Scalable Metal-Free Synthesis of N-N Compounds for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of N-N Compounds for Advanced Pharmaceutical Intermediates
The strategic development of efficient synthetic routes for nitrogen-nitrogen bonded compounds remains a critical priority for the global pharmaceutical and fine chemical industries. As detailed in the recent patent CN115745847A, a groundbreaking methodology has emerged that leverages hypervalent iodine chemistry to construct N-N bonds with unprecedented efficiency. This technology addresses the longstanding challenges associated with hydrazine derivative synthesis, offering a robust pathway for producing high-value intermediates used in drugs like Celecoxib and advanced organic materials. For R&D leaders and procurement strategists, this metal-free oxidative coupling represents a significant shift towards greener, more cost-effective manufacturing paradigms that align with modern sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the N-N bond has relied on methodologies that are often fraught with operational complexities and economic inefficiencies. Traditional approaches frequently involve the diazotization of pre-functionalized nitrogen compounds, a process that typically requires harsh acidic conditions and generates stoichiometric amounts of hazardous waste. Alternatively, methods utilizing organometallic reagents for pre-functionalization introduce significant cost burdens due to the high price of precious metal catalysts and the absolute necessity for rigorous metal removal steps to meet pharmaceutical purity standards. Furthermore, nucleophilic substitution strategies involving leaving groups often suffer from limited substrate scope and the formation of difficult-to-separate by-products, which drastically reduces overall yield and complicates downstream processing for supply chain teams.
The Novel Approach
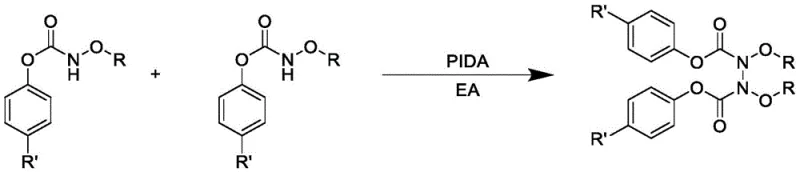
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes iodobenzene diacetate (PIDA) as a mild, metal-free oxidant to drive the direct self-coupling of N-alkoxycarbamates. This transformative reaction proceeds smoothly under an air atmosphere at room temperature, eliminating the need for expensive inert gas protection or energy-intensive heating protocols. The reaction mechanism facilitates high atom utilization, ensuring that the majority of the starting material mass is incorporated into the final product rather than lost as waste. By employing commercially available solvents like ethyl acetate and avoiding toxic heavy metals, this method not only simplifies the regulatory compliance landscape but also significantly lowers the barrier to entry for large-scale commercial production.
Mechanistic Insights into PIDA-Mediated Oxidative Coupling
The core of this technological breakthrough lies in the unique reactivity of the hypervalent iodine species, which acts as a potent single-electron oxidant to generate nitrogen-centered radicals from the N-alkoxycarbamate precursors. Unlike transition metal catalysis which often relies on complex coordination cycles, this radical-mediated pathway allows for the direct homocoupling of two nitrogen centers to form the stable N-N bond found in Formula I. The reaction exhibits remarkable chemoselectivity, tolerating a wide array of electronic environments on the aromatic ring without compromising the integrity of sensitive functional groups. This mechanistic elegance ensures that the resulting hydrazine derivatives possess high structural fidelity, a critical parameter for R&D directors focused on impurity profiling and batch-to-batch consistency in API synthesis.
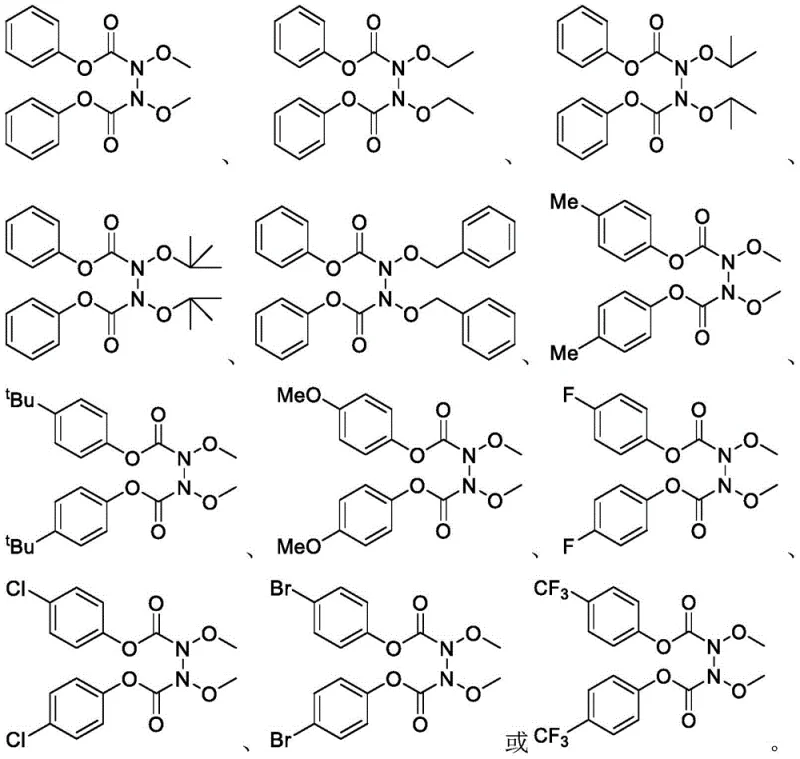
Furthermore, the versatility of this coupling strategy is evidenced by its broad substrate scope, accommodating diverse alkoxy groups such as methyl, ethyl, isopropyl, tert-butyl, and benzyl moieties. The system also demonstrates exceptional tolerance for various para-substituents on the phenyl ring, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl. This flexibility allows chemists to access a vast library of structurally distinct N-N compounds from readily available starting materials, facilitating rapid analog synthesis for drug discovery programs without the need for extensive route scouting or protecting group manipulations.
