Advanced Catalyst-Free Route for High-Purity 2,4-Disubstituted Thiophene Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more sustainable and cost-effective manufacturing processes for critical intermediates. A significant breakthrough in this domain is documented in patent CN103694217A, which discloses a novel preparation method for 2,4-disubstituted thiophene derivatives. These compounds are pivotal building blocks in the synthesis of pharmaceuticals, agrochemicals, and functional materials, yet their traditional production has often been hindered by complex catalytic systems and harsh reaction environments. This patent introduces a transformative approach that utilizes simple enyne ether compounds and potassium sulfide as raw materials, operating under remarkably mild conditions of room temperature and ambient air. By eliminating the need for expensive transition metal catalysts and rigorous anhydrous conditions, this technology offers a compelling value proposition for industrial-scale production, addressing key pain points related to cost, safety, and environmental compliance in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4-disubstituted thiophene derivatives has relied heavily on methodologies that impose significant operational burdens on chemical manufacturers. Conventional literature often describes routes that necessitate the use of precious metal catalysts such as palladium or heavy metals like copper, which not only escalate the raw material costs but also introduce severe challenges in downstream processing. The removal of trace metal residues to meet stringent pharmaceutical purity specifications requires additional purification steps, such as specialized chromatography or scavenging treatments, which drastically reduce overall throughput and increase waste generation. Furthermore, many existing protocols demand harsh reaction conditions, including high-temperature reflux and extended reaction times, which consume substantial energy and pose safety risks associated with thermal runaway or pressure buildup in large-scale reactors. These factors collectively contribute to a high cost of goods sold (COGS) and a fragile supply chain that is vulnerable to fluctuations in the availability of specialized catalysts.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN103694217A presents a streamlined and economically superior pathway that fundamentally reshapes the production economics of thiophene intermediates. The core innovation lies in the utilization of potassium sulfide (K2S) as a sulfur source in conjunction with enyne ether precursors, facilitating a direct cyclization that bypasses the need for any transition metal catalysis. This catalyst-free system operates efficiently at room temperature, typically between 10°C and 30°C, and tolerates the presence of air, thereby removing the capital expenditure associated with inert gas blanketing and specialized pressure vessels. The simplicity of the reagent profile, combined with the use of common solvents like N,N-dimethylformamide (DMF), ensures that the process is highly scalable and robust. By shifting to this methodology, manufacturers can achieve high yields ranging from 56% to 90% while simultaneously simplifying the workup procedure, as the absence of metal catalysts eliminates the need for complex metal scavenging steps.
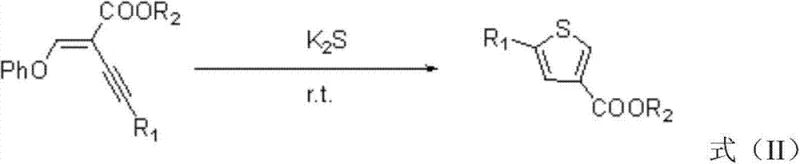
Mechanistic Insights into Potassium Sulfide-Mediated Cyclization
The mechanistic elegance of this transformation provides deep insights into why this method achieves such high efficiency under mild conditions. The reaction initiates with the nucleophilic attack of the sulfide ion (S²⁻) derived from potassium sulfide on the electron-deficient double bond of the enyne ether substrate. This initial addition generates a carbanion intermediate which subsequently undergoes an intramolecular cyclization involving the alkyne moiety. The electronic properties of the enyne system are perfectly tuned to facilitate this cascade, where the initial sulfur addition activates the adjacent triple bond for nucleophilic attack by the newly formed thiolate species. This concerted sequence leads to the formation of the thiophene ring skeleton with high regioselectivity, ensuring that the 2,4-substitution pattern is established cleanly without significant formation of isomeric byproducts. The final step involves protonation to aromatize the ring and release the phenoxide leaving group, driving the equilibrium towards the desired thiophene product.
