Scalable Cobalt-Catalyzed Synthesis of Indolo[1,2-a]quinazolin-6(5H)-one Intermediates
Introduction to Advanced Heterocyclic Synthesis
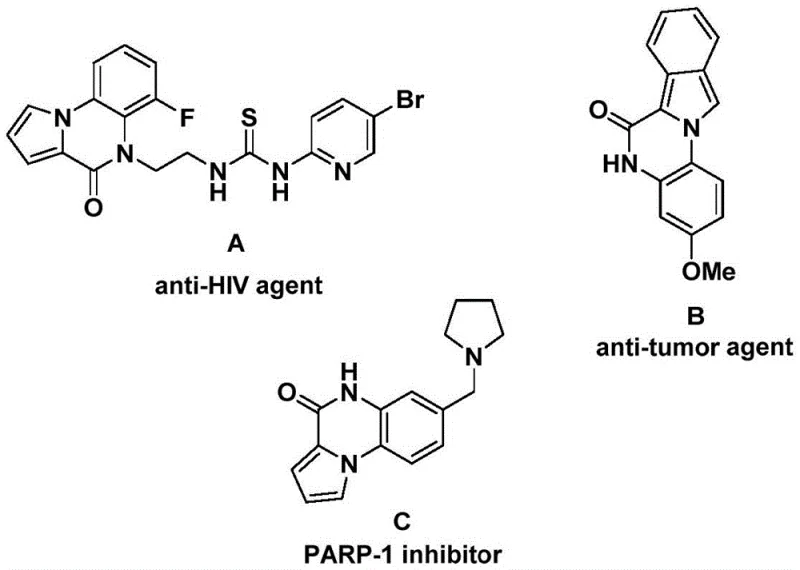
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. Patent CN112321593B discloses a groundbreaking preparation method for indolo[1,2-a]quinazolin-6(5H)-one compounds, a privileged structural motif found in numerous bioactive molecules. As illustrated in the structural diversity of known pharmacophores, this core skeleton is integral to the design of anti-HIV agents, anti-tumor molecules, and PARP-1 inhibitors, highlighting its critical role in drug discovery pipelines. The ability to access these complex architectures efficiently is paramount for accelerating the development of next-generation therapeutics targeting oncology and infectious diseases.
This novel methodology represents a significant leap forward in organic synthesis by leveraging transition metal cobalt catalysis to achieve C-H activation and carbonylation in a single operational step. Unlike traditional approaches that often rely on expensive and toxic precious metals, this invention utilizes a robust cobalt catalyst system combined with a solid carbon monoxide surrogate. This strategic shift not only enhances the safety profile of the reaction by avoiding high-pressure CO gas but also drastically improves the economic feasibility of the process. For research and development teams seeking reliable pharmaceutical intermediate suppliers, this technology offers a pathway to high-purity intermediates with superior cost-efficiency and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolo[1,2-a]quinazolin-6(5H)-one framework has been fraught with synthetic challenges, primarily relying on palladium-catalyzed carbonylation reactions. These conventional methods typically necessitate the use of stoichiometric amounts of expensive palladium catalysts, which poses significant cost barriers for large-scale manufacturing. Furthermore, traditional carbonylation protocols often require the handling of toxic carbon monoxide gas under high pressure, demanding specialized reactor equipment and stringent safety measures that complicate process scale-up. The reliance on precious metals also introduces complications in downstream processing, where rigorous purification steps are needed to remove trace metal residues to meet strict pharmaceutical regulatory standards.
The Novel Approach
The inventive process described in CN112321593B overcomes these hurdles by introducing a cobalt-catalyzed C-H activation strategy that operates under markedly milder and safer conditions. By employing cobalt chloride as the catalyst and phenyl 1,3,5-tricarboxylate as a solid carbon monoxide source, the reaction eliminates the need for gaseous CO infrastructure. This approach not only simplifies the operational setup but also ensures a steady, controlled release of CO in situ, leading to higher reaction efficiency and better selectivity. The method demonstrates exceptional substrate compatibility, tolerating a wide range of functional groups including halogens, esters, and alkoxy substituents, thereby enabling the rapid synthesis of diverse analogues for structure-activity relationship studies without the need for extensive protecting group manipulations.

Mechanistic Insights into Cobalt-Catalyzed C-H Carbonylation
The mechanistic pathway of this transformation involves a sophisticated catalytic cycle initiated by the oxidation of the cobalt(II) precursor to a high-valent cobalt(III) species by silver carbonate. This active cobalt(III) intermediate subsequently coordinates with the 2-pyridinecarboxamide derivative, facilitating the crucial C-H bond activation at the C2 position of the indole ring. This step is pivotal as it forms a stable cobalt(III) metallacycle, setting the stage for the subsequent carbonyl insertion. The use of phenyl 1,3,5-tricarboxylate serves as an elegant solution for CO delivery; upon thermal decomposition, it releases carbon monoxide which inserts into the cobalt-carbon bond to generate an acyl-cobalt(III) intermediate. This sequence ensures that the carbonylation occurs with high regioselectivity and minimal formation of decarbonylated byproducts.
