Advanced Gold-Catalyzed Synthesis of Tetrahydrofuran Dihydroquinolines for Commercial Pharmaceutical Applications
Advanced Gold-Catalyzed Synthesis of Tetrahydrofuran Dihydroquinolines for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN112574225A introduces a groundbreaking advancement in this domain by detailing a highly efficient, gold-catalyzed 1,1-carboalkoxylation strategy for synthesizing tetrahydrofuran-fused 1,4-dihydroquinoline derivatives. This innovation represents a significant leap forward in synthetic organic chemistry, specifically addressing the challenges associated with forming densely functionalized tricyclic nitrogen-containing heterocycles. Unlike traditional approaches that often necessitate elevated temperatures, prolonged reaction times, or hazardous reagents, this novel protocol achieves exceptional conversion rates under remarkably mild conditions, specifically at room temperature. The ability to construct these valuable cores in a single step with high atom economy positions this technology as a transformative solution for the manufacturing of high-purity pharmaceutical intermediates, directly impacting the cost structures and supply chain reliability for global drug developers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused heterocyclic systems like tetrahydrofuran dihydroquinolines has relied on multi-step sequences involving harsh cyclization conditions that frequently compromise yield and purity. Conventional transition metal-catalyzed reactions often require stoichiometric amounts of oxidants or extreme thermal energy to drive the formation of carbon-oxygen and carbon-nitrogen bonds simultaneously, leading to significant generation of chemical waste and increased safety risks in a production environment. Furthermore, many established protocols suffer from limited substrate scope, failing to tolerate sensitive functional groups that are increasingly common in modern drug design, thereby necessitating additional protection and deprotection steps that inflate both time and material costs. The reliance on internal cyclization pathways in prior art often restricts the structural diversity accessible to chemists, limiting the exploration of novel chemical space for drug discovery programs. These inefficiencies create substantial bottlenecks in the supply chain, where the inability to scale processes reliably without sacrificing quality remains a persistent challenge for procurement teams managing complex API pipelines.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112574225A leverages a sophisticated gold-catalyzed exocyclization mechanism that proceeds with unprecedented efficiency and selectivity. By utilizing a cationic gold(I) catalyst, specifically IPrAuNTf2, the process activates the alkyne moiety to facilitate a 1,1-carboalkoxylation followed by a 1,2-nitrogen migration, effectively assembling the tricyclic core in a single operational step. This approach eliminates the need for external additives or harsh thermal inputs, as the reaction reaches completion within merely one hour at ambient temperature, drastically reducing energy consumption and operational complexity. The versatility of this method is demonstrated by its broad tolerance for various substituents on the aromatic ring and the alkyne terminus, allowing for the rapid generation of diverse libraries of compounds without the need for extensive process re-optimization.  This streamlined pathway not only enhances the speed of synthesis but also ensures a cleaner reaction profile, which simplifies downstream purification and significantly improves the overall mass balance of the manufacturing process.
This streamlined pathway not only enhances the speed of synthesis but also ensures a cleaner reaction profile, which simplifies downstream purification and significantly improves the overall mass balance of the manufacturing process.
Mechanistic Insights into Gold-Catalyzed 1,1-Carboalkoxylation
The mechanistic elegance of this transformation lies in the unique ability of the gold catalyst to stabilize reactive carbene intermediates through pi-coordination, thereby lowering the activation energy barrier for the cyclization event. Upon coordination of the gold species to the alkyne triple bond, the proximal oxygen atom of the oxetane ring performs a nucleophilic attack, triggering a ring-expansion event that generates a transient exocyclic gold carbene species. This critical intermediate then undergoes a facile 1,2-nitrogen migration, where the nitrogen atom shifts to the carbene center, ultimately forging the new carbon-nitrogen bond that completes the dihydroquinoline framework. 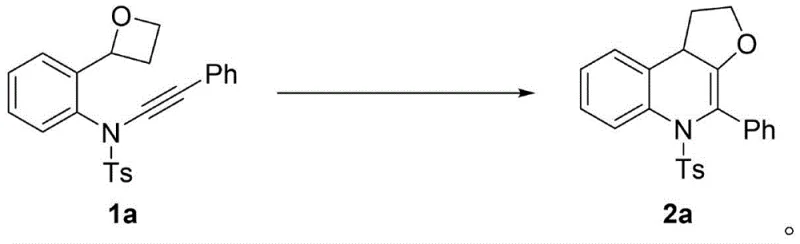 The precision of this migratory insertion is governed by the electronic properties of the ligand system on the gold center, with the N-heterocyclic carbene ligand in IPrAuNTf2 providing the optimal steric and electronic environment to suppress competing side reactions such as polymerization or non-selective hydration. Understanding this catalytic cycle is paramount for R&D directors aiming to replicate these results, as it highlights the importance of maintaining strict anhydrous conditions and inert atmospheres to preserve the integrity of the active gold species throughout the reaction timeline.
