Advanced Synthesis of Naphthalenolactam Derivatives for Potent Tumor Cell Proliferation Inhibition
The pharmaceutical industry continuously seeks novel scaffolds capable of disrupting tumor cell proliferation with high specificity and potency. A significant breakthrough in this domain is documented in patent CN101602707A, which discloses a comprehensive class of naphthalenolactam derivatives engineered for superior oncological applications. These compounds are characterized by the strategic incorporation of high-biological-activity functional groups, including acetylene bonds, cyano groups, amino groups, and amide structures, onto the robust benzo[c,d]indole core. The innovation lies not merely in the final structures but in the versatile synthetic methodology that allows for precise modulation of the molecule's electronic and steric properties. By systematically modifying the 1-position imine and introducing diverse electrophilic substituents at the 6-position, researchers can fine-tune the interaction of these derivatives with cellular targets. This level of structural control is paramount for developing next-generation therapeutic agents that overcome resistance mechanisms often encountered in current chemotherapy regimens.
For procurement specialists and supply chain managers, the significance of this patent extends beyond biological efficacy to encompass manufacturability and scalability. The disclosed synthetic routes utilize readily available starting materials and standard industrial reagents, mitigating the risks associated with exotic or unstable precursors. The ability to synthesize a wide array of derivatives from a common naphthalenolactam parent suggests a platform technology approach, where a single production line can be adapted to generate multiple high-value intermediates. This flexibility is crucial for maintaining supply continuity in the volatile pharmaceutical market. Furthermore, the detailed experimental examples provided in the patent, ranging from gram-scale preparations with yields exceeding 80% in certain steps, offer a clear roadmap for commercial scale-up. Understanding these technical nuances allows stakeholders to assess the true potential of these compounds as reliable pharmaceutical intermediates for large-scale drug development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the naphthalenolactam scaffold has presented significant challenges due to the inherent stability and low reactivity of the fused ring system. Conventional methods often struggled to achieve selective substitution at the 6-position without compromising the integrity of the lactam ring or requiring harsh conditions that led to extensive byproduct formation. Traditional electrophilic aromatic substitution on similar polycyclic systems frequently resulted in mixtures of regioisomers, necessitating complex and costly purification processes that drastically reduced overall throughput. Additionally, modifying the nitrogen atom at the 1-position was often hindered by the low nucleophilicity of the imine hydrogen, requiring strong bases or high temperatures that could degrade sensitive functional groups elsewhere in the molecule. These limitations severely restricted the chemical space accessible to medicinal chemists, resulting in a limited library of derivatives with suboptimal pharmacokinetic profiles. The inability to efficiently introduce diverse side chains, such as propargyl or aminoethyl groups, further constrained the optimization of biological activity against specific tumor cell lines.
The Novel Approach
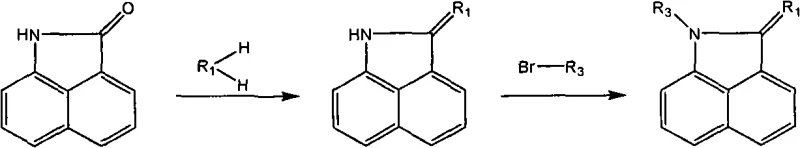
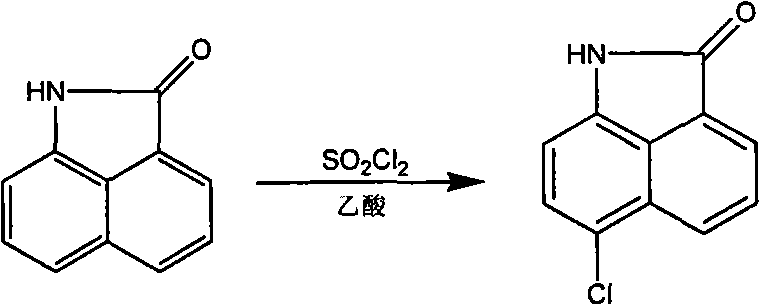
The methodology outlined in the patent represents a paradigm shift by employing a sequential strategy that decouples the modification of the aromatic core from the functionalization of the nitrogen center. By first utilizing phosphorus oxychloride (POCl3) as a potent dehydrating agent, the process facilitates a clean dehydrocondensation between the C2 carbonyl and active methylene compounds or aromatic amines. This step establishes the exocyclic double bond essential for biological activity while preserving the core structure. Subsequently, the introduction of electrophilic groups at the 6-position is achieved under controlled conditions—such as using SO2Cl2 in acetic acid for chlorination or liquid bromine in 1,2-dichloroethane for bromination—ensuring high regioselectivity. Finally, the N-alkylation at the 1-position is performed using specific halogenated alkanes in basic media, allowing for the precise attachment of bioactive side chains. This modular approach not only simplifies the synthesis but also enables the rapid generation of a diverse library of derivatives, significantly accelerating the lead optimization process for oncology drug discovery.
