Advanced Copper-Catalyzed Aerobic Oxidation for Scalable Nitrile Production
Introduction to Patent CN102964196B Technology
The pharmaceutical and fine chemical industries constantly seek robust methodologies for constructing nitrogen-containing heterocycles and building blocks, with nitriles serving as pivotal intermediates for amines, amides, and tetrazoles. Patent CN102964196B introduces a transformative approach to nitrile synthesis, shifting away from hazardous traditional protocols toward a sustainable, copper-catalyzed aerobic oxidation strategy. This technology leverages a synergistic catalytic system comprising copper salts, nitrogen-based ligands, and nitroxyl radical co-catalysts to efficiently convert primary alcohols directly into nitriles using aqueous ammonia and molecular oxygen. By operating under mild conditions, often at room temperature or slightly elevated temperatures up to 55°C, this method addresses critical pain points regarding safety, atom economy, and operational simplicity that have long plagued nitrile manufacturing.
The significance of this innovation lies in its ability to utilize benign starting materials while achieving high yields across a broad substrate scope. Unlike legacy processes that rely on stoichiometric amounts of toxic cyanating agents or harsh dehydrating conditions, this catalytic cycle utilizes atmospheric oxygen as the terminal oxidant, generating water as the primary byproduct. This aligns perfectly with modern green chemistry principles, offering a pathway to reduce the environmental footprint of chemical synthesis. For R&D teams and process chemists, understanding the nuances of this copper-mediated transformation provides a powerful tool for optimizing synthetic routes for active pharmaceutical ingredients (APIs) and advanced intermediates.
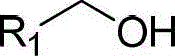
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nitrile compounds has relied heavily on nucleophilic substitution reactions involving alkyl halides and metal cyanides such as sodium cyanide or potassium cyanide. While effective, these methods present severe safety liabilities due to the extreme toxicity of cyanide salts, requiring specialized containment facilities and rigorous waste treatment protocols to prevent environmental contamination. Furthermore, these substitution reactions often suffer from poor atom economy and generate substantial amounts of inorganic salt waste. Alternative routes involving the dehydration of primary amides typically necessitate the use of aggressive dehydrating agents like phosphorus oxychloride or thionyl chloride, which are corrosive, moisture-sensitive, and produce acidic gaseous byproducts that demand scrubbing systems. These conventional pathways also frequently require high temperatures and prolonged reaction times, leading to energy inefficiencies and potential decomposition of thermally sensitive substrates.
The Novel Approach
In stark contrast, the methodology disclosed in CN102964196B utilizes a catalytic oxidative ammonolysis strategy that fundamentally redefines the safety and efficiency profile of nitrile synthesis. By employing a copper salt catalyst in conjunction with a TEMPO-based co-catalyst and a bidentate nitrogen ligand, the system activates molecular oxygen to drive the oxidation of the alcohol intermediate to an aldehyde, which subsequently condenses with ammonia and undergoes further oxidation to the nitrile. This one-pot transformation eliminates the need for isolating unstable aldehyde or imine intermediates. The reaction proceeds smoothly in common protic solvents like ethanol under an oxygen balloon or air atmosphere, drastically simplifying the equipment requirements. The mild thermal profile, often effective at room temperature, preserves sensitive functional groups that would otherwise degrade under the harsh conditions of traditional dehydration or cyanation protocols.
Mechanistic Insights into Cu/TEMPO-Catalyzed Aerobic Oxidation
The catalytic cycle underpinning this transformation involves a sophisticated interplay between the copper center and the nitroxyl radical co-catalyst. Initially, the copper species, stabilized by ligands such as 2,2'-bipyridine or 1,10-phenanthroline, coordinates with the alcohol substrate and facilitates its oxidation to the corresponding aldehyde. The TEMPO co-catalyst acts as a hydrogen atom transfer mediator, shuttling electrons and protons to regenerate the active copper-oxygen species. This cooperative catalysis ensures that the oxidation potential is sufficiently high to drive the reaction forward while maintaining selectivity for the primary alcohol over other oxidizable functionalities. The presence of aqueous ammonia allows for the rapid formation of a hemiaminal intermediate from the generated aldehyde, which is then swiftly oxidized by the same catalytic system to yield the final nitrile product. This mechanism avoids the accumulation of free aldehyde, minimizing side reactions such as aldol condensation.
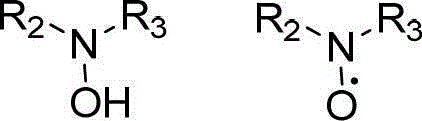
Impurity control is inherently built into this mechanistic pathway due to the high chemoselectivity of the copper/TEMPO system. Because the reaction avoids strong acids or bases and operates at neutral to slightly basic pH levels provided by ammonia, acid-catalyzed polymerization or base-mediated elimination side reactions are suppressed. The use of molecular oxygen as the oxidant ensures that the only stoichiometric byproduct is water, which simplifies downstream processing and purification. For process chemists, this means that the crude reaction mixture typically contains fewer organic impurities compared to routes using chlorinating or dehydrating reagents. The robustness of the catalyst system also tolerates a wide range of electronic environments on the aromatic ring, from electron-donating methoxy groups to electron-withdrawing halogens, ensuring consistent quality and yield across diverse substrate libraries without the need for extensive re-optimization.
