Advanced Metal-Free Synthesis of Functionalized Allenones for Pharmaceutical Applications
Introduction to Patent CN113620762A and Allenone Technology
The landscape of organic synthesis is continuously evolving, driven by the demand for greener, more efficient, and safer methodologies, particularly in the realm of pharmaceutical intermediate production. Patent CN113620762A introduces a groundbreaking approach to the preparation of allenone compounds, a class of highly functionalized molecules with significant potential in drug discovery and material science. This technology leverages the power of N-heterocyclic carbene (NHC) organocatalysis combined with radical chemistry to achieve what was previously difficult with traditional methods. By utilizing a metal-free catalytic system, this invention addresses critical pain points regarding environmental pollution and product purity that have long plagued the fine chemical industry. The ability to synthesize these complex structures under mild conditions represents a paradigm shift, offering a reliable pathway for creating high-value building blocks.
Furthermore, the versatility of the resulting allenone compounds cannot be overstated. These molecules serve as pivotal intermediates capable of undergoing diverse transformations such as electrophilic additions and cyclization reactions, which are essential for constructing complex pharmacophores. The patent highlights that these compounds are not merely theoretical constructs but practical tools with wide application prospects in biochemistry and pesticide research. For R&D directors and procurement specialists alike, understanding the underlying chemistry of this patent is crucial. It signifies a move towards sustainable manufacturing processes that do not compromise on yield or structural complexity, thereby aligning perfectly with the modern goals of cost reduction in fine chemical manufacturing and environmental stewardship.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allenone compounds via the functionalization of 1,3-enyne compounds has relied heavily on transition metal catalysis systems, predominantly involving copper (Cu) and nickel (Ni). While these metal-catalyzed pathways have served the industry for years, they come with inherent and significant drawbacks that limit their utility in modern high-purity applications. The primary concern is the inevitable introduction of metal residues into the final product, which necessitates rigorous and costly purification steps to meet the stringent regulatory standards of the pharmaceutical sector. Additionally, these metal systems often require harsh reaction conditions, including extreme temperatures or pressures, which can lead to safety hazards and increased energy consumption. The substrate scope in these traditional methods is frequently narrow, failing to accommodate sensitive functional groups that are common in advanced drug candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113620762A utilizes an organic small-molecule N-heterocyclic carbene catalyst to drive the reaction through a radical mechanism. This novel approach effectively bypasses the need for toxic transition metals, ensuring a strictly non-metallic reaction environment that simplifies downstream processing. The reaction proceeds by coupling a 1,3-eneyne compound with an aldehyde and a radical precursor, facilitated by the unique electronic properties of the NHC catalyst. This strategy not only improves atom utilization rates but also significantly enhances the safety profile of the production process. By operating under mild conditions, typically between 60°C and 80°C, the method preserves the integrity of sensitive functional groups, thereby expanding the designability of the target molecules.
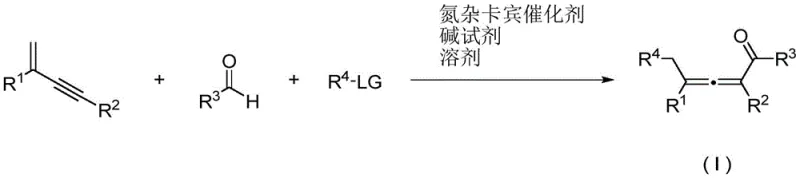
The visual representation of this transformation underscores its elegance and efficiency. As shown in the reaction scheme, three distinct components converge to form the highly functionalized allenone core in a single pot. This convergence is a hallmark of ideal synthetic design, minimizing waste and maximizing throughput. For supply chain managers, this translates to a streamlined process that reduces the number of unit operations required, directly impacting the lead time for high-purity pharmaceutical intermediates. The elimination of metal catalysts also removes the burden of sourcing and disposing of expensive heavy metals, contributing to substantial cost savings and a reduced environmental footprint. This technological leap positions the NHC-catalyzed route as a superior alternative for the commercial scale-up of complex organic intermediates.
Mechanistic Insights into NHC-Catalyzed Radical Coupling
To fully appreciate the robustness of this synthesis, one must delve into the mechanistic intricacies of the N-heterocyclic carbene catalytic cycle. The process begins with the deprotonation of the NHC precursor by a base reagent, generating the active carbene species. This nucleophilic carbene then attacks the carbonyl carbon of the aldehyde substrate, forming a classical Breslow intermediate. This key intermediate is pivotal as it undergoes a single-electron transfer (SET) process with the radical precursor, generating an acyl radical species. Simultaneously, the 1,3-eneyne compound participates in the radical manifold, where the addition of a radical species generates either a propargyl or an allene radical. The synergy between the acyl radical derived from the aldehyde and the allene radical derived from the enyne drives the coupling reaction forward with high selectivity.
