Revolutionizing Benzothienoisoquinoline Production: A Scalable Cu-Catalyzed Synthetic Route for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN110606855B introduces a groundbreaking approach to synthesizing polysubstituted benzothienoisoquinoline and its derivatives, a class of compounds with significant potential in medicinal chemistry and optoelectronic materials. This invention addresses the longstanding challenge of accessing these stable molecular structures through a novel copper-catalyzed transformation that operates under remarkably mild conditions. Unlike traditional methods that often require stringent inert atmospheres or expensive precious metal catalysts, this process leverages abundant sulfur powder and inexpensive copper salts in an open air environment. The resulting compounds exhibit excellent chemical properties and structural stability, making them ideal candidates for further functionalization in drug discovery pipelines. 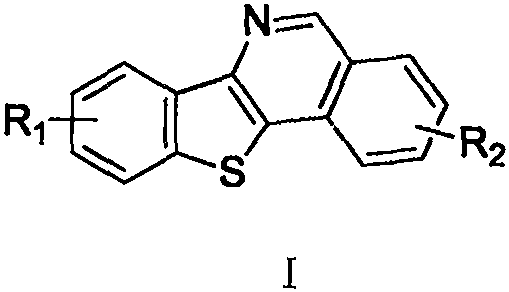
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused thiophene-isoquinoline systems has been plagued by synthetic inefficiencies that hinder commercial viability. Conventional routes often rely on multi-step sequences involving harsh reaction conditions, such as extremely high temperatures or the use of strong, corrosive acids that complicate downstream processing. Furthermore, many established protocols necessitate the use of palladium or rhodium-based catalysts, which not only inflate the raw material costs but also introduce significant challenges regarding heavy metal residue removal in pharmaceutical applications. The requirement for strictly anhydrous and oxygen-free environments adds another layer of operational complexity, demanding specialized equipment and increasing energy consumption. These factors collectively contribute to prolonged lead times and reduced overall atom economy, creating bottlenecks for supply chain managers aiming to scale production reliably.
The Novel Approach
In stark contrast, the methodology disclosed in CN110606855B represents a paradigm shift towards green and sustainable chemical manufacturing. By utilizing a copper-catalyzed system, the inventors have eliminated the dependency on noble metals, thereby drastically simplifying the purification process and reducing the environmental footprint. The reaction proceeds smoothly in an air atmosphere, removing the need for costly inert gas purging and allowing for simpler reactor configurations. This one-pot strategy directly converts ketoxime esters, o-chlorobenzaldehydes, and sulfur powder into the target heterocycles with high selectivity. The simplicity of the operation means that experimental procedures are streamlined, requiring fewer unit operations and less specialized labor. This approach not only enhances the economic feasibility of producing these valuable intermediates but also aligns perfectly with modern regulatory demands for cleaner production technologies.
Mechanistic Insights into Cu-Catalyzed Cyclization
The core of this innovation lies in the intricate interplay between the copper catalyst, sulfur source, and organic substrates under thermal conditions. The reaction mechanism likely involves the initial activation of the carbon-sulfur bond formation followed by a cascade of cyclization events driven by the copper species. The use of elemental sulfur powder is particularly noteworthy, as it serves as an efficient sulfur donor without the hazards associated with gaseous hydrogen sulfide or other volatile sulfur reagents. The copper catalyst facilitates the oxidative coupling and subsequent ring closure, stabilizing the transition states required to form the rigid benzothienoisoquinoline core. 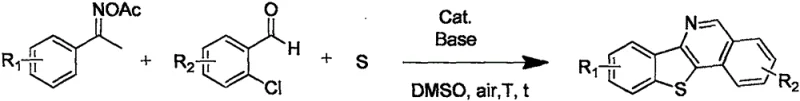
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on product quality. The high atom economy of the reaction ensures that most starting materials are incorporated into the final product, minimizing the generation of complex by-product mixtures that are difficult to separate. The mild reaction temperatures, typically ranging between 120°C and 150°C, prevent thermal degradation of sensitive functional groups on the aromatic rings. This tolerance allows for the introduction of diverse substituents, such as halogens, alkoxy groups, and alkyl chains, without compromising the integrity of the molecule. Consequently, the resulting impurity profile is cleaner, reducing the burden on analytical teams and accelerating the timeline for regulatory filing and approval.
How to Synthesize Polysubstituted Benzothienoisoquinoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal profiles to maximize yield and purity. The process begins with the precise weighing and mixing of the ketoxime ester and o-chlorobenzaldehyde precursors alongside the sulfur powder and base. A suitable copper salt, such as cuprous chloride or cupric acetate, is then introduced along with a polar aprotic solvent like DMSO or DMF to ensure homogeneous dissolution and effective catalysis. The detailed standardized synthesis steps see the guide below.
- Mix ketoxime ester compounds, o-chlorobenzaldehyde compounds, sulfur powder, a copper catalyst, and an organic solvent in a reaction vessel.
- Heat the reactants to 120-150°C in an air atmosphere for 1-8 hours to facilitate the cyclization reaction.
- Purify the crude reaction mixture to isolate the high-purity polysubstituted benzothienoisoquinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The shift from precious metal catalysts to abundant copper salts fundamentally alters the cost structure of the manufacturing process, offering substantial cost savings on raw material procurement. Additionally, the elimination of inert atmosphere requirements reduces utility costs and equipment maintenance needs, further enhancing the overall economic efficiency of the production line. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term supply agreements with global pharmaceutical partners.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with inexpensive copper compounds results in a significant decrease in direct material costs. Furthermore, the simplified workup procedure reduces the consumption of purification solvents and adsorbents, leading to lower waste disposal fees. By avoiding the need for specialized inert gas infrastructure, capital expenditure on reactor setup is also minimized, allowing for faster ROI on new production lines dedicated to these high-value intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including ketoxime esters and o-chlorobenzaldehydes, are commercially available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The stability of the reagents allows for bulk purchasing and long-term storage without significant degradation, enabling strategic stockpiling to buffer against market volatility. This reliability ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and strengthening relationships with downstream clients.
- Scalability and Environmental Compliance: The one-pot nature of the reaction simplifies the scale-up process from gram to ton scale, as there are fewer intermediate isolation steps that typically cause yield losses. The use of air as an oxidant and the absence of toxic gaseous reagents align with strict environmental regulations, reducing the permitting burden for new facilities. This eco-friendly profile enhances the corporate sustainability image and ensures compliance with increasingly rigorous green chemistry standards imposed by international regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this Cu-catalyzed method over traditional synthesis?
A: This method utilizes cheap copper catalysts instead of expensive noble metals, operates under mild air atmosphere conditions without inert gas protection, and employs readily available raw materials, significantly reducing environmental pollution and production costs.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process features a simple reaction system, requires less reaction equipment, and uses a one-pot method that saves development time. The mild conditions and wide range of available materials make it highly adaptable for mass production and popularization.
Q: What types of substituents are compatible with this benzothienoisoquinoline synthesis?
A: The method supports a broad scope of substrates, including various halogen groups, alkyls, substituted aryls, and heterocyclic groups containing nitrogen, oxygen, or sulfur atoms, allowing for the creation of diverse derivatives with stable molecular structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Benzothienoisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods described in CN110606855B for the production of advanced heterocyclic building blocks. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of polysubstituted benzothienoisoquinoline meets the highest industry standards for pharmaceutical and electronic applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next product launch. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for complex organic syntheses.
