Advanced Copper-Catalyzed Synthesis of 3-Chloro-4-Amino Maleimide Derivatives for Pharmaceutical Applications
The landscape of medicinal chemistry is constantly evolving, driven by the demand for complex scaffolds that can mimic natural products or inhibit specific biological pathways. Among these, 3,4-bifunctional maleimides have emerged as privileged structures, serving as core skeletons for a wide array of bioactive molecules ranging from marine alkaloids to potent antitumor agents. As illustrated in the structural diversity of compounds like Isogranulatimide and Himanimide A, the ability to efficiently construct these densely functionalized heterocycles is paramount for drug discovery efforts 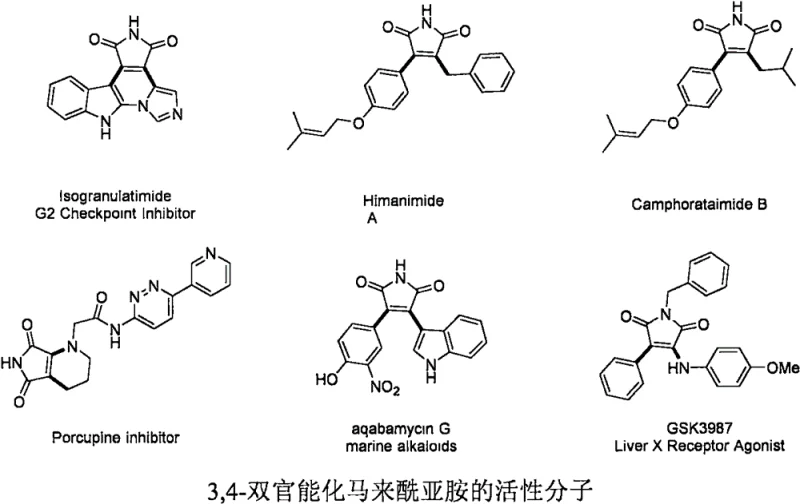 . Addressing this synthetic challenge, the recent patent CN112661690A discloses a groundbreaking preparation method for 3-chloro-4-amido maleimide compounds. This technology leverages a transition metal-catalyzed radical tandem reaction, utilizing inexpensive N-chlorosuccinimide and secondary amines to achieve high-yield functionalization under mild oxidative conditions, offering a robust solution for the reliable pharmaceutical intermediate supplier seeking to optimize their pipeline.
. Addressing this synthetic challenge, the recent patent CN112661690A discloses a groundbreaking preparation method for 3-chloro-4-amido maleimide compounds. This technology leverages a transition metal-catalyzed radical tandem reaction, utilizing inexpensive N-chlorosuccinimide and secondary amines to achieve high-yield functionalization under mild oxidative conditions, offering a robust solution for the reliable pharmaceutical intermediate supplier seeking to optimize their pipeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of halogenated and aminated maleimide derivatives has been fraught with significant chemical and operational hurdles. Conventional routes often rely on multi-step sequences involving the protection and deprotection of sensitive functional groups, which drastically increases the overall process time and material costs. Furthermore, classical halogenation methods frequently employ hazardous reagents or require cryogenic conditions that are difficult to maintain on a large industrial scale. The lack of regioselectivity in direct electrophilic substitution on the maleimide ring often leads to complex mixtures of isomers, necessitating rigorous and yield-depleting purification steps. Additionally, many existing protocols depend on precious metal catalysts or exotic ligands that are not only cost-prohibitive but also pose challenges regarding residual metal removal, a critical quality attribute for any high-purity OLED material or API intermediate intended for human consumption.
The Novel Approach
In stark contrast to these legacy methods, the invention described in CN112661690A introduces a streamlined, one-pot strategy that fundamentally simplifies the construction of the 3-chloro-4-amino motif. By employing N-chlorosuccinimide (NCS) as a dual-purpose reagent—serving as both the chlorine source and the oxidant precursor—the process eliminates the need for separate halogenation and amination steps. The core innovation lies in the in-situ generation of a chloroamination reagent facilitated by a copper catalyst under an oxygen atmosphere. This approach not only enhances atom economy but also ensures excellent regioselectivity, directly installing the chlorine and amino groups at the 3 and 4 positions of the maleimide ring respectively. The general reaction scheme, depicted below, highlights the convergence of N-substituted maleimides, secondary amines, and NCS to form the target scaffold with remarkable efficiency 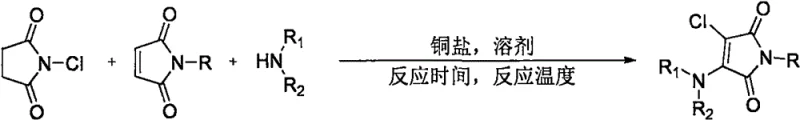 .
.
Mechanistic Insights into Copper-Catalyzed Radical Tandem Reaction
The success of this transformation hinges on a sophisticated radical mechanism orchestrated by the transition metal copper catalyst. Under the specified reaction conditions, typically involving cuprous chloride in a solvent like toluene, the copper species activates the N-chlorosuccinimide to generate reactive nitrogen-centered radicals or chlorinating species in situ. These active intermediates then engage the electron-deficient double bond of the N-substituted maleimide. The presence of molecular oxygen is crucial, acting as a terminal oxidant to regenerate the active copper catalyst and drive the radical propagation cycle forward. This oxidative environment prevents the accumulation of reduced copper species that could otherwise stall the reaction, ensuring a continuous turnover number that supports high conversion rates even with low catalyst loading (typically 10 mol%).
