Advanced Bicyclic Lactam Synthesis via Controllable Sulfonyl Radical Cyclization for Commercial Scale
Introduction to Controllable Bicyclization Technology
The landscape of organic synthesis is continuously evolving with the introduction of more efficient and atom-economical methodologies for constructing complex molecular architectures. A significant breakthrough in this domain is documented in Chinese Patent CN113336692B, which discloses a novel method for the controllable bicyclization of 1,6-diene compounds initiated by sulfonyl radicals. This technology represents a paradigm shift in how chemists approach the synthesis of fused ring systems, particularly those containing nitrogen heterocycles which are ubiquitous in pharmaceutical agents and functional materials. By leveraging a copper-catalyzed oxidative system, this process enables the direct transformation of readily available 1,6-dienes and sulfonyl hydrazides into highly functionalized bicyclic lactams. The significance of this invention lies not only in its chemical elegance but also in its practical applicability for industrial manufacturing, offering a robust pathway to access structural motifs that were previously difficult or costly to produce.
For research and development teams focused on pipeline expansion, the ability to rapidly generate diverse bicyclic scaffolds is invaluable. The patent highlights that this method operates under remarkably mild conditions compared to traditional radical cyclizations, which often require harsh initiators or strict exclusion of moisture and oxygen. The core innovation involves the generation of sulfonyl radicals from sulfonyl hydrazides, which then undergo a cascade addition to the diene system. This results in the formation of two new carbon-carbon bonds and one carbon-nitrogen bond in a single operational step, drastically improving step economy. As we delve deeper into the technical specifics, it becomes clear that this methodology addresses several critical pain points in modern synthetic chemistry, including reagent stability, operational simplicity, and product purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of bicyclic frameworks similar to those described in this patent has been fraught with synthetic challenges. Traditional approaches often rely on multi-step sequences involving separate cyclization events, protecting group manipulations, and extensive purification between stages. For instance, classical radical cyclizations frequently necessitate the use of toxic tin hydrides or stoichiometric amounts of hazardous halogenated reagents, which pose significant environmental and safety liabilities in a commercial setting. Furthermore, many existing protocols demand rigorous exclusion of air and moisture, requiring specialized equipment such as Schlenk lines or gloveboxes, which increases capital expenditure and operational complexity. The lack of stereocontrol in many free-radical processes also leads to complex mixtures of diastereomers, complicating isolation and reducing overall yield. These factors collectively contribute to higher production costs and longer lead times, making conventional routes less attractive for large-scale manufacturing of pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN113336692B offers a streamlined and highly efficient alternative. The reaction utilizes a simple combination of a copper salt catalyst and a peroxide oxidant to drive the transformation under ambient air atmosphere. This eliminates the need for inert gas protection, a feature that significantly lowers the barrier to entry for implementation in standard chemical reactors. The use of sulfonyl hydrazides as radical precursors is particularly advantageous due to their stability, low cost, and ease of handling compared to volatile sulfonyl chlorides. The process achieves high levels of diastereoselectivity, often exceeding a ratio of 20:1, which ensures that the desired isomer is produced predominantly, thereby reducing the burden on downstream purification processes.
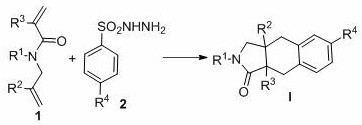
As illustrated in the general reaction scheme above, the transformation converts linear 1,6-diene precursors into complex fused ring systems with high precision. The versatility of this approach is evidenced by its tolerance to a wide range of substituents on both the diene and the sulfonyl hydrazide components. Whether the substrate bears electron-donating groups like methoxy or electron-withdrawing groups like halogens, the reaction proceeds smoothly to afford the target bicyclic products in good to excellent yields. This broad substrate scope makes the technology highly adaptable for the synthesis of diverse libraries of compounds, facilitating rapid structure-activity relationship (SAR) studies in drug discovery programs.
Mechanistic Insights into Copper-Catalyzed Radical Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing the process and ensuring reproducibility at scale. The reaction is believed to proceed through a copper-mediated single-electron transfer (SET) mechanism. Initially, the copper(II) catalyst interacts with the sulfonyl hydrazide in the presence of the oxidant, likely tert-butyl hydroperoxide (TBHP), to generate a sulfonyl radical species. This highly reactive intermediate then adds selectively to one of the terminal double bonds of the 1,6-diene substrate. The resulting carbon-centered radical undergoes an intramolecular 5-exo-trig cyclization onto the second alkene moiety, forming the five-membered ring characteristic of the pyrrolidine core. Subsequent oxidation and nucleophilic capture by the nitrogen atom, or potentially a radical-polar crossover event, leads to the closure of the second ring and the formation of the final lactam structure. The copper catalyst plays a dual role, facilitating both the initiation of the radical chain and the termination steps that restore the catalytic cycle.
