Advanced One-Step Synthesis of Trisubstituted Indoles for Commercial Pharmaceutical Manufacturing
Advanced One-Step Synthesis of Trisubstituted Indoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly indole derivatives which serve as critical cores in numerous bioactive molecules. Patent CN113121403B introduces a groundbreaking one-step methodology for the preparation of trisubstituted indole compounds, specifically targeting the synthesis of 2-sulfoximine-3,6-dibromoindole derivatives. This innovation represents a significant leap forward in process chemistry by eliminating the need for transition metal catalysts, which have traditionally been a bottleneck in terms of cost and purification complexity. The general structure of these high-value intermediates is depicted below, showcasing the versatile substitution patterns achievable through this novel route.
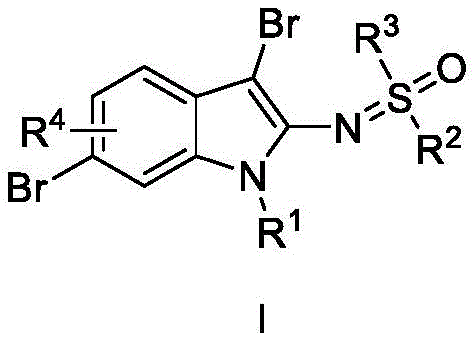
By leveraging a metal-free catalytic system, this technology addresses the growing demand for greener and more sustainable manufacturing processes in the production of high-purity pharmaceutical intermediates. The ability to introduce multiple functional groups simultaneously in a single operational step drastically reduces the number of unit operations required, thereby minimizing waste generation and energy consumption. For R&D directors and process chemists, this patent offers a robust alternative to classical indole functionalization strategies, promising higher atom economy and simplified downstream processing. The structural diversity allowed by the variable R groups ensures that this platform technology can be adapted for a wide array of drug discovery programs requiring specific indole-based building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct functionalization of the indole nucleus, especially at multiple positions, has relied heavily on transition metal catalysis involving precious metals such as palladium, rhodium, or ruthenium. These conventional methods often necessitate harsh reaction conditions, including elevated temperatures and prolonged reaction times, which can lead to thermal degradation of sensitive substrates and the formation of complex impurity profiles. Furthermore, the use of stoichiometric amounts of oxidants and additives frequently complicates the work-up procedure, requiring extensive chromatographic purification to remove trace metal residues that are strictly regulated in final active pharmaceutical ingredients. The economic burden of recovering or disposing of these expensive catalysts, combined with the environmental impact of heavy metal waste, poses significant challenges for procurement managers aiming to reduce the overall cost of goods sold in API manufacturing.
The Novel Approach
In stark contrast, the method disclosed in patent CN113121403B utilizes a unique two-component reaction between 1-substituted indoles and N-bromosulfoximines in acetonitrile at room temperature. As illustrated in the specific reaction scheme below, this approach achieves rapid tri-substitution within merely 5 minutes, demonstrating exceptional reaction efficiency without the need for any external catalysts or additives. The mild conditions preserve the integrity of the indole core while facilitating the introduction of bromine and sulfoximine moieties with high regioselectivity. This streamlined process not only accelerates the timeline from bench to kilogram scale but also significantly simplifies the isolation of the target compound, as the absence of metal catalysts removes the necessity for specialized scavenging resins or complex extraction protocols typically associated with organometallic chemistry.

Mechanistic Insights into Metal-Free C-H Functionalization
The underlying mechanism of this transformation relies on the inherent electrophilic nature of the N-bromosulfoximine reagent, which acts as both a brominating agent and a source of the sulfoximine functionality. Unlike traditional electrophilic aromatic substitutions that might require strong Lewis acids to activate the electrophile, the sulfur(VI) center in the N-bromosulfoximine possesses sufficient reactivity to engage directly with the electron-rich indole system. The reaction likely proceeds through an initial electrophilic attack at the C3 position of the indole, followed by subsequent functionalization at the C2 and C6 positions, driven by the specific electronic properties of the intermediate species formed in situ. This cascade-like behavior allows for the construction of a highly substituted scaffold in a single pot, bypassing the need for protecting group strategies or stepwise functionalization that would otherwise increase the step count and reduce overall yield.
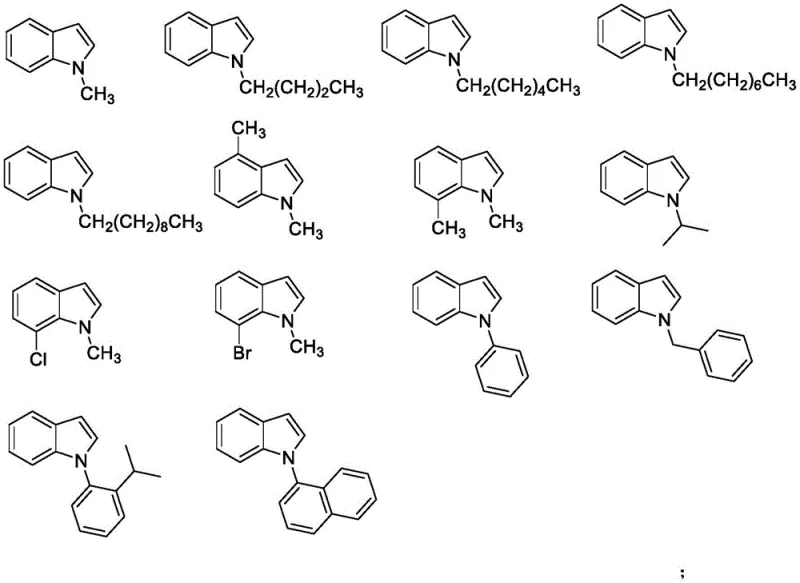
From an impurity control perspective, the simplicity of the reaction matrix is a major advantage for ensuring product quality. Since the reaction does not involve metal catalysts, the risk of metal-catalyzed side reactions such as homocoupling or over-oxidation is virtually eliminated. The primary by-products are likely derived from the decomposition of the sulfoximine reagent or minor regioisomers, which can be effectively separated using standard neutral alumina chromatography as described in the patent examples. The broad substrate scope, encompassing various N-alkyl and N-aryl indoles as shown in the substrate diversity chart, indicates that the electronic and steric demands of the mechanism are quite forgiving. This robustness is critical for commercial applications where feedstock variability must be accommodated without compromising the consistency of the final intermediate.