From an impurity control perspective, this mechanistic pathway offers distinct advantages over ionic alternatives. The radical nature of the reaction tends to be more tolerant of various functional groups on the amine and maleimide substrates, reducing the formation of side products associated with nucleophilic attacks on carbonyl groups. The specific choice of cuprous chloride over other copper salts, such as copper acetate or copper fluoride, is critical; experimental data indicates that CuCl provides the optimal redox potential to facilitate the homolytic cleavage of the N-Cl bond without promoting undesirable decomposition pathways. This precise control over the radical generation step results in a cleaner reaction profile, thereby simplifying downstream purification and enhancing the overall purity of the final high-purity pharmaceutical intermediate.
How to Synthesize 3-Chloro-4-Amino Maleimide Efficiently
Implementing this novel synthetic route requires careful attention to reaction parameters to maximize yield and reproducibility. The protocol generally involves mixing the N-substituted maleimide substrate with an excess of N-chlorosuccinimide and the secondary amine in an organic solvent, followed by the addition of the copper catalyst. The system must be rigorously degassed and backfilled with oxygen to establish the necessary oxidative environment before heating. Detailed standardized operating procedures, including specific molar ratios and workup techniques, are essential for transferring this laboratory-scale success to pilot plant operations. For a comprehensive breakdown of the exact experimental steps and conditions validated in the patent examples, please refer to the technical guide below.
- Combine N-phenylmaleimide, N-chlorosuccinimide (NCS), and cuprous chloride catalyst in toluene solvent at room temperature.
- Add the secondary amine (e.g., morpholine) and perform three cycles of evacuation and oxygen filling to ensure an oxidative environment.
- Stir the mixture at 100°C for approximately 23 hours, then cool, dilute with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for complex heterocyclic intermediates. The shift away from precious metals and multi-step syntheses directly addresses the volatility often seen in the pricing of specialized reagents. By utilizing commodity chemicals like N-chlorosuccinimide and simple cyclic amines, manufacturers can decouple their production costs from the fluctuations of the rare earth or precious metal markets. Furthermore, the simplified workflow reduces the manpower and equipment time required per batch, leading to substantial operational expenditure savings without compromising on the quality of the output.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive catalysts and reagents with inexpensive, bulk-available alternatives. The use of cuprous chloride, a base metal salt, instead of palladium or rhodium complexes eliminates the need for costly metal scavenging steps, which are often mandatory to meet regulatory limits for residual metals in pharmaceutical ingredients. Additionally, the high atom economy of the tandem reaction means less waste is generated per kilogram of product, reducing the burden on waste treatment facilities and lowering the overall environmental compliance costs associated with production.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical intermediates is a top priority for any organization managing complex drug development timelines. This synthesis route relies on raw materials that are widely produced and stocked by global chemical suppliers, minimizing the risk of shortages due to geopolitical issues or single-source dependencies. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-dry or inert atmospheres beyond standard oxygen sparging, further ensures that production can continue uninterrupted even in varying facility conditions, thereby reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, but this method is inherently designed for expansion. The use of toluene, a common industrial solvent with well-established recovery and recycling protocols, facilitates easy integration into existing manufacturing infrastructure. The reaction generates minimal hazardous byproducts compared to traditional halogenation methods that might produce stoichiometric amounts of acidic waste. This green chemistry profile aligns with modern sustainability goals, making it easier to obtain regulatory approvals and maintain a social license to operate in increasingly environmentally conscious jurisdictions.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthetic method is vital for making informed sourcing and development decisions. The following questions address common inquiries regarding the scope, catalyst sensitivity, and practical application of this patented technology. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a factual basis for evaluating its fit within your specific project requirements.
Q: What is the critical catalyst for this maleimide functionalization?
A: The patent identifies cuprous chloride (CuCl) as the optimal transition metal catalyst. While other copper salts like copper acetate showed low activity, and copper fluoride or bromide showed no reaction, CuCl provided high yields up to 90%.
Q: Can this method accommodate different amine substrates?
A: Yes, the methodology demonstrates broad substrate scope. It successfully utilizes cyclic secondary amines such as morpholine, piperidine, and pyrrolidine to generate diverse 3-chloro-4-amino maleimide derivatives.
Q: Is this synthesis suitable for large-scale manufacturing?
A: The process utilizes inexpensive, commercially available raw materials like N-chlorosuccinimide and toluene. The reaction conditions (100°C, atmospheric oxygen) and simple workup procedures suggest high potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-4-Amino Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a promising patent to a commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative copper-catalyzed route described in CN112661690A can be effectively translated into a robust manufacturing process. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-chloro-4-amino maleimide meets the exacting standards required for downstream drug synthesis.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in fine chemical intermediates can accelerate your development pipeline and secure your supply chain for the future.
