Advanced Gold-Catalyzed Synthesis of Imidazo[1,2-a]pyridine Intermediates for Commercial API Production
Advanced Gold-Catalyzed Synthesis of Imidazo[1,2-a]pyridine Intermediates for Commercial API Production
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly the imidazo[1,2-a]pyridine scaffold, which serves as the core structure for several blockbuster drugs. Patent CN112442025A introduces a groundbreaking methodology for synthesizing these valuable compounds through a gold-catalyzed intramolecular cyclization strategy. This innovation addresses critical limitations in existing manufacturing processes by utilizing a specialized gold complex to drive the aromatization of propargylamine precursors with exceptional efficiency. The significance of this chemical transformation extends beyond academic interest, directly impacting the supply chain stability for high-demand sedative-hypnotics and anxiolytics. As shown in the structural overview of key market drugs, the versatility of this scaffold is evident in its presence in molecules like Alpidem, Zolpidem, and Necopidem, underscoring the commercial necessity for reliable production methods. ![Structures of marketed drugs containing the imidazo[1,2-a]pyridine core such as Alpidem and Zolpidem](/insights/img/imidazo-pyridine-gold-catalysis-pharma-supplier-20260306090158-01.webp)
This patented process represents a significant leap forward in green chemistry principles applied to fine chemical manufacturing, specifically targeting the reduction of hazardous waste streams often associated with traditional heterocycle synthesis. By employing a catalytic amount of a gold species in conjunction with specific additives, the reaction achieves high atom economy, ensuring that the majority of starting material atoms are incorporated into the final product rather than lost as byproducts. The operational simplicity of the method, which avoids the need for exotic reagents or extreme pressure conditions, makes it an attractive candidate for technology transfer from laboratory discovery to kilogram-scale production. For procurement specialists and supply chain managers, understanding the nuances of this new pathway is essential for evaluating potential cost reductions and securing a stable supply of high-purity pharmaceutical intermediates. The following analysis dissects the technical merits and commercial implications of this gold-catalyzed approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the imidazo[1,2-a]pyridine ring system has relied heavily on multi-component coupling reactions that often suffer from harsh reaction conditions and limited substrate scope. A prominent example found in prior art involves the use of copper catalysts, such as cuprous chloride, combined with trifluoromethanesulfonic acid promoters to facilitate the cyclization of 2-aminopyridines with aldehydes and alkynes. These traditional protocols typically require elevated temperatures reaching 120°C and extended reaction times, which can lead to thermal degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, the reliance on stoichiometric amounts of acidic promoters generates significant quantities of salt waste, complicating downstream purification and increasing the environmental burden of the manufacturing process. The inability of these older methods to tolerate a wide range of electronic substituents often necessitates the development of bespoke synthetic routes for each new derivative, thereby inflating research and development costs and delaying time-to-market for new drug candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112442025A leverages the unique pi-acidity of gold catalysts to activate the alkyne moiety towards nucleophilic attack by the pyridine nitrogen, facilitating a smooth 5-endo-dig cyclization. This novel approach operates under significantly milder thermal conditions, typically between 80°C and 120°C, with a preferred operating temperature of 100°C, which minimizes energy consumption and reduces the risk of thermal runaway incidents in large-scale reactors. The use of a specialized gold catalyst system allows for the direct conversion of N-(1,3-diphenyl-2-alkynyl-1-yl)pyridin-2-amine substrates into the desired fused heterocycles with high selectivity. As illustrated in the general reaction scheme, this transformation proceeds efficiently in common organic solvents like 1,2-dichloroethane or toluene, eliminating the need for corrosive liquid acids. ![General reaction scheme for gold-catalyzed synthesis of imidazo[1,2-a]pyridine from alkynyl amine substrate](/insights/img/imidazo-pyridine-gold-catalysis-pharma-supplier-20260306090158-03.webp)
The versatility of this new synthetic route is further enhanced by its compatibility with a diverse array of substituents on the aromatic rings, including electron-donating methyl and methoxy groups as well as electron-withdrawing halogens. This broad substrate adaptability means that a single standardized protocol can be applied to synthesize a library of analogues, streamlining the process development workflow for medicinal chemists. The reaction times, ranging from 20 to 26 hours, are competitive with existing methods while offering superior control over the reaction outcome. By shifting from a brute-force acid-catalyzed condensation to a sophisticated transition-metal catalyzed cyclization, this invention provides a cleaner, more predictable, and economically viable pathway for producing high-value imidazo[1,2-a]pyridine intermediates required for the next generation of therapeutic agents.