How to Synthesize Nitrile Compounds Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the catalytic components to ensure maximum turnover. The patent specifies that a combination of copper iodide, bipyridine, and TEMPO in ethanol provides an optimal balance of activity and cost. The procedure involves charging the catalysts into a reaction vessel, purging with oxygen, and then introducing the alcohol substrate and aqueous ammonia. The reaction is typically stirred at ambient temperature for approximately 24 hours, although kinetics can be accelerated by mild heating to 55°C if necessary. Monitoring via thin-layer chromatography (TLC) allows for precise determination of the endpoint, preventing over-oxidation or degradation. Following completion, standard extraction and column chromatography techniques yield the pure nitrile product with high recovery rates.
- Prepare the catalytic system by mixing copper salt (e.g., CuI), ligand (e.g., bipyridine), and TEMPO co-catalyst in an alcoholic solvent like ethanol.
- Add the primary alcohol substrate and aqueous ammonia to the reaction mixture under an oxygen or air atmosphere.
- Stir the reaction at mild temperatures (room temperature to 55°C) until completion, monitored by TLC, followed by standard purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, adopting this copper-catalyzed technology offers profound strategic benefits that extend beyond mere chemical efficiency. The shift from toxic cyanide sources to benign alcohols and ammonia fundamentally alters the risk profile of the manufacturing process, potentially lowering insurance costs and regulatory compliance burdens associated with handling scheduled hazardous substances. The reliance on commodity chemicals like ethanol and air as the oxidant means that raw material sourcing is resilient against market volatility, ensuring a stable supply chain even during global disruptions. Furthermore, the simplified workup procedures reduce the consumption of auxiliary materials and solvents during purification, contributing to a leaner and more cost-effective production model.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous cyanating reagents directly lowers the bill of materials for nitrile production. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, as the process often proceeds effectively at room temperature. The high atom economy of the oxidative pathway means that a greater proportion of the starting mass is converted into the desired product, minimizing waste disposal costs. By avoiding the need for specialized corrosion-resistant equipment required for strong acids or dehydrating agents, capital expenditure for new production lines can be significantly optimized.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, primarily benzyl alcohols and aliphatic alcohols, are widely available from multiple global suppliers, reducing dependency on single-source vendors. The use of aqueous ammonia and oxygen removes the logistical challenges of transporting and storing toxic gases or solids. This accessibility ensures that production schedules can be maintained with minimal risk of raw material shortages. The robustness of the reaction also implies fewer batch failures due to sensitivity to moisture or oxygen, leading to more predictable lead times and reliable delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The green nature of this process aligns with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and toxic waste discharge. Generating water as the primary byproduct simplifies wastewater treatment and reduces the environmental footprint of the facility. The scalability is demonstrated by the use of simple stirring and gas sparging, which translates easily from liter-scale reactors to multi-ton vessels without complex engineering modifications. This ease of scale-up allows manufacturers to respond rapidly to increased market demand without lengthy process validation periods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented nitrile synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the safety advantages of this nitrile synthesis method compared to traditional cyanation?
A: Unlike traditional methods that utilize highly toxic cyanide salts (NaCN/KCN) or require hazardous dehydration agents, this patent describes a green oxidative protocol using aqueous ammonia and oxygen. This eliminates the handling of lethal cyanide sources, significantly improving operational safety and reducing environmental hazards associated with toxic waste disposal.
Q: Can this copper-catalyzed method tolerate sensitive functional groups on the aromatic ring?
A: Yes, the methodology demonstrates excellent functional group tolerance. The mild reaction conditions (often room temperature to 55°C) allow for the successful conversion of substrates containing halogens (F, Cl, Br), methoxy groups, amino groups, and even heterocycles like pyridine and triazole without significant side reactions or dehalogenation.
Q: Is this process suitable for large-scale commercial manufacturing?
A: The process is highly amenable to scale-up due to its use of inexpensive and readily available reagents (alcohols, ammonia, air/oxygen) and simple catalyst systems. The absence of high-pressure equipment requirements and the use of common solvents like ethanol facilitate straightforward translation from laboratory benchtop to multi-ton industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrile Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively evaluated the copper-catalyzed aerobic oxidation pathway described in CN102964196B and integrated its principles into our process development workflow. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of nitrile intermediate meets the highest standards required for API synthesis.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for your specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and economics of your supply chain. Let us help you transform your nitrile sourcing strategy with safer, greener, and more efficient solutions.