The choice of catalyst structure plays a critical role in modulating the reactivity and selectivity of this transformation. The patent specifies several classes of nitrogen-containing heterocyclic compounds, including imidazole, thiazole, and triazole-based azacarbenes, as effective catalysts. Among these, seven-membered thiazole azacarbenes have demonstrated particularly high yields, suggesting that the steric and electronic environment of the catalyst backbone is finely tuned to stabilize the radical intermediates. This level of control is essential for minimizing byproduct formation and ensuring high purity. Understanding these mechanistic details allows chemists to optimize reaction parameters such as solvent choice and stoichiometry, further enhancing the efficiency of the process.
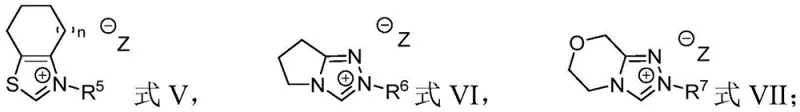
Moreover, the radical nature of this mechanism offers unique advantages in terms of functional group tolerance. Unlike ionic mechanisms that might be quenched by protic solvents or sensitive groups, radical pathways are generally more robust. The patent data indicates that a wide array of substituents, ranging from electron-withdrawing halogens to electron-donating alkoxy groups, are well-tolerated on the aromatic rings of the substrates. This broad compatibility is a testament to the gentle yet effective nature of the NHC-radical dual catalysis system. For R&D teams, this means that the synthetic route can be adapted to a diverse library of analogues without the need for extensive re-optimization, accelerating the drug discovery timeline.
How to Synthesize Functionalized Allenone Compounds Efficiently
Implementing this advanced synthesis in a laboratory or pilot plant setting requires careful attention to reaction conditions and reagent quality. The general protocol involves the precise mixing of the 1,3-eneyne compound, the aldehyde, and the radical precursor in an anhydrous solvent such as acetonitrile or dichloromethane. The addition of the NHC catalyst and the base reagent must be conducted under an inert atmosphere, typically argon or nitrogen, to prevent the quenching of radical species by oxygen. Temperature control is also paramount; maintaining the reaction mixture between 60°C and 80°C ensures optimal kinetics without promoting decomposition. The detailed standardized synthesis steps provided below outline the specific molar ratios and workup procedures necessary to achieve the high yields reported in the patent examples.
- Prepare the reaction system by mixing 1,3-eneyne compounds, aldehyde compounds, and radical precursors in an inert atmosphere.
- Add the N-heterocyclic carbene catalyst and alkali reagent to initiate the formation of the active acyl radical intermediate.
- Maintain the reaction temperature between 60-80°C to facilitate the radical coupling and obtain the target allenone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this NHC-catalyzed synthesis offers transformative benefits for procurement and supply chain operations. The most immediate impact is seen in the realm of cost reduction in manufacturing. By eliminating the need for expensive transition metal catalysts like palladium, copper, or nickel, the raw material costs are significantly lowered. Furthermore, the absence of metal residues removes the necessity for complex and costly purification steps such as scavenging or recrystallization specifically designed to meet heavy metal limits. This simplification of the downstream process not only saves money but also reduces the time required to release batches for quality control, thereby enhancing overall operational efficiency.
- Cost Reduction in Manufacturing: The economic advantages of this metal-free protocol are multifaceted. Firstly, organic small-molecule catalysts are generally more affordable and easier to source than specialized transition metal complexes. Secondly, the high atom economy of the reaction means that a larger proportion of the starting materials ends up in the final product, reducing waste disposal costs. Thirdly, the mild reaction conditions translate to lower energy consumption for heating and cooling, contributing to a smaller utility bill. These factors combine to create a leaner, more cost-effective production model that improves profit margins without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available commercial starting materials. The aldehydes and 1,3-enynes required for this synthesis are commodity chemicals that can be sourced from multiple vendors, reducing the risk of supply disruptions associated with specialized reagents. Additionally, the robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in environmental parameters, leading to more consistent batch-to-batch results. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the simplicity of the operation. The reaction does not require high-pressure equipment or cryogenic conditions, making it compatible with standard glass-lined or stainless steel reactors found in most multipurpose plants. From an environmental standpoint, the metal-free nature of the process drastically reduces the toxicity of the effluent, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. This alignment with green chemistry principles enhances the corporate sustainability profile of any manufacturer adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these allenone compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the scope, limitations, and practical implementation of the technology for potential partners and stakeholders. Understanding these nuances is key to leveraging the full potential of this innovative synthetic method in your own R&D or production workflows.
Q: What are the advantages of using NHC catalysis over traditional metal catalysts for allenone synthesis?
A: NHC catalysis eliminates the risk of heavy metal contamination, which is critical for pharmaceutical applications. It also operates under milder conditions with higher atom economy compared to copper or nickel systems.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process uses commercially available raw materials and simple operational steps without requiring complex protection/deprotection strategies, making it highly suitable for commercial scale-up.
Q: What types of functional groups are tolerated in this reaction?
A: The method exhibits broad substrate universality, tolerating various groups including halogens, esters, ethers, and heterocycles, allowing for diverse structural modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allenone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of the technologies described in CN113620762A to revolutionize the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into robust, commercial-scale processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation.
We invite you to collaborate with us to explore the possibilities of this metal-free allenone synthesis for your specific projects. Whether you require custom synthesis services or large-scale manufacturing, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with cutting-edge chemical solutions.