Mechanistic Insights into AuLCl-Catalyzed Cyclization
The success of this synthetic transformation hinges on the precise design of the gold catalyst system, specifically the use of the AuLCl complex where L represents a bulky, electron-rich phosphine ligand. The steric bulk of the ligand, as depicted in the catalyst structure, plays a critical role in stabilizing the active gold(I) species and preventing the formation of inactive gold clusters that could deactivate the catalytic cycle. 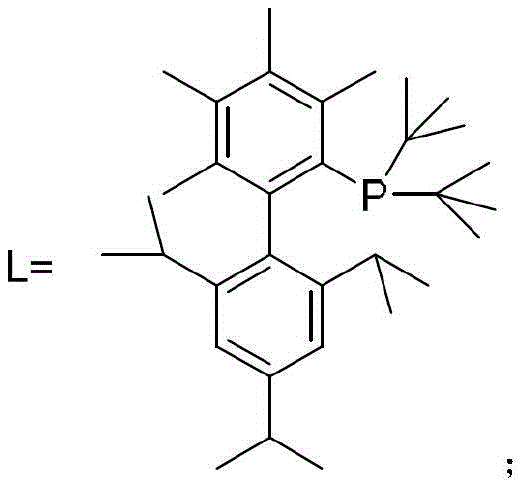
Upon activation by a silver or sodium salt additive, such as NaBArF4 or AgNTf2, the chloride ligand is abstracted to generate a highly electrophilic cationic gold center. This cationic gold species coordinates to the triple bond of the alkyne substrate, lowering the energy barrier for the nucleophilic attack by the adjacent pyridine nitrogen atom. The resulting vinyl-gold intermediate then undergoes protodeauration to release the final aromatic product and regenerate the active catalyst, completing the turnover cycle. This mechanism ensures that the gold catalyst is used in truly catalytic amounts, typically ranging from 1 to 5 mol%, which is crucial for minimizing the residual heavy metal content in the final API intermediate. The choice of additive is also pivotal, as non-coordinating anions like BArF4- or NTf2- ensure that the gold center remains sufficiently Lewis acidic to activate the alkyne without being poisoned by the solvent or substrate. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters for optimal yield and purity, ensuring that the final product meets the stringent quality standards required for pharmaceutical applications.
How to Synthesize 3-benzyl-2-phenylimidazo[1,2-a]pyridine Efficiently
The practical implementation of this gold-catalyzed protocol involves a straightforward procedure that can be easily adapted for both small-scale discovery and larger pilot plant operations. The process begins with the precise weighing of the N-(1,3-diphenyl-2-alkynyl-1-yl)pyridin-2-amine substrate, the AuLCl catalyst, and the appropriate additive, which are then suspended in a dry, degassed solvent to prevent catalyst deactivation by oxygen or moisture.
- Mix the N-(1,3-diphenyl-2-alkynyl-1-yl)pyridin-2-amine substrate with the AuLCl gold catalyst and a silver or borate additive in a suitable solvent like 1,2-dichloroethane.
- Heat the reaction mixture to a temperature range of 80-120°C, preferably maintaining 100°C, and allow the cyclization to proceed for 20 to 26 hours.