Following the CO insertion, the acyl-cobalt(III) species undergoes reductive elimination to forge the new C-C bond, closing the quinazolinone ring system. The final step involves hydrolysis or protonolysis to release the desired indolo[1,2-a]quinazolin-6(5H)-one product and regenerate the active catalyst species. From an impurity control perspective, the mild reaction conditions and the specific coordination environment provided by the pivalic acid additive help suppress common side reactions such as homocoupling or over-oxidation. This mechanistic precision results in a cleaner reaction profile, reducing the burden on purification processes and ensuring that the final API intermediates meet the stringent purity specifications required for clinical applications.
How to Synthesize Indolo[1,2-a]quinazolin-6(5H)-one Efficiently
The synthesis of these valuable heterocycles is streamlined through a one-pot procedure that combines all necessary reagents in a common organic solvent such as dioxane. The process begins by charging a reaction vessel with the cobalt catalyst, silver carbonate oxidant, and the specific 2-pyridinecarboxamide substrate, followed by the addition of the CO surrogate and additives. The mixture is then heated to a temperature range of 130-150°C, allowing the cascade of C-H activation and carbonylation to proceed to completion over a period of 20 to 40 hours. Detailed standardized synthetic steps for optimizing yield and purity are provided in the technical guide below.
- Combine cobalt chloride catalyst, silver carbonate oxidant, pivalic acid additive, triethylamine base, and the 2-pyridinecarboxamide derivative substrate in dioxane solvent.
- Add phenyl 1,3,5-tricarboxylate as the carbon monoxide substitute to the reaction mixture under stirring.
- Heat the reaction mixture to 130-150°C for 20-40 hours, then filter and purify the crude product via column chromatography to isolate the target indolo[1,2-a]quinazolin-6(5H)-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed technology translates into tangible strategic benefits regarding cost stability and supply continuity. The shift from precious palladium catalysts to base metal cobalt systems fundamentally alters the cost structure of the synthesis, removing exposure to the volatile pricing of noble metals. Additionally, the use of solid reagents instead of hazardous gases simplifies logistics and storage requirements, reducing the overhead associated with specialized safety compliance and handling protocols. This operational simplicity ensures a more robust supply chain capable of responding rapidly to fluctuating market demands for key pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive cobalt chloride results in a substantial decrease in raw material costs per kilogram of product. Moreover, the elimination of high-pressure carbon monoxide gas cylinders removes the need for costly autoclave reactors and specialized gas handling infrastructure, leading to significant capital expenditure savings. The simplified workup procedure, which often requires only filtration and standard chromatography, further reduces labor and solvent consumption costs, enhancing the overall economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, including cobalt chloride, silver carbonate, and phenyl formate derivatives, are commodity chemicals that are readily available from multiple global suppliers. This abundance mitigates the risk of supply disruptions that often plague processes dependent on specialized or single-source catalysts. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent batch-to-batch reproducibility and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work effectively from milligram to gram scales with consistent yields, indicating a clear path to multi-kilogram and ton-scale production. The avoidance of toxic CO gas and the use of less hazardous solvents align with green chemistry principles, simplifying waste treatment and environmental permitting processes. This environmental friendliness not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing operation, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis platform. These insights are derived directly from the experimental data and optimization studies detailed in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: What are the advantages of using cobalt over palladium for this synthesis?
A: Cobalt catalysts are significantly more cost-effective and earth-abundant compared to precious metal palladium catalysts. Furthermore, this cobalt-catalyzed protocol utilizes phenyl formate as a safe solid CO surrogate, eliminating the need for hazardous high-pressure carbon monoxide gas equipment often required in traditional carbonylation methods.
Q: What is the substrate scope of this cobalt-catalyzed reaction?
A: The method demonstrates excellent functional group tolerance, successfully accommodating substrates with electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as chloro and ester functionalities. This broad compatibility allows for the rapid generation of diverse libraries of indolo[1,2-a]quinazolin-6(5H)-one derivatives for SAR studies.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is designed for scalability. It employs commercially available reagents, operates at moderate temperatures (130-150°C), and utilizes a simple workup procedure involving filtration and standard column chromatography, making it highly viable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[1,2-a]quinazolin-6(5H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the race to bring life-saving medicines to market. Our team of expert chemists has extensively evaluated the cobalt-catalyzed C-H activation technology described in CN112321593B and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from laboratory discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of indolo[1,2-a]quinazolin-6(5H)-one intermediate delivered meets the highest quality standards.
We invite you to collaborate with us to leverage this advanced chemistry for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and cost-effective supply chain for your critical pharmaceutical intermediates.