The precision of this migratory insertion is governed by the electronic properties of the ligand system on the gold center, with the N-heterocyclic carbene ligand in IPrAuNTf2 providing the optimal steric and electronic environment to suppress competing side reactions such as polymerization or non-selective hydration. Understanding this catalytic cycle is paramount for R&D directors aiming to replicate these results, as it highlights the importance of maintaining strict anhydrous conditions and inert atmospheres to preserve the integrity of the active gold species throughout the reaction timeline.
From an impurity control perspective, the high selectivity of this gold-catalyzed pathway minimizes the formation of regioisomers and oligomeric byproducts that typically plague alkyne functionalization reactions. The mild reaction conditions prevent the degradation of sensitive functional groups, ensuring that the final product retains the intended substitution pattern required for downstream biological activity. Moreover, the use of a well-defined homogeneous catalyst allows for predictable kinetics, enabling process chemists to accurately model reaction progress and determine precise endpoints to maximize yield while minimizing residual metal content. This level of control is essential for meeting the stringent purity specifications demanded by regulatory bodies for pharmaceutical ingredients, as it reduces the burden on purification units and ensures a consistent quality profile across different production batches.
How to Synthesize Tetrahydrofuran Dihydroquinoline Efficiently
The implementation of this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent quality and atmospheric control to fully realize the benefits outlined in the patent documentation. The procedure begins with the dissolution of the alkynylamine precursor in a suitable organic solvent, with dichloromethane identified as the optimal medium for balancing solubility and reaction rate. Following the establishment of an inert nitrogen atmosphere to exclude moisture and oxygen, the gold catalyst is introduced at room temperature, initiating the cascade sequence that rapidly converts the starting material into the desired heterocyclic product. Detailed standardized synthesis steps see the guide below.
- Dissolve the alkynylamine precursor in an organic solvent such as dichloromethane under an inert nitrogen atmosphere.
- Add the IPrAuNTf2 gold catalyst at room temperature and stir the mixture for approximately 1 hour until reaction completion.
- Concentrate the reaction mixture and purify the residue via silica gel column chromatography to isolate the target heterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this gold-catalyzed methodology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The ability to conduct the reaction at room temperature translates directly into substantial energy savings, as there is no requirement for heating mantles, oil baths, or cryogenic cooling systems that drive up utility costs in large-scale manufacturing facilities. Additionally, the shortened reaction time of approximately one hour significantly increases throughput capacity, allowing existing reactor vessels to produce more batches per week without the need for capital investment in new equipment. This intensification of the process timeline enhances supply chain agility, enabling manufacturers to respond more rapidly to fluctuating market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the reduction in solvent volume due to higher concentration feasibility contribute to a leaner cost structure for producing these complex heterocycles. By avoiding multi-step sequences that require isolation of unstable intermediates, the process minimizes material loss and labor hours associated with handling and transfer operations. Furthermore, the high atom economy ensures that a greater proportion of the raw material input is converted into valuable product rather than waste, aligning with green chemistry principles that are increasingly valued by corporate sustainability mandates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to variations in ambient temperature or minor fluctuations in reagent quality, leading to more consistent batch-to-batch performance. This reliability reduces the risk of production delays caused by failed runs or out-of-specification results, ensuring a steady flow of materials to downstream formulation units. The broad substrate scope also allows for the sourcing of diverse starting materials from multiple suppliers, mitigating the risk of supply disruptions associated with single-source dependencies for specialized reagents.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scales is facilitated by the exothermic nature of the cyclization being manageable under ambient conditions, removing the need for complex heat exchange systems often required for highly exothermic traditional cyclizations. The simplified workup procedure, involving basic concentration and chromatography, reduces the volume of aqueous waste streams generated, easing the burden on wastewater treatment facilities. This environmental compatibility supports compliance with increasingly rigorous industrial discharge regulations, preventing potential fines and operational shutdowns related to effluent management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology, derived from a detailed analysis of the experimental data and process parameters provided in the intellectual property disclosure. These insights are intended to clarify the operational feasibility and strategic value of adopting this gold-catalyzed route for the production of tetrahydrofuran dihydroquinoline derivatives. Stakeholders are encouraged to review these points to understand how this innovation aligns with their specific development goals and supply chain requirements.
Q: What are the primary advantages of this gold-catalyzed method over traditional cyclization routes?
A: This method operates at room temperature with high atom economy, eliminating the need for harsh heating or stoichiometric oxidants often required in conventional transition metal catalysis.
Q: Is the catalyst loading significant for large-scale production costs?
A: The process utilizes low catalyst loading (typically 5 mol%), and the high turnover efficiency combined with mild conditions significantly reduces overall operational expenditures compared to multi-step alternatives.
Q: What is the biological potential of the synthesized tetrahydrofuran dihydroquinolines?
A: Preliminary evaluations indicate potent cytotoxicity against various cancer cell lines including JeKo-1 and HepG2, suggesting strong value as intermediates for oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrofuran Dihydroquinoline Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this cutting-edge academic research into reliable commercial reality for global pharmaceutical clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of tetrahydrofuran dihydroquinoline intermediates meets the highest standards of quality and consistency required for drug development.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to delivering superior value and supply chain security for your critical pharmaceutical projects.