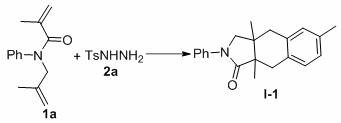
The specific example shown above demonstrates the efficacy of using copper acetate as the catalyst and TBHP as the oxidant in acetonitrile solvent. In this representative case, the reaction yields the bicyclic product I-1 with high efficiency. The choice of copper acetate is particularly strategic; it offers a balance of solubility in organic solvents and appropriate redox potential to mediate the radical generation without promoting excessive side reactions. The high diastereoselectivity observed suggests that the cyclization transition state is highly organized, likely controlled by steric interactions between the substituents on the nitrogen atom and the forming ring system. This level of control is essential for producing pharmaceutical intermediates where stereochemical purity is a regulatory requirement. Furthermore, the mechanism avoids the formation of persistent radical intermediates that could lead to polymerization or oligomerization, ensuring that the reaction remains clean and manageable.
How to Synthesize Bicyclic Lactams Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires attention to specific operational parameters to maximize yield and safety. The protocol is designed to be straightforward, utilizing common reagents and standard heating equipment. The key to success lies in maintaining the correct stoichiometry of the oxidant and ensuring adequate mixing to facilitate mass transfer in the heterogeneous reaction mixture if any solids are present. The reaction time is typically around 20 hours, allowing for complete conversion of the starting materials as monitored by thin-layer chromatography (TLC) or gas chromatography (GC). Post-reaction workup is equally simple, involving standard liquid-liquid extraction and column chromatography, which are well-established unit operations in the fine chemical industry.
- Combine 1,6-diene compound, sulfonyl hydrazide, copper acetate catalyst, and TBHP oxidant in acetonitrile solvent within a reaction vessel.
- Heat the reaction mixture to 90°C under 1 atm air atmosphere with stirring for approximately 20 hours until TLC indicates completion.
- Extract the reaction mixture with ethyl acetate, dry the organic phase over anhydrous sodium sulfate, and purify the residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this sulfonyl radical-initiated bicyclization method offers substantial benefits for procurement and supply chain management. The primary advantage stems from the drastic simplification of the reaction conditions. By operating under air atmosphere rather than requiring inert gas blanketing, manufacturers can utilize standard reactor vessels without the need for specialized nitrogen or argon purging systems. This reduction in infrastructure requirements translates directly into lower capital expenditures and reduced operational overhead. Additionally, the reagents employed, such as copper acetate and TBHP, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of supply chain disruptions associated with exotic or proprietary catalysts.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of expensive and hazardous reagents traditionally used in radical chemistry. The avoidance of tin hydrides or stoichiometric halogen sources not only reduces raw material costs but also significantly lowers waste disposal expenses, as the process generates less toxic byproduct stream. Furthermore, the high diastereoselectivity achieved minimizes the loss of material during purification, effectively increasing the overall throughput of the manufacturing line. The simplified workup procedure, which relies on common solvents like ethyl acetate and hexane, further contributes to cost efficiency by reducing solvent consumption and processing time.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable and commercially abundant starting materials. Sulfonyl hydrazides are robust solids that can be stored for extended periods without degradation, unlike some sensitive liquid reagents that require cold chain logistics. The tolerance of the reaction to ambient conditions means that production schedules are less vulnerable to delays caused by utility failures, such as interruptions in nitrogen supply. This reliability ensures consistent delivery of high-purity pharmaceutical intermediates to downstream customers, fostering stronger long-term partnerships and trust within the supply network.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne quantities is facilitated by its inherent safety profile. The absence of pyrophoric reagents and the use of moderate temperatures (80-100°C) reduce the risk of thermal runaways, making the process safer for large-scale operations. From an environmental standpoint, the method aligns with green chemistry principles by utilizing air as a benign reactant component and generating minimal hazardous waste. The high atom economy of the bicyclization reaction ensures that a large proportion of the starting material ends up in the final product, reducing the environmental footprint per unit of production and aiding in compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the reaction scope and operational details. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these nuances is critical for successful technology transfer and scale-up.
Q: What are the optimal reaction conditions for this bicyclization?
A: The patent specifies using copper acetate (20 mol%) as the catalyst and tert-butyl hydroperoxide (TBHP) as the oxidant in acetonitrile at 90°C under air atmosphere.
Q: Does this method require inert gas protection?
A: No, a key advantage of this protocol is its operation under 1 atm air atmosphere, eliminating the need for expensive inert gas setups like nitrogen or argon.
Q: What is the diastereoselectivity of the products?
A: The method demonstrates excellent stereocontrol, with many examples showing a diastereomeric ratio (d.r.) greater than 20:1, simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bicyclic Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the sulfonyl radical-initiated bicyclization described in CN113336692B. Our team of expert chemists is well-versed in adapting such cutting-edge academic and patent literature into robust, commercial-grade manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of complex intermediates. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to request specific COA data and route feasibility assessments to validate the suitability of these bicyclic lactams for your next-generation drug candidates. Together, we can accelerate the development of life-saving medicines through innovative chemistry and reliable manufacturing excellence.