- Upon completion, cool the solution, add silica gel, remove the solvent under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this gold-catalyzed synthesis offers tangible benefits that extend beyond mere chemical elegance, directly addressing key pain points in the sourcing of complex heterocyclic intermediates. The shift towards a catalytic process with high atom economy inherently reduces the volume of raw materials required per unit of product, leading to a leaner and more cost-effective manufacturing footprint. By eliminating the need for stoichiometric amounts of strong acids and the associated neutralization steps, the process significantly simplifies the workup procedure, reducing the consumption of auxiliary chemicals and the generation of saline wastewater. This simplification translates into shorter batch cycles and lower utility costs, providing a competitive edge in the pricing of the final intermediate. Furthermore, the mild reaction conditions reduce the stress on reactor equipment, potentially extending the lifespan of capital assets and lowering maintenance overheads for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The utilization of a gold catalyst, while seemingly expensive on a per-gram basis, is offset by its high turnover number and the ability to use it in very low loadings, effectively distributing the catalyst cost across a large mass of product. The elimination of harsh acidic conditions removes the requirement for specialized corrosion-resistant equipment, allowing the reaction to be run in standard glass-lined or stainless steel reactors commonly available in multipurpose plants. Additionally, the simplified purification process, which often requires only standard silica gel chromatography or crystallization, reduces the demand for expensive preparative HPLC resources or complex extraction protocols. These factors combine to lower the overall cost of goods sold (COGS), making the final imidazo[1,2-a]pyridine derivatives more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method against variations in substrate electronics ensures a consistent supply of intermediates regardless of the specific substitution pattern required by the drug developer. Since the starting materials are readily accessible anilines and alkynes, the supply chain is not dependent on obscure or single-source reagents that could pose availability risks. The tolerance of the reaction to various functional groups means that late-stage functionalization strategies can be employed, allowing for greater flexibility in responding to changing clinical demands without needing to revalidate entirely new synthetic routes. This flexibility acts as a buffer against supply disruptions, ensuring that project timelines are met even when specific building blocks face temporary shortages.
- Scalability and Environmental Compliance: The absence of noxious gas evolution and the minimal generation of aqueous waste align perfectly with increasingly stringent environmental regulations governing pharmaceutical manufacturing. The use of common solvents like toluene or dichloroethane facilitates solvent recovery and recycling, further enhancing the sustainability profile of the process. From a scalability perspective, the exotherm of the reaction is manageable under the specified temperature range, reducing the risk of hot spots in large reactors and ensuring safe operation at the 100 kg to multi-ton scale. This safety profile, combined with the green chemistry metrics, makes the technology highly attractive for companies aiming to reduce their carbon footprint and meet corporate sustainability goals while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold-catalyzed synthesis for imidazo[1,2-a]pyridine production. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for technical teams.
Q: What are the advantages of using gold catalysis over traditional copper methods for this synthesis?
A: The gold-catalyzed method described in CN112442025A operates under milder conditions compared to traditional copper-catalyzed three-component couplings, offering better functional group tolerance and higher atom economy without generating significant waste gas or water.
Q: Is this synthetic route scalable for industrial API intermediate production?
A: Yes, the process utilizes standard solvents like 1,2-dichloroethane or toluene and moderate temperatures (80-120°C), which are highly amenable to scale-up in standard stainless steel reactors used for commercial pharmaceutical manufacturing.
Q: What types of substituents are tolerated on the pyridine and phenyl rings?
A: The method demonstrates excellent substrate adaptability, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens (fluorine, chlorine, bromine) at various positions on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazo[1,2-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new pharmaceutical therapies, and we are committed to delivering excellence in every batch. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every shipment of imidazo[1,2-a]pyridine derivatives meets the highest industry standards. Our dedication to quality assurance means that you can rely on us as a trusted partner for your most challenging synthetic projects, with a track record of delivering complex heterocycles on time and within specification.
We invite you to contact our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how optimizing your supply chain with our proven synthetic routes can reduce your overall development costs. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to transparency and technical excellence. Let us help you accelerate your drug development timeline with our reliable supply of high-purity pharmaceutical intermediates.
