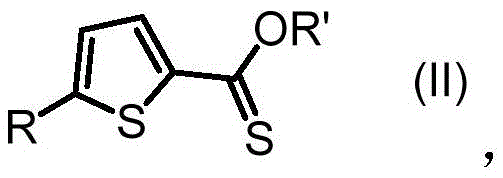
Scalable Synthesis of 2-Thiophene Thiocarboxylates via Direct Ring Expansion for Advanced Intermediates
Introduction to Patent CN112375065A
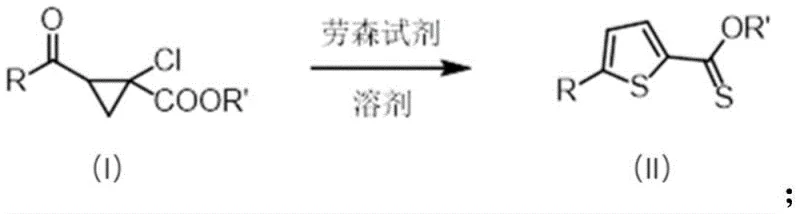
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct heterocyclic scaffolds, particularly thiophene derivatives which serve as critical building blocks for bioactive molecules and advanced electronic materials. Patent CN112375065A introduces a groundbreaking methodology for the preparation of 2-thiophene thiocarboxylate derivatives, specifically addressing the limitations of traditional multi-step syntheses. This innovation centers on a direct, one-pot transformation where 2-acyl-1-chlorocyclopropylcarboxylates undergo a ring-expansion and thionation reaction upon contact with Lawesson's reagent. The significance of this patent lies in its ability to generate complex thiophene structures under remarkably mild conditions, utilizing readily available reagents while maintaining high atom economy. By bypassing the need for harsh sulfurization agents or sensitive metal catalysts, this process offers a robust alternative for the commercial scale-up of complex pharmaceutical intermediates. The technology described herein not only streamlines the synthetic route but also enhances the overall safety profile of the manufacturing process, making it an attractive option for reliable agrochemical intermediate supplier networks and pharmaceutical manufacturers alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted thiophene rings has relied heavily on classical condensation reactions such as the Gewald reaction, Paal-Knorr synthesis, or various metal-catalyzed cross-coupling strategies. These conventional methods often suffer from significant drawbacks that hinder their efficiency in large-scale cost reduction in pharmaceutical intermediates manufacturing. For instance, many traditional routes require the use of elemental sulfur or inorganic sulfides under strongly alkaline conditions, which can lead to poor functional group tolerance and difficult waste treatment protocols. Furthermore, metal-catalyzed approaches frequently necessitate the use of expensive noble metals like palladium or rhodium, introducing substantial raw material costs and creating stringent regulatory hurdles regarding residual metal limits in final drug substances. Additionally, these older methods often involve multiple synthetic steps to install the necessary functionality, resulting in cumulative yield losses and increased processing time. The sensitivity of many thiophene precursors to oxidation also mandates the use of rigorous inert atmosphere techniques, adding complexity and equipment costs to the production line.
The Novel Approach
In stark contrast, the methodology disclosed in CN112375065A utilizes a unique ring-opening and closing mechanism driven by Lawesson's reagent to directly convert cyclopropane precursors into the desired thiophene system. This novel approach fundamentally simplifies the synthetic landscape by consolidating what would traditionally be multiple steps into a single thermal transformation. The reaction proceeds smoothly in common organic solvents such as toluene or xylene at temperatures ranging from 50°C to 200°C, with optimal results often observed around 110°C. A key advantage of this system is its insensitivity to atmospheric oxygen, allowing the reaction to proceed without the absolute necessity for degassed solvents or nitrogen blanketing, which drastically reduces operational overhead. The versatility of the method is demonstrated by its compatibility with a wide array of substituents on the aromatic ring, including halogens, alkoxy groups, and esters, enabling the rapid generation of diverse chemical libraries. This direct access to high-purity OLED material precursors and pharmaceutical intermediates represents a significant leap forward in process chemistry efficiency.
Mechanistic Insights into Lawesson's Reagent Mediated Cyclization
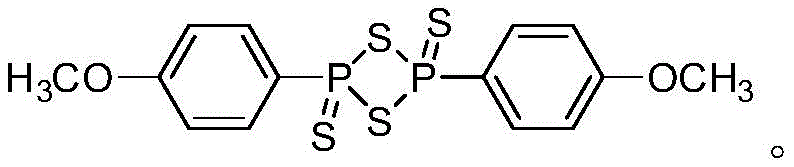
The core of this transformative process lies in the reactivity of Lawesson's reagent, a dimeric organophosphorus compound known for its ability to convert carbonyl groups into thiocarbonyls. In the context of this patent, the reagent acts not merely as a thionating agent but as a facilitator for a complex rearrangement cascade. The reaction initiates with the interaction between the carbonyl oxygen of the acyl-cyclopropane substrate and the phosphorus-sulfur centers of the reagent. This interaction weakens the strained carbon-carbon bonds of the cyclopropane ring, promoting a ring-opening event that generates a reactive intermediate capable of intramolecular cyclization. The presence of the chlorine atom on the cyclopropane ring further assists in this process by acting as a leaving group or stabilizing the transition state during the ring expansion. As the system rearranges, the sulfur atoms from the Lawesson's reagent are incorporated into the newly forming heterocyclic ring, ultimately yielding the stable aromatic thiophene structure. This mechanism avoids the formation of unstable radical species often seen in elemental sulfur reactions, thereby minimizing the generation of polymeric byproducts and tars that complicate purification.
