Revolutionizing Ortho-Cyanation: A Green Transition Metal Catalysis Strategy for Commercial Scale
Revolutionizing Ortho-Cyanation: A Green Transition Metal Catalysis Strategy for Commercial Scale
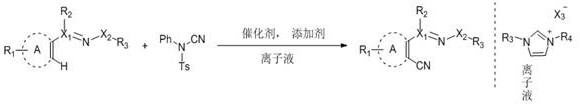
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the synthesis of high-value pharmaceutical intermediates. A groundbreaking approach detailed in patent CN108690007B introduces a highly efficient transition metal-catalyzed C-H coupling reaction that utilizes N1,N3-disubstituted imidazole-type ionic liquids as the reaction medium. This innovation addresses critical bottlenecks in the production of ortho-cyanated aromatic and unsaturated aliphatic ring compounds, which are ubiquitous scaffolds in modern medicinal chemistry. By replacing hazardous traditional solvents with recyclable ionic liquids and employing N-cyano-N-phenyl-p-toluenesulfonamide as a benign cyanide source, this technology offers a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their process chemistry. The method operates under remarkably mild conditions, often at room temperature, eliminating the need for energy-intensive heating protocols and enhancing the overall safety profile of the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group onto an aromatic ring has relied on classical transformations such as the Rosenmund-von Braun reaction or the Sandmeyer reaction, both of which present significant operational and environmental challenges. These traditional pathways typically necessitate the use of stoichiometric amounts of highly toxic copper(I) cyanide (CuCN), posing severe health risks to personnel and creating substantial hazardous waste streams that require costly disposal procedures. Furthermore, alternative cross-coupling strategies involving aromatic halides demand rigorous pre-activation of the substrate, adding extra synthetic steps that reduce overall atom economy and increase production lead times. The reliance on highly polar, toxic organic solvents and expensive transition metal ligands in existing C-H activation methods further exacerbates the cost burden, making large-scale cost reduction in pharmaceutical intermediates manufacturing difficult to achieve. Additionally, many conventional protocols require harsh reaction conditions, such as elevated temperatures or strictly anaerobic environments, which complicate reactor design and limit the functional group tolerance necessary for complex molecule synthesis.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages the unique physicochemical properties of ionic liquids to facilitate a direct C-H cyanation that bypasses the need for substrate pre-functionalization. By utilizing N-cyano-N-phenyl-p-toluenesulfonamide as a solid, stable, and low-toxicity cyanating agent, the process eliminates the handling risks associated with gaseous HCN or soluble metal cyanides. The reaction proceeds efficiently at room temperature within an ionic liquid matrix, which not only solubilizes the reactants effectively but also stabilizes the cationic organometallic intermediates formed during the catalytic cycle. This approach allows for the rapid construction of molecular libraries containing ortho-cyanated derivatives with excellent yields and high regioselectivity. Moreover, the ability to recycle the ionic liquid solvent and the catalyst system multiple times without replenishment represents a transformative advantage for industrial scalability, directly addressing the economic and environmental pain points of legacy synthesis routes.

Mechanistic Insights into Transition Metal-Catalyzed C-H Activation
The core of this technological advancement lies in the sophisticated interplay between the transition metal catalyst, typically a rhodium, ruthenium, or cobalt complex, and the directing groups present on the substrate. The mechanism initiates with the coordination of the metal center to a heteroatom (such as nitrogen in pyridyl or triazolyl rings), which directs the activation of the proximal C-H bond through a concerted metalation-deprotonation (CMD) pathway or oxidative addition. The ionic liquid environment plays a pivotal role here, as its high polarity and potential for hydrogen bonding can stabilize the charged transition states, thereby lowering the activation energy barrier for the C-H cleavage step. Once the metallacycle is formed, the N-cyano-N-phenyl-p-toluenesulfonamide acts as an electrophilic cyanide source, transferring the cyano group to the metal center followed by reductive elimination to release the ortho-cyanated product. This catalytic cycle is highly efficient, requiring only low catalyst loadings (0.01 to 0.20 molar equivalents) to drive the reaction to completion, demonstrating exceptional turnover numbers that are critical for high-purity pharmaceutical intermediates production where metal residue limits are stringent.
From an impurity control perspective, the specificity of the C-H activation ensures that side reactions such as poly-cyanation or attack at non-directed positions are minimized, resulting in a cleaner crude reaction profile. The use of silver additives, such as silver acetate or silver bistrifluoromethylsulfonylimide, serves to abstract halide ligands from the metal precursor, generating the active cationic species necessary for the catalytic cycle while simultaneously scavenging potential halide impurities. The robustness of the ionic liquid system also prevents the decomposition of sensitive functional groups that might occur in protic or strongly nucleophilic organic solvents. Consequently, the final product exhibits a superior purity profile, reducing the burden on downstream purification processes like chromatography or crystallization. This level of control is essential for ensuring the quality of commercial scale-up of complex pharmaceutical intermediates, where consistency and reproducibility are paramount for regulatory compliance.
How to Synthesize Ortho-Cyanated Derivatives Efficiently
The practical implementation of this synthesis route is designed for straightforward operation, making it accessible for both laboratory-scale optimization and pilot plant production. The protocol involves a simple one-pot procedure where the substrate, cyanating agent, catalyst, additive, and ionic liquid are combined in a standard reactor. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and workup procedures required to achieve the reported high yields. This simplicity reduces the training burden on operators and minimizes the risk of human error during batch preparation. The mild reaction conditions mean that standard glass-lined or stainless steel reactors can be used without the need for specialized high-pressure or high-temperature equipment, further lowering the capital expenditure required for adoption.
