Advanced Metal-Free Synthesis of 2-Ethyl-4,6-Disubstituted Pyridines for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and cost-effective synthetic routes for heterocyclic scaffolds, particularly pyridine derivatives which serve as critical building blocks in drug discovery. Patent CN111138346B introduces a groundbreaking methodology for the preparation of 2-ethyl-4,6-disubstituted pyridine compounds, addressing long-standing challenges in heterocyclic chemistry. This innovation utilizes a metal-free cycloaddition strategy between alkenyl imines and ketones, facilitated by a sodium hydride and tetrabutylammonium bisulfate system. Unlike traditional methods that rely on precious transition metals, this approach offers a streamlined pathway that significantly simplifies downstream processing and reduces environmental impact. For R&D directors and procurement specialists, this technology represents a pivotal shift towards greener, more economical manufacturing of high-purity pharmaceutical intermediates.
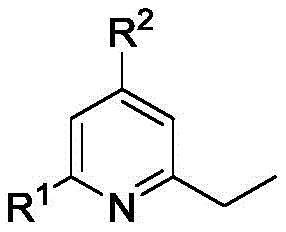
The significance of this patent lies in its ability to construct complex polysubstituted pyridine cores with excellent functional group tolerance. The structural versatility allows for various substituents at the 4 and 6 positions, including phenyl, methylphenyl, chlorophenyl, methoxyphenyl, fluorophenyl, furan, and pyridine groups. This broad substrate scope is essential for medicinal chemists aiming to explore structure-activity relationships (SAR) without being constrained by synthetic limitations. By leveraging this technology, manufacturers can access a diverse library of pyridine derivatives that are vital for developing next-generation therapeutics and crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted pyridines has been plagued by reliance on expensive and potentially toxic transition metal catalysts. For instance, earlier methodologies often employed ruthenium trichloride (RuCl3) catalyzed oxidation of 1,4-dihydropyridines or copper-catalyzed cycloadditions. 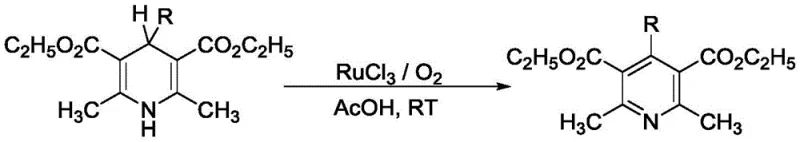 These conventional routes present substantial drawbacks for industrial application, primarily due to the high cost of the catalysts and the stringent requirements for removing residual metal impurities from the final product. In the context of pharmaceutical manufacturing, residual heavy metals are strictly regulated, necessitating additional purification steps such as scavenging or recrystallization, which inevitably increase production costs and reduce overall yield. Furthermore, some prior art methods suffer from poor regioselectivity or require harsh reaction conditions that limit the compatibility of sensitive functional groups, thereby restricting the chemical space available for exploration.
These conventional routes present substantial drawbacks for industrial application, primarily due to the high cost of the catalysts and the stringent requirements for removing residual metal impurities from the final product. In the context of pharmaceutical manufacturing, residual heavy metals are strictly regulated, necessitating additional purification steps such as scavenging or recrystallization, which inevitably increase production costs and reduce overall yield. Furthermore, some prior art methods suffer from poor regioselectivity or require harsh reaction conditions that limit the compatibility of sensitive functional groups, thereby restricting the chemical space available for exploration.
The Novel Approach
In stark contrast, the novel approach disclosed in CN111138346B eliminates the need for transition metals entirely, utilizing a base-mediated cycloaddition mechanism. This metal-free strategy not only drastically reduces the raw material costs associated with catalyst procurement but also simplifies the workup procedure, as there is no need for complex metal removal protocols. The reaction operates under mild conditions, typically at room temperature, which minimizes energy consumption and enhances operational safety compared to high-temperature or high-pressure alternatives. The use of readily available starting materials, specifically alkenyl imines and ketones, ensures a robust supply chain and facilitates rapid scale-up. This paradigm shift enables the efficient construction of the pyridine ring system with moderate to excellent yields, making it an attractive option for both laboratory research and commercial production.
Mechanistic Insights into Base-Mediated Cycloaddition
The core of this innovative synthesis lies in the base-mediated activation of the substrates to facilitate ring closure. The reaction mechanism involves the deprotonation of the ketone substrate by sodium hydride to generate a reactive enolate species, which subsequently undergoes nucleophilic attack on the alkenyl imine. 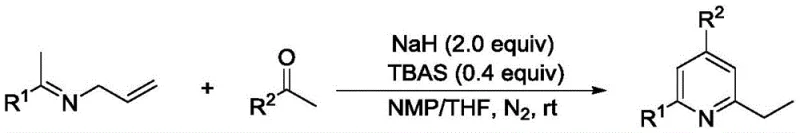 The presence of tetrabutylammonium bisulfate (TBAS) acts as a phase transfer catalyst, enhancing the solubility and reactivity of the ionic species in the organic solvent mixture of NMP and THF. This synergistic effect promotes the [3+3] cycloaddition process, leading to the formation of the dihydropyridine intermediate which spontaneously aromatizes to yield the final 2-ethyl-4,6-disubstituted pyridine product. Understanding this mechanistic pathway is crucial for optimizing reaction parameters and ensuring consistent quality across different batches.
