Scalable Synthesis of 2,5-Disubstituted Thiophenes for Advanced Material and Pharma Applications
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the dual demands of environmental sustainability and economic efficiency in the fine chemical sector. A significant breakthrough in this domain is documented in Chinese Patent CN102584784A, which outlines a novel preparation method for 2,5-disubstituted thiophene compounds. These sulfur-containing conjugated aromatic structures are pivotal building blocks in the manufacture of active pharmaceutical ingredients (APIs), agrochemicals, and advanced optoelectronic materials such as OLEDs and liquid crystals. The patent introduces a streamlined approach that bypasses traditional limitations by utilizing readily available inorganic sodium salts, specifically sodium hydrosulfide (NaHS), sodium sulfide (Na2S), or sodium sulfide hydrate (Na2S·9H2O), reacting directly with 1,3-diyne compounds. This innovation represents a paradigm shift from complex, multi-step protocols to a more atom-economical and operationally simple process, offering substantial value to manufacturers seeking reliable supply chains for high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiophene derivatives has relied heavily on classical methodologies such as the Paal-Knorr reaction, which involves the sulfuration of 1,4-diketone compounds. While effective, these traditional routes often necessitate the use of hazardous and malodorous sulfurizing agents like phosphorus pentasulfide (P2S5) or Lawesson's reagent, posing significant safety and environmental disposal challenges for large-scale production facilities. Furthermore, alternative methods involving transition metal catalysis, such as copper-catalyzed reactions between diiodo-alkadienes and potassium sulfide, introduce the critical issue of metal residue contamination. For pharmaceutical and electronic grade applications, removing trace heavy metals to meet stringent regulatory specifications requires additional purification steps, such as scavenging or extensive chromatography, which drastically increases production costs and extends lead times. The preparation of the 1,4-diketone precursors themselves can also be synthetically tedious, adding further complexity to the overall manufacturing workflow.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in the patent leverages a direct cyclization strategy using 1,3-diyne compounds and inexpensive inorganic sulfides in a polar aprotic solvent system. By eliminating the need for transition metal catalysts entirely, this process inherently produces a cleaner crude product profile, significantly reducing the burden on downstream purification units. The reaction conditions are remarkably mild, typically proceeding efficiently at temperatures between 0°C and 80°C, which minimizes energy consumption and thermal degradation risks associated with sensitive functional groups. Moreover, the versatility of this approach allows for the successful synthesis of both symmetric and asymmetric 2,5-disubstituted thiophenes, accommodating a wide range of substituents including alkyl, aryl, and heteroaryl groups. This flexibility makes it an ideal candidate for the cost reduction in electronic chemical manufacturing, where diverse molecular architectures are often required to tune material properties.
Mechanistic Insights into Catalyst-Free Thiophene Cyclization
The core of this technological advancement lies in the unique reactivity of the 1,3-diyne system towards nucleophilic attack by sulfide anions. Unlike standard alkynes, the conjugated diyne structure possesses enhanced electrophilicity at the internal carbons, facilitating the initial addition of the sulfur species without the need for external activation by Lewis acids or transition metals. In the preferred solvent system, typically N,N-dimethylformamide (DMF), the sodium salt dissociates effectively to provide a high concentration of reactive sulfide ions. These ions attack the triple bond of the 1,3-diyne substrate, initiating a cascade of intramolecular cyclization events that rapidly construct the thiophene ring. The absence of a metal catalyst not only simplifies the reaction mechanism but also ensures that the final product is free from metal impurities, a critical quality attribute for clients requiring high-purity OLED material or pharmaceutical intermediates. The reaction pathway is highly selective, favoring the formation of the 2,5-disubstituted isomer with minimal side products, thereby maximizing atom economy.
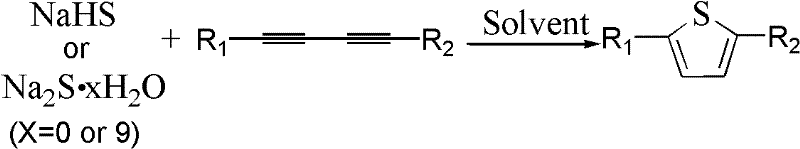
Controlling the impurity profile in this synthesis is largely managed through the choice of solvent and reaction stoichiometry. Experimental data indicates that while various solvents like THF, toluene, and acetonitrile were screened, DMF provided superior solubility for both the organic diyne substrate and the inorganic salt, leading to homogeneous reaction conditions and higher yields. The patent highlights that maintaining a specific molar ratio between the 1,3-diyne and the sodium salt, preferably between 1:0.5 and 1:3, is crucial for driving the reaction to completion while minimizing the formation of polymeric byproducts or unreacted starting materials. Furthermore, the mild thermal conditions prevent the decomposition of sensitive substituents, ensuring that functional groups such as methoxy, bromo, or long-chain alkyls remain intact throughout the cyclization process. This robustness allows for the commercial scale-up of complex polymer additives and liquid crystal precursors without compromising structural integrity.