How to Synthesize N-N Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The process is designed to be operationally simple, requiring standard glassware and ambient conditions, which minimizes the need for specialized equipment. The following guide outlines the fundamental steps derived from the optimized embodiments within the patent, serving as a foundational protocol for process chemists aiming to replicate these results or adapt them for specific target molecules.
- Prepare the reaction mixture by adding N-alkoxycarbamate substrate and PIDA oxidant into ethyl acetate solvent at a molar ratio of approximately 1: 2.
- Maintain the reaction under an air atmosphere at room temperature (25°C) for 16 to 24 hours to ensure complete conversion.
- Filter the mixture to remove solid impurities, concentrate the filtrate under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this PIDA-mediated coupling technology offers tangible strategic benefits that extend beyond mere chemical novelty. The elimination of transition metal catalysts removes a major cost center associated with catalyst procurement and, more importantly, the downstream purification processes required to reduce metal residues to ppm levels. This simplification of the workflow translates directly into reduced processing time and lower utility consumption, as the reaction does not require cryogenic cooling or high-temperature heating, thereby enhancing the overall energy efficiency of the manufacturing site.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the use of commodity chemicals; both the N-alkoxycarbamate substrates and the PIDA oxidant are commercially available at low cost compared to specialized organometallic reagents. By removing the dependency on precious metals like palladium or copper, manufacturers can avoid the volatility of metal markets and the substantial expenses linked to metal scavenger resins. This results in a significantly leaner cost structure for the production of complex hydrazine intermediates, allowing for better margin management in competitive bidding scenarios.
- Enhanced Supply Chain Reliability: The reliance on air-stable reagents and common solvents like ethyl acetate ensures that the supply chain is robust against disruptions often seen with specialty gases or moisture-sensitive reagents. Since the reaction tolerates an air atmosphere, there is no need for complex nitrogen or argon blanketing systems, reducing the risk of batch failure due to operator error or equipment malfunction. This operational resilience ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, providing a competitive edge in fast-paced market environments.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles by minimizing hazardous waste generation. The high atom economy means less waste disposal cost, and the absence of heavy metals simplifies wastewater treatment protocols. These factors make the process highly scalable from gram to multi-ton quantities without encountering the exponential increase in safety risks or regulatory hurdles typically associated with scaling up energetic or toxic reactions, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-N coupling technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using PIDA over traditional metal catalysts for N-N coupling?
A: Using PIDA (iodobenzene diacetate) eliminates the need for expensive and toxic transition metal catalysts. This simplifies the purification process by removing the requirement for heavy metal scavenging steps, thereby reducing production costs and environmental impact while maintaining high atom economy.
Q: What is the substrate scope for this N-N coupling reaction?
A: The method demonstrates excellent tolerance for various functional groups. It successfully couples N-alkoxycarbamates with diverse R groups (methyl, ethyl, tert-butyl, benzyl) and aromatic substituents (fluoro, chloro, bromo, trifluoromethyl, methoxy), making it highly versatile for synthesizing complex pharmaceutical intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates under mild conditions (room temperature, air atmosphere) using commercially available solvents like ethyl acetate. The absence of stringent inert gas requirements and the simplicity of the workup procedure make it highly amenable to scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free oxidative coupling technology for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-N compound delivered adheres to the highest international quality standards required by top-tier global pharma companies.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this PIDA-mediated protocol. We encourage you to contact our technical procurement team today to request specific COA data for our catalog of hydrazine derivatives and to discuss detailed route feasibility assessments tailored to your unique supply chain needs.