Mechanistic Insights into POCl3-Mediated Dehydrocondensation and Electrophilic Substitution
The core of this synthetic innovation relies on the activation of the naphthalenolactam carbonyl group by phosphorus oxychloride. Mechanistically, POCl3 reacts with the oxygen of the C2 carbonyl to form a highly reactive imidoyl chloride intermediate. This activation significantly increases the electrophilicity of the C2 carbon, making it susceptible to nucleophilic attack by the active methylene group of reagents like malononitrile or dimethyl malonate. The subsequent elimination of HCl drives the formation of the exocyclic double bond, yielding the 2-ylidene derivative. This dehydration condensation is critical because it extends the conjugation of the pi-system, which is often correlated with enhanced intercalation or binding affinity to biological targets. The choice of solvent, such as toluene or chlorobenzene, and the temperature range of 100-120°C are optimized to balance reaction kinetics with the thermal stability of the intermediates. This step sets the foundation for the entire molecular architecture, ensuring that the subsequent modifications occur on a stable and well-defined scaffold.
Following the establishment of the C2 substituent, the electrophilic substitution at the 6-position exploits the electron density distribution of the naphthalene ring. The presence of the electron-withdrawing lactam and the exocyclic double bond influences the reactivity of the aromatic protons, directing incoming electrophiles primarily to the 6-position. For instance, nitration using a mixture of concentrated nitric and sulfuric acids proceeds via the formation of the nitronium ion, which attacks the activated ring position. Similarly, halogenation with SO2Cl2 or Br2 involves the generation of positive halogen species that selectively substitute the hydrogen at C6. This regioselectivity is vital for maintaining batch-to-batch consistency and minimizing the formation of difficult-to-remove isomers. The final N-alkylation step involves the deprotonation of the imine nitrogen by bases like sodium methoxide or potassium tert-butoxide, generating a nucleophilic anion that displaces the halide in alkylating agents like bromoacetylene. This sequence ensures that each functional group is installed with high fidelity, resulting in derivatives with defined impurity profiles suitable for rigorous biological testing.
How to Synthesize Naphthalenolactam Derivatives Efficiently
The synthesis of these high-value oncology intermediates follows a robust, multi-step protocol designed for reproducibility and scalability. The process begins with the activation of the naphthalenolactam core, followed by sequential functionalization to install the desired pharmacophores. Each step has been optimized to maximize yield and purity, utilizing standard laboratory equipment and commercially available reagents. The detailed procedural parameters, including stoichiometry, temperature profiles, and workup procedures, are critical for achieving the high-quality standards required for pharmaceutical applications. By adhering to these optimized conditions, manufacturers can ensure the consistent production of derivatives that meet stringent specifications for downstream biological evaluation.
- Perform dehydrocondensation of naphthalenolactam with active methylene compounds or aromatic amines using POCl3 in chlorobenzene or toluene at 100-120°C.
- Introduce electrophilic substituents at the 6-position via chlorination, bromination, or nitration using SO2Cl2, Br2, or HNO3/H2SO4 respectively.
