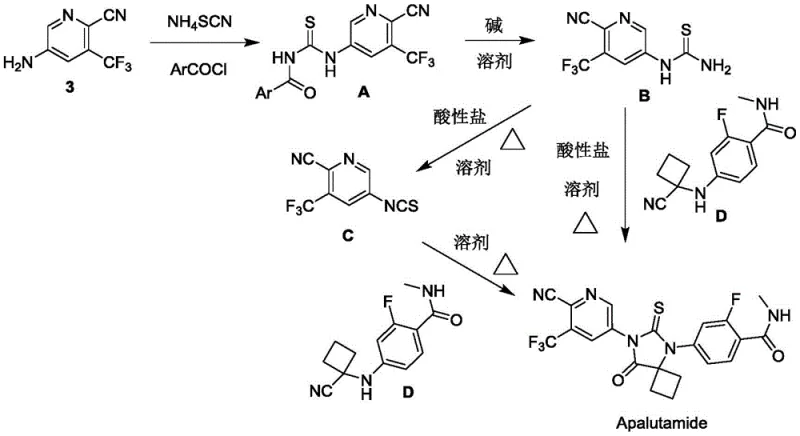
Following the generation of compound B, the final cyclization to form the spiro-hydantoin ring of Apalutamide is achieved through an acid-salt catalyzed condensation with the benzamide intermediate D. The use of acid salts such as potassium bisulfate or sodium bisulfate provides a controlled proton source that activates the thiourea for nucleophilic attack while buffering the reaction medium to prevent acid-catalyzed decomposition of the cyano group. This one-pot transformation is particularly elegant as it combines the generation of the isothiocyanate equivalent and the subsequent cyclization in a single operational sequence. The result is a highly efficient closure of the heterocyclic ring system with excellent stereochemical integrity, yielding the final active pharmaceutical ingredient with minimal impurity burden. This level of mechanistic control is essential for meeting the strict impurity profiles mandated by regulatory bodies for oncology drugs.
How to Synthesize Apalutamide Intermediates Efficiently
Implementing this synthesis requires careful attention to solvent selection and thermal profiles to maximize the yield of the thiourea intermediates. The patent specifies the use of acetone or acetonitrile for the initial acylation, followed by a switch to alcoholic solvents like methanol for the hydrolysis step to ensure complete solubility of the inorganic bases. Operators must maintain reflux conditions for 4 to 8 hours during the formation of derivative A to drive the equilibrium towards completion, followed by a precise pH adjustment to 8.0 during workup to precipitate the product cleanly. For the final cyclization, a biphasic system involving chlorobenzene and DMA is recommended to manage the heat of reaction and facilitate the removal of water generated during the condensation. Adhering to these standardized parameters ensures reproducible results across different batch sizes.
- React 2-cyano-3-trifluoromethyl-5-aminopyridine with ammonium thiocyanate and aromatic acyl chloride to form derivative A.
- Hydrolyze derivative A under alkaline conditions to obtain compound B (thiourea intermediate).
- Condense compound B with o-fluorobenzamide intermediate D using acid salts to cyclize into Apalutamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this thiophosgene-free route offers profound advantages in terms of cost stability and vendor risk management. By removing the dependency on hazardous gas supplies, manufacturers can avoid the premium pricing and logistical complexities associated with transporting and storing toxic reagents. This simplification of the raw material portfolio allows for more accurate forecasting and reduces the likelihood of production stoppages due to supply chain disruptions. Additionally, the elimination of extensive safety mitigation infrastructure translates directly into lower overhead costs, which can be passed down as competitive pricing for the final intermediate. The robustness of the chemistry also means less batch failure, ensuring a consistent flow of materials to downstream formulation partners and strengthening the overall reliability of the drug product supply chain.
- Cost Reduction in Manufacturing: The exclusion of thiophosgene removes the need for expensive scrubbing systems and specialized containment vessels, leading to significant capital expenditure savings. Furthermore, the avoidance of column chromatography in favor of crystallization reduces solvent usage and waste disposal costs, driving down the variable cost per kilogram. The higher yields reported in the patent examples indicate better atom economy, meaning less raw material is wasted, which directly improves the gross margin profile of the manufacturing campaign. These cumulative efficiencies create a leaner cost structure that is resilient against market volatility in raw material pricing.
- Enhanced Supply Chain Reliability: Sourcing ammonium thiocyanate and common aromatic acyl chlorides is far more straightforward than securing regulated toxic gases, ensuring uninterrupted production schedules. The use of commodity solvents like acetone and chlorobenzene further mitigates the risk of solvent shortages that can plague fine chemical manufacturing. This accessibility allows for multi-vendor sourcing strategies, reducing dependency on single suppliers and enhancing negotiation leverage. Consequently, lead times for high-purity pharmaceutical intermediates can be stabilized, providing downstream partners with greater certainty in their own production planning and inventory management.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily transferable from pilot plants to commercial-scale reactors without exothermic runaway risks. The reduction in hazardous waste generation aligns with green chemistry principles, facilitating easier permitting and compliance with environmental protection regulations. This sustainability angle is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals. The simplified workup procedures also reduce the energy load required for solvent recovery, contributing to a lower overall carbon footprint for the manufacturing lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway. They are derived from the specific experimental data and advantageous effects claimed in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer. The answers reflect the objective capabilities of the method as described in the intellectual property, providing a clear basis for further discussion with manufacturing partners.
Q: How does this method improve safety over conventional Apalutamide synthesis?
A: This method eliminates the use of thiophosgene, a highly toxic gas, replacing it with safer ammonium thiocyanate and acid salts, significantly reducing industrial safety risks.
Q: What are the yield advantages of the new thiourea derivative route?
A: Experimental data indicates yields ranging from 67% to 91% for intermediates and up to 82% for the final cyclization, offering robust efficiency for scale-up.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process avoids column chromatography and uses common solvents like acetone and chlorobenzene, making it highly adaptable for multi-kilogram production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apalutamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to adapt and optimize this advanced thiourea-based synthesis for your specific commercial needs. Our engineering team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest industry standards. Our commitment to quality assurance means that you receive intermediates that are ready for immediate use in your downstream API synthesis, minimizing your internal testing burden.
We invite you to engage with our technical procurement team to discuss how this greener, safer route can benefit your supply chain strategy. Request a Customized Cost-Saving Analysis today to quantify the potential economic impact of switching to this thiophosgene-free method. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable and cost-effective supply of high-quality Apalutamide intermediates for your global operations.