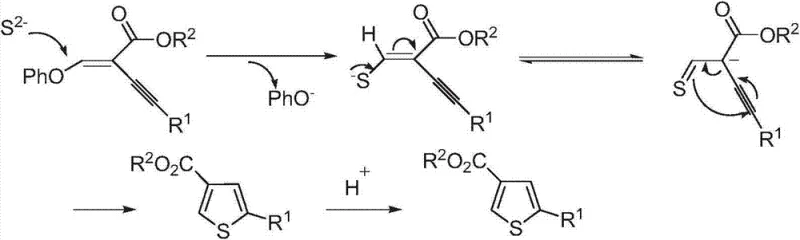
From an impurity control perspective, this mechanism offers distinct advantages that are critical for R&D directors focused on product quality. Because the reaction proceeds through a well-defined ionic pathway rather than a radical or metal-mediated cycle, the formation of complex polymeric side products or homocoupling impurities is significantly suppressed. The mild thermal conditions further mitigate the risk of thermal degradation of sensitive functional groups on the aromatic rings, such as methoxy or chloro substituents, which might otherwise decompose under the high temperatures required by traditional reflux methods. Additionally, the stoichiometric use of potassium sulfide ensures complete consumption of the starting material without the accumulation of unreacted intermediates that could complicate purification. The resulting crude product typically exhibits a high degree of purity, allowing for straightforward isolation via standard extraction and crystallization techniques, which is a major benefit for maintaining consistent batch-to-batch quality in commercial manufacturing.
How to Synthesize 2,4-Disubstituted Thiophene Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The process is designed to be operationally simple, leveraging the high reactivity of the enyne ether precursor with the inexpensive sulfide source. The key to success lies in maintaining the correct molar ratio of reactants and ensuring adequate mixing in the polar aprotic solvent to facilitate the ionic mechanism. While the reaction is tolerant of air, good agitation is necessary to ensure the solid potassium sulfide remains suspended and reactive throughout the two-hour duration. The following guide outlines the standardized procedure derived from the patent examples, providing a clear roadmap for technical teams to replicate these results effectively.
- Mix enyne ether compounds and potassium sulfide in a molar ratio of 1: 2.
- Dissolve the mixture in N,N-dimethylformamide (DMF) solvent under air conditions.
- Stir the reaction at room temperature (10°C-30°C) for 2 hours, followed by separation and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates directly into tangible strategic advantages that enhance the overall competitiveness of the supply base. The elimination of precious metal catalysts represents a massive shift in cost structure, removing the volatility associated with the pricing of palladium and copper markets. Furthermore, the simplified downstream processing reduces the consumption of auxiliary materials such as silica gel for chromatography or specialized scavenger resins, leading to substantial cost savings in waste disposal and material handling. The robustness of the reaction conditions also means that the process can be executed in standard multipurpose reactors without requiring specialized linings or high-pressure ratings, thereby maximizing asset utilization and reducing capital barriers for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive transition metal catalysts, which are often the single largest cost driver in fine chemical synthesis. By substituting these with commodity-grade potassium sulfide, the raw material cost profile is drastically improved, allowing for more competitive pricing in long-term supply agreements. Additionally, the reduction in purification complexity lowers the labor and utility costs associated with extended reaction times and complex workups, contributing to a leaner and more efficient manufacturing operation that can better absorb market fluctuations.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable raw materials such as enyne ethers and inorganic sulfides ensures a resilient supply chain that is less susceptible to geopolitical disruptions or shortages of specialized reagents. Since the reaction does not require strict inert atmosphere conditions, the logistical burden of transporting and storing sensitive catalysts under nitrogen is eliminated, simplifying inventory management and reducing the risk of production delays due to reagent degradation. This stability allows for more accurate forecasting and planning, ensuring consistent delivery schedules for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metals align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Scaling this process from kilogram to tonnage levels does not introduce significant safety hazards related to exotherms or high pressures, making it an ideal candidate for continuous manufacturing or large-batch production. The reduced generation of heavy metal-contaminated waste streams simplifies effluent treatment and lowers the environmental compliance burden, enhancing the sustainability profile of the final product and supporting corporate ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiophene synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this route for potential integration into their supply chains. Understanding these details is crucial for assessing the feasibility of adopting this method for specific project requirements.
Q: What are the primary advantages of this thiophene synthesis method over conventional routes?
A: Unlike conventional methods requiring expensive palladium or copper catalysts and harsh reflux conditions, this patented process operates at room temperature in air without any transition metal catalysts, significantly reducing raw material costs and simplifying purification.
Q: What is the substrate scope for the R1 group in this thiophene derivative synthesis?
A: The method demonstrates excellent universality, successfully accommodating C1-20 alkyl groups, benzyl groups, and various aryl groups including phenyl, substituted phenyl, and naphthyl moieties with high conversion rates.
Q: What are the specific reaction conditions required for this cyclization?
A: The reaction proceeds efficiently under mild conditions, specifically at temperatures between 10°C and 30°C in DMF solvent, completing within approximately 2 hours without the need for inert atmosphere protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Disubstituted Thiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation pharmaceutical and agrochemical products. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN103694217A and validated its potential for commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2,4-disubstituted thiophene derivative we supply meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in catalyst-free heterocyclic synthesis can drive value and efficiency in your supply chain.