The presence of tetrabutylammonium bisulfate (TBAS) acts as a phase transfer catalyst, enhancing the solubility and reactivity of the ionic species in the organic solvent mixture of NMP and THF. This synergistic effect promotes the [3+3] cycloaddition process, leading to the formation of the dihydropyridine intermediate which spontaneously aromatizes to yield the final 2-ethyl-4,6-disubstituted pyridine product. Understanding this mechanistic pathway is crucial for optimizing reaction parameters and ensuring consistent quality across different batches.
From an impurity control perspective, the metal-free nature of this reaction inherently reduces the risk of metal-catalyzed side reactions that often lead to complex impurity profiles. The primary byproducts are typically derived from incomplete conversion or minor competitive pathways, which are easily managed through standard chromatographic purification. The high selectivity of the base-mediated system ensures that the desired regioisomer is formed predominantly, simplifying the isolation process. For quality control teams, this translates to a cleaner crude product and higher purity specifications in the final API intermediate, aligning perfectly with the rigorous standards required by global regulatory bodies.
How to Synthesize 2-Ethyl-4,6-Disubstituted Pyridines Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and atmospheric conditions to maximize yield and reproducibility. The process begins with the preparation of a basic mixture containing sodium hydride and the phase transfer catalyst in a specific solvent ratio, followed by the controlled addition of substrates under an inert nitrogen atmosphere. Detailed standard operating procedures regarding mixing rates, temperature monitoring, and quenching protocols are essential for safe and effective execution. Adhering to these standardized steps ensures that the reaction proceeds smoothly to completion, typically within 14 hours, providing a reliable method for generating high-quality pyridine derivatives.
- Prepare a mixed solution of sodium hydride and tetrabutylammonium bisulfate in NMP/THF (3: 1) under nitrogen atmosphere.
- Add alkenyl imine and ketone substrates to the mixture using a micro-injector maintaining a molar ratio of 2: 1:1.3 for NaH:imine:ketone.
- Stir the reaction at room temperature for at least 14 hours until TLC indicates disappearance of starting materials, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits. The elimination of precious metal catalysts directly correlates to a significant reduction in raw material expenditure, as sodium hydride and ammonium salts are commoditized chemicals with stable pricing and widespread availability. Furthermore, the simplified purification process reduces the consumption of solvents and adsorbents, contributing to lower waste disposal costs and a smaller environmental footprint. This efficiency gain is critical for maintaining competitiveness in the global fine chemical market.
- Cost Reduction in Manufacturing: The absence of expensive transition metals like ruthenium or copper removes a major cost driver from the bill of materials. Additionally, the mild reaction conditions at room temperature reduce energy costs associated with heating or cooling reactors. The streamlined workup procedure minimizes labor hours and equipment usage, leading to substantial overall cost savings in the manufacturing of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as acetophenones and alkenyl imines ensures a stable supply chain不受 geopolitical fluctuations affecting rare metal markets. The robustness of the reaction conditions allows for flexible production scheduling and reduces the risk of batch failures due to sensitive catalyst handling. This reliability is paramount for meeting tight delivery deadlines and maintaining continuous supply to downstream customers.
- Scalability and Environmental Compliance: The metal-free nature of the process simplifies regulatory compliance regarding heavy metal residues, facilitating faster approval times for new drug applications. The reduced generation of hazardous waste and the use of common organic solvents make this process more environmentally sustainable. Scalability is enhanced by the straightforward reaction setup, allowing for seamless transition from gram-scale laboratory synthesis to ton-scale commercial production without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN111138346B operates under metal-free conditions using sodium hydride and a phase transfer catalyst, eliminating the need for costly ruthenium or copper catalysts.
Q: What are the typical reaction conditions for this pyridine synthesis?
A: The reaction proceeds under mild conditions at room temperature in a mixture of NMP and THF solvents, requiring approximately 14 hours for completion as monitored by TLC.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of readily available raw materials, mild reaction conditions, and the absence of toxic heavy metals make this process highly suitable for commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ethyl-4,6-Disubstituted Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis technology described in CN111138346B for producing high-value pyridine intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality and supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-ethyl-4,6-disubstituted pyridine meets the highest industry standards.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced synthetic route for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to obtain specific COA data and route feasibility assessments, and let us help you optimize your supply chain with cost-effective, high-purity chemical solutions.