From an impurity control perspective, this mechanism offers distinct advantages for reducing lead time for high-purity pharmaceutical intermediates. Because the reaction pathway is highly specific and driven by the thermodynamic stability of the aromatic thiophene product, side reactions are minimized. The patent data indicates that the reaction mixture remains homogeneous during the process, with the product precipitating or remaining soluble depending on the specific solvent choice, allowing for straightforward isolation. Unlike metal-catalyzed couplings which can produce homocoupling byproducts or dehalogenated impurities, this thionation-cyclization route produces a cleaner crude profile. The ability to purify the final product simply by recrystallization or short-path filtration, as evidenced by the experimental examples, suggests that the impurity spectrum is narrow and manageable. This level of control is critical for meeting the stringent quality specifications required by global regulatory bodies for active pharmaceutical ingredients.
How to Synthesize 2-Thiophene Thiocarboxylate Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory scale optimization and pilot plant production. The process begins by dissolving the 2-acyl-1-chlorocyclopropylcarboxylate starting material in an anhydrous solvent, with toluene being the preferred medium due to its favorable boiling point and solubility characteristics. Lawesson's reagent is then added in a molar ratio ranging from 1:1 to 1:10 relative to the substrate, with examples showing excellent results using 5 equivalents. The mixture is heated, typically to 110°C, and stirred for a duration of 1 to 20 hours depending on the specific electronic nature of the substituents. Monitoring via TLC ensures complete conversion before the mixture is cooled and processed. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined below to ensure reproducibility.
- Dissolve the 2-acyl-1-chlorocyclopropylcarboxylate substrate (Formula I) in an anhydrous aromatic solvent such as toluene or xylene under ambient conditions.
- Add Lawesson's reagent (1.25 to 5 equivalents) to the reaction mixture and heat to a temperature range of 50°C to 200°C, typically refluxing at 110°C.
- Maintain stirring for 1 to 20 hours until TLC indicates complete conversion, then isolate the product (Formula II) via filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of transition metal catalysts removes a major cost driver and supply chain bottleneck, as the pricing and availability of noble metals can be volatile. Furthermore, the simplified workup procedure, which often avoids labor-intensive column chromatography in favor of recrystallization, significantly reduces processing time and solvent consumption. This efficiency gain allows for faster batch turnover and lower utility costs per kilogram of product. The robustness of the reaction conditions, particularly its tolerance to oxygen, reduces the risk of batch failure due to equipment leaks or operator error, thereby enhancing overall supply reliability. These factors collectively contribute to a more resilient and cost-effective manufacturing framework for high-value heterocyclic intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the substitution of expensive metal catalysts with Lawesson's reagent, which is a commodity chemical available at a fraction of the cost of palladium or rhodium complexes. Additionally, the high yields reported, often exceeding 80% and reaching up to 91% in optimized examples, mean that less raw material is wasted, directly improving the cost of goods sold (COGS). The ability to use inexpensive solvents like toluene, which can be easily recovered and recycled, further drives down operational expenditures. By avoiding complex purification steps, the labor and energy costs associated with downstream processing are also substantially reduced, creating a leaner production model.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials and reagents mitigates the risk of supply disruptions that often plague specialized catalytic systems. Since the reaction does not require ultra-dry or oxygen-free environments to succeed, the logistical burden on the manufacturing facility is lightened, allowing for more flexible scheduling and reduced downtime. The scalability of the process from gram to kilogram scales without significant re-optimization ensures that supply can be ramped up quickly to meet market demand. This reliability is crucial for maintaining continuous production lines for downstream API synthesis, preventing costly delays in the broader pharmaceutical supply chain.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this process offers a greener alternative to traditional thiophene synthesis. The absence of heavy metals simplifies waste stream treatment and reduces the environmental footprint of the manufacturing site. The use of closed systems at moderate temperatures minimizes the release of volatile organic compounds (VOCs) compared to high-temperature pyrolysis methods. Furthermore, the high atom economy of the transformation means less chemical waste is generated per unit of product, aligning with modern green chemistry principles and facilitating easier compliance with increasingly strict environmental regulations. This sustainability profile enhances the long-term viability of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within patent CN112375065A, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using Lawesson's reagent for thiophene synthesis compared to metal catalysis?
A: The use of Lawesson's reagent eliminates the need for expensive transition metal catalysts like palladium or rhodium, significantly reducing raw material costs and removing the regulatory burden of heavy metal residue testing in pharmaceutical intermediates. Furthermore, the reaction is insensitive to atmospheric oxygen, simplifying operational requirements by negating the need for rigorous inert gas protection.
Q: What yields can be expected from this cyclopropane ring-expansion methodology?
A: Experimental data from the patent indicates robust yields generally exceeding 60%, with optimized conditions frequently achieving yields above 80% and even reaching 91% for specific substrates like ethyl-(4-methoxyphenyl)-2-thiophenecarboxylate. This consistency supports reliable commercial production planning.
Q: Is the purification process scalable for industrial manufacturing?
A: Yes, the process is highly scalable because the reaction mixture can often be purified through simple recrystallization or short-column filtration rather than complex chromatographic separations. The use of common solvents like toluene further facilitates solvent recovery and recycling in a plant setting.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Thiophene Thiocarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocyclic synthesis plays in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the technology described in CN112375065A and possesses the capability to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to plant floor is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of 2-thiophene thiocarboxylate meets the highest international standards for pharmaceutical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free methodology. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line.