- Substitute the active hydrogen at the 1-position imine using halogenated alkanes like bromoacetylene or bromoethylamine under basic conditions.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in patent CN101602707A offers distinct advantages that align with the goals of cost efficiency and supply chain resilience. The reliance on commodity chemicals such as phosphorus oxychloride, malononitrile, and common halogenated solvents reduces the dependency on specialized or single-source suppliers. This accessibility of raw materials translates directly into lower procurement costs and reduced risk of supply disruptions. Furthermore, the reaction conditions, while requiring careful temperature control, do not necessitate extreme pressures or cryogenic environments, which simplifies the engineering requirements for production facilities. The ability to perform these reactions in standard glass-lined or stainless steel reactors makes the technology transfer from lab to plant straightforward and cost-effective. For procurement managers, this means a more predictable cost structure and the ability to negotiate better terms with chemical vendors due to the high volume availability of the necessary inputs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts, which are often expensive and require complex removal steps to meet residual metal specifications, represents a significant operational saving. The process relies on organic reagents and simple inorganic acids or bases, which are inexpensive and easy to handle. Additionally, the high yields reported in the patent examples, such as the 94% yield in the chlorination step and 83% in the initial condensation, indicate a material-efficient process that minimizes waste generation. This efficiency reduces the cost of goods sold (COGS) by maximizing the output per unit of input. The simplified purification methods, often involving recrystallization or standard column chromatography, further contribute to cost reduction by avoiding expensive preparative HPLC or complex distillation setups.
- Enhanced Supply Chain Reliability: The use of stable intermediates and robust reaction conditions enhances the overall reliability of the supply chain. Unlike processes that involve unstable intermediates requiring immediate consumption, several intermediates in this route, such as the 6-substituted naphthalenolactams, can be isolated and stored. This capability allows for the decoupling of production stages, enabling manufacturers to build inventory buffers against demand fluctuations. The modularity of the synthesis also means that if a specific alkylating agent becomes unavailable, the production of the core scaffold can continue, allowing for flexibility in the final product portfolio. This resilience is critical for maintaining continuous supply to pharmaceutical clients who depend on timely delivery for their clinical trial timelines.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, as demonstrated by the successful execution of reactions on molar scales that can be directly translated to kilogram or tonne production. The waste streams generated are primarily aqueous acidic or basic solutions and organic solvents that can be recovered and recycled, aligning with modern environmental compliance standards. The absence of heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing process. This adherence to green chemistry principles not only mitigates regulatory risks but also appeals to environmentally conscious partners in the pharmaceutical value chain. The potential for solvent recovery, particularly of high-boiling solvents like chlorobenzene or DMF, further enhances the economic and environmental sustainability of the operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these naphthalenolactam derivatives. The answers are derived directly from the experimental data and technical disclosures within the patent documentation. Understanding these details helps stakeholders make informed decisions regarding the integration of these intermediates into their research and development pipelines. The clarity provided here aims to bridge the gap between academic discovery and industrial application.
Q: What is the primary biological activity of these naphthalenolactam derivatives?
A: According to patent CN101602707A, these derivatives exhibit significant inhibitory activity against tumor cell proliferation, specifically targeting 7721 human liver cancer cells, MCF-7 human breast cancer cells, Hela human cervical cancer cells, and BGC-823 human gastric cancer cells.
Q: How is the 6-position of the naphthalenolactam ring modified?
A: The 6-position is modified through electrophilic substitution reactions. Chlorine is introduced using SO2Cl2 in acetic acid, bromine using liquid Br2 in 1,2-dichloroethane, and nitro groups using concentrated HNO3 in concentrated H2SO4 under controlled temperatures.
Q: What reagents are used for the N-alkylation at the 1-position?
A: The active hydrogen at the 1-position is substituted using various halogenated alkanes such as bromoacetylene, bromoethylamine, or dibromoethane. The reaction typically employs basic conditions using systems like acetonitrile/t-BuOK or DMF/NaOCH3.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalenolactam Derivatives Supplier
The technical potential of the naphthalenolactam derivatives described in patent CN101602707A is immense, offering a promising avenue for the development of novel antitumor agents. As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring these complex molecules from the bench to the market. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of intermediate meets the exacting standards of the global pharmaceutical industry. We understand the critical nature of oncology research and are committed to providing materials that support the acceleration of life-saving therapies.
We invite you to initiate a dialogue regarding your specific supply chain needs and explore how our manufacturing capabilities can optimize your project economics. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for any of the derivatives discussed. By partnering with us, you gain access to a reliable supply of high-purity pharmaceutical intermediates backed by decades of chemical engineering expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