How to Synthesize Trisubstituted Indole Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant production. The protocol involves mixing the indole substrate and the N-bromosulfoximine reagent in acetonitrile, followed by a brief stirring period at ambient temperature. Monitoring the reaction progress is straightforward using thin-layer chromatography (TLC), with the disappearance of the starting indole serving as the endpoint indicator. Following the reaction, the solvent is removed under reduced pressure, and the crude product is purified via column chromatography. For detailed operational parameters and safety considerations regarding the handling of N-bromosulfoximines, please refer to the standardized synthesis guide below.
- Combine 1-substituted indole, N-bromosulfoximine, and acetonitrile in a dry pressure-resistant tube with a magnetic stirrer.
- Stir the mixture at room temperature (15-25°C) for approximately 5 minutes until TLC monitoring indicates the disappearance of the indole starting material.
- Remove the solvent via rotary evaporation and purify the crude residue using neutral alumina chromatography with a petroleum ether and ethyl acetate mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology offers tangible strategic benefits that extend beyond mere chemical novelty. The elimination of precious metal catalysts directly translates to a reduction in raw material costs and removes the logistical complexities associated with sourcing and managing hazardous metal wastes. Additionally, the use of acetonitrile, a common and relatively inexpensive solvent, alongside commercially available or easily synthesized N-bromosulfoximines, ensures a stable and reliable supply chain for the necessary reagents. The short reaction time of approximately 5 minutes implies a high throughput potential, allowing manufacturing facilities to maximize reactor utilization rates and significantly shorten the production cycle time for these valuable intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver for this technology is the complete avoidance of expensive transition metals like palladium and rhodium, which are subject to volatile market pricing and supply constraints. By removing the catalyst cost entirely and simplifying the purification process to a single column chromatography step, the overall cost of production is substantially lowered. Furthermore, the mild reaction conditions eliminate the energy costs associated with heating reactors to high temperatures or maintaining cryogenic conditions, contributing to a more energy-efficient manufacturing footprint that aligns with modern sustainability goals.
- Enhanced Supply Chain Reliability: The reliance on simple, non-specialized reagents enhances the resilience of the supply chain against disruptions. Since the method does not depend on proprietary ligands or sensitive organometallic complexes that may have long lead times or single-source suppliers, procurement teams can secure materials from multiple vendors with greater ease. The stability of the reagents, such as the yellow solid N-bromosulfoximines which can be stored in a refrigerator, further supports inventory management strategies by allowing for bulk purchasing and storage without significant degradation risks, ensuring continuous production capability.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the absence of exothermic hazards typically associated with rapid metal-catalyzed oxidations. The reaction operates safely at room temperature and atmospheric pressure (or slight pressure in sealed tubes), reducing the engineering controls required for scale-up. From an environmental compliance standpoint, the metal-free nature of the process simplifies wastewater treatment and reduces the regulatory burden related to heavy metal discharge limits. This green chemistry profile not only mitigates environmental risk but also enhances the marketability of the final pharmaceutical products to eco-conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-step trisubstituted indole synthesis. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the method's capabilities and limitations for potential licensees or manufacturing partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or new drug development pipelines.
Q: What are the primary advantages of this metal-free indole functionalization method?
A: The primary advantages include the elimination of expensive transition metal catalysts like palladium or rhodium, operation under mild room temperature conditions, and significantly reduced reaction times (approx. 5 minutes), which collectively lower production costs and simplify purification processes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes cheap and easily obtainable raw materials, avoids hazardous solvents, and operates under simple conditions without the need for specialized high-pressure or high-temperature equipment, making it highly suitable for commercial scale-up.
Q: What is the substrate scope for the indole starting materials?
A: The method demonstrates excellent substrate suitability, accommodating various N-substituents including alkyl, phenyl, benzyl, 2-isopropylphenyl, and naphthyl groups, as well as substituted indoles with methyl, chloro, or bromo groups on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in accelerating the delivery of complex pharmaceutical intermediates to the global market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in patent CN113121403B can be seamlessly transitioned from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of trisubstituted indole meets the exacting standards required for GMP-compliant API synthesis.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthesis route for their specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, as well as obtain specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain and reduce time-to-market with our advanced manufacturing capabilities and commitment to quality excellence.