How to Synthesize 2,5-Disubstituted Thiophenes Efficiently
Implementing this synthesis route in a production environment requires careful attention to the sequential addition of reagents and the control of reaction parameters to ensure reproducibility and safety. The process begins with the preparation of a dry reaction vessel to prevent moisture interference, followed by the dissolution of the 1,3-diyne compound in the selected aprotic solvent. The inorganic sulfur source is then introduced, and the mixture is agitated under controlled thermal conditions until monitoring indicates full conversion. The simplicity of the workup procedure, involving standard aqueous quenching and organic extraction, further enhances its appeal for industrial adoption. For detailed operational parameters and specific stoichiometric recommendations tailored to your specific substrate, please refer to the standardized synthesis guide below.
- In a dry reaction vessel, sequentially add an aprotic organic solvent such as DMF, the 1,3-diyne substrate, and an inorganic sodium salt like NaHS or Na2S.
- Stir the reaction mixture at a controlled temperature ranging from 0°C to 80°C for a duration between 10 minutes and 48 hours until completion.
- Quench the reaction with saturated brine, extract the product with an organic solvent like ether or ethyl acetate, and purify via chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this catalyst-free methodology offers compelling advantages that directly address common pain points in the global supply chain for fine chemicals. The primary driver for cost optimization is the substitution of expensive, specialized sulfurizing reagents and precious metal catalysts with commodity-grade inorganic salts. Sodium sulfide and sodium hydrosulfide are produced on a massive industrial scale, ensuring stable pricing and abundant availability, which shields manufacturers from the volatility often seen in the market for organometallic catalysts. Additionally, the elimination of metal catalysts removes the necessity for costly metal scavenging resins and the associated validation testing required to prove low residual metal levels in the final API or material. This streamlining of the purification process translates into significant cost savings in manufacturing overhead and reduces the overall cycle time from raw material intake to finished goods release.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived principally from the drastic simplification of the reagent profile and the reduction in waste treatment costs. By avoiding the use of toxic phosphorus-based reagents or expensive transition metals, the facility avoids the high expenses associated with hazardous waste disposal and specialized containment infrastructure. The high atom economy of the reaction means that a larger proportion of the raw material mass is converted into the desired product, reducing the cost per kilogram of the active intermediate. Furthermore, the ability to run the reaction at near-ambient temperatures significantly lowers energy consumption compared to high-temperature reflux processes, contributing to a lower carbon footprint and reduced utility bills for the production site.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of universally available raw materials that are not subject to the geopolitical restrictions or supply bottlenecks often affecting specialized catalytic ligands. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating market demand without lengthy campaign changeovers. Since the process does not rely on sensitive catalysts that may degrade upon storage or require inert atmosphere handling, the logistics of raw material storage and transport are simplified, reducing the risk of production delays due to material spoilage. This reliability is essential for maintaining just-in-time delivery schedules for downstream customers in the pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: Scaling this chemistry from laboratory benchtop to multi-ton commercial production is facilitated by the homogeneous nature of the reaction mixture and the absence of exothermic hazards typically associated with strong oxidizers or reactive metal powders. The straightforward workup involving liquid-liquid extraction is easily adaptable to large-scale separation equipment, ensuring consistent product quality across different batch sizes. From an environmental compliance standpoint, the process generates significantly less hazardous waste, aligning with increasingly strict global regulations on chemical manufacturing emissions. The use of DMF, while requiring proper recovery systems, is a well-understood solvent in the industry, and the lack of heavy metals simplifies the effluent treatment process, making it easier to obtain and maintain environmental operating permits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiophene synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these details is crucial for R&D teams assessing process robustness and procurement teams evaluating total cost of ownership.
Q: Does this thiophene synthesis method require transition metal catalysts?
A: No, the patented process described in CN102584784A operates without transition metal catalysts. It utilizes inexpensive inorganic sodium salts such as sodium hydrosulfide or sodium sulfide, which eliminates the risk of heavy metal contamination in the final pharmaceutical or electronic grade product.
Q: What is the typical yield range for this cyclization reaction?
A: The method demonstrates robust efficiency with yields generally ranging from 72% to 99% for various substituted substrates. Optimal conditions using DMF as the solvent at temperatures between 25°C and 80°C consistently provide high conversion rates.
Q: Can this method produce asymmetric thiophene derivatives?
A: Yes, the protocol is highly versatile and specifically supports the synthesis of asymmetric 2,5-disubstituted thiophenes. This capability is critical for developing specialized liquid crystal materials and functionalized organic semiconductors where precise structural control is required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Disubstituted Thiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch is free from critical impurities and residual metals. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this catalyst-free cyclization, guaranteeing a consistent supply of high-purity 2,5-disubstituted thiophenes for your most demanding applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data for our existing inventory and to request route feasibility assessments for custom derivatives, ensuring that your project timelines are met with precision and reliability.