- Charge a clean reactor with the aromatic heterocycle or alkenyl substrate, N-cyano-N-phenyl-p-toluenesulfonamide as the cyanide source, a rhodium or ruthenium catalyst, a silver additive, and an imidazole-based ionic liquid solvent.
- Stir the reaction mixture at room temperature for approximately 24 hours to allow the C-H activation and subsequent cyanation coupling to proceed to completion under mild conditions.
- Extract the product using cyclohexane, separate the organic layer, remove the solvent under reduced pressure, and purify the residue via silica gel column chromatography while recycling the ionic liquid layer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-based cyanation technology offers compelling strategic benefits that extend beyond mere technical feasibility. The primary value driver is the drastic simplification of the supply chain for raw materials; by eliminating the need for pre-halogenated starting materials, manufacturers can source cheaper, more abundant bulk chemicals, thereby insulating the production process from volatility in the pricing of specialized halogenated intermediates. Furthermore, the replacement of toxic volatile organic solvents with non-flammable ionic liquids significantly reduces the costs associated with solvent recovery, ventilation, and hazardous waste disposal, leading to substantial operational expenditure savings. The enhanced safety profile also translates to lower insurance premiums and reduced regulatory compliance burdens, making the facility more resilient to audits and inspections. Ultimately, this process enables a more agile and cost-effective manufacturing model that can respond quickly to market demands for key drug building blocks.
- Cost Reduction in Manufacturing: The economic viability of this process is heavily underpinned by the recyclability of the reaction medium. Since the ionic liquid and the transition metal catalyst can be recovered and reused for multiple cycles without a noticeable decline in performance, the consumption of expensive noble metals like rhodium and ruthenium is minimized. This closed-loop system effectively decouples production volume from raw material consumption, driving down the variable cost per kilogram of the final product. Additionally, the avoidance of stoichiometric copper salts and the reduction in purification steps due to higher selectivity contribute to a leaner cost structure. These factors collectively ensure that the cost reduction in pharmaceutical intermediates manufacturing is realized through fundamental process intensification rather than superficial margin squeezing.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on hazardous reagents that face strict transportation and storage regulations. By utilizing solid, stable cyanating agents and non-volatile solvents, this method mitigates the risk of supply disruptions caused by logistics constraints or regulatory bans on toxic substances. The mild reaction conditions also mean that production is less susceptible to utilities failures, such as steam or cooling water interruptions, ensuring consistent output even in challenging operational environments. This reliability is crucial for maintaining the steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers, fostering stronger long-term partnerships and trust within the value chain.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new engineering challenges, but the homogeneous nature of this ionic liquid system facilitates smooth scale-up from gram to ton quantities. The absence of gas evolution and the low vapor pressure of the solvent simplify reactor pressure management and venting requirements, allowing for larger batch sizes without proportional increases in safety infrastructure. From an environmental standpoint, the process aligns perfectly with green chemistry principles by reducing E-factors and eliminating the release of volatile organic compounds (VOCs) into the atmosphere. This alignment not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the company as a sustainable and responsible reliable pharmaceutical intermediate supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-H cyanation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when integrating this methodology into their production workflows. Understanding these nuances is vital for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of using ionic liquids over traditional organic solvents in this cyanation reaction?
A: Ionic liquids offer superior thermal and chemical stability, negligible vapor pressure which enhances operational safety, and excellent solubility for polar intermediates. Crucially, they enable the recycling of the catalytic system without significant loss of activity, drastically reducing waste generation compared to volatile organic compounds.
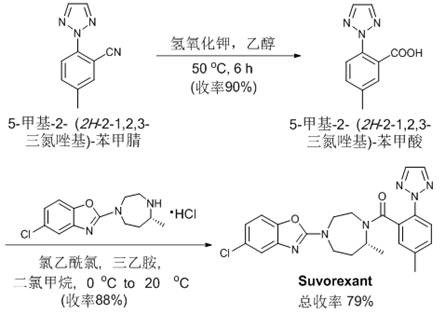
Q: Can this method be applied to the synthesis of complex drug molecules like Suvorexant?
A: Yes, the patent explicitly demonstrates the utility of the resulting ortho-cyanated products as key precursors for pharmaceuticals. Specifically, the cyano group generated can be derivatized into carboxyl or heterocyclic functionalities required for the synthesis of the anti-insomnia drug Suvorexant, bypassing lengthy traditional synthetic routes.
Q: Is the catalyst system reusable, and how does this impact production costs?
A: The catalytic system, comprising the transition metal and the ionic liquid solvent, can be recycled and reused for at least five cycles without the need for fresh additives. This reusability significantly lowers the cost of goods sold (COGS) by minimizing the consumption of expensive noble metal catalysts and reducing solvent disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Cyanated Aromatic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN108690007B can be faithfully reproduced on an industrial scale. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of ortho-cyanated intermediates meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and consistency makes us the ideal partner for companies looking to secure a stable supply of these critical building blocks.
We invite you to explore how this advanced cyanation technology can optimize your specific project requirements and deliver tangible value to your organization. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timelines and enhance your competitive edge in the market.
