Advanced Manufacturing of Lorlatinib Intermediate via Photocatalytic Bromination
Advanced Manufacturing of Lorlatinib Intermediate via Photocatalytic Bromination
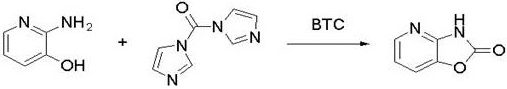
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical oncology therapeutics, particularly third-generation ALK inhibitors like Lorlatinib (PF-06463922). A pivotal breakthrough in this domain is detailed in patent CN111943885A, which discloses a novel synthetic method for 2-amino-5-bromo-3-hydroxypyridine, a key building block in the Lorlatinib value chain. This patent outlines a robust three-step sequence involving ring closure, photocatalytic bromination, and hydrolysis, starting from the commercially accessible 2-amino-3-hydroxypyridine. By shifting away from traditional hazardous reagents and complex coupling strategies, this technology offers a compelling alternative for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains. The integration of photochemical techniques and safer cyclization agents represents a significant leap forward in process chemistry, addressing both economic and environmental constraints inherent in modern cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lorlatinib and its precursors has relied on routes that present substantial challenges for large-scale operations. As illustrated in prior art such as Reaction Formula 1, conventional pathways often employ Suzuki-Miyaura coupling reactions which necessitate the use of expensive palladium catalysts and rigorous purification steps to remove trace metal residues to meet stringent regulatory limits. Furthermore, alternative routes involving the protection and activation of amino and hydroxyl groups, as seen in Reaction Formula 2, introduce multiple synthetic steps that drastically reduce overall atom economy and increase the accumulation of organic waste. These traditional methods frequently utilize N-bromosuccinimide (NBS) for bromination, which generates stoichiometric amounts of succinimide byproduct, complicating isolation procedures and increasing the burden on waste treatment facilities. The reliance on toxic gases like phosgene for cyclization in older methodologies also poses severe safety risks, requiring specialized containment infrastructure that drives up capital expenditure and operational complexity for manufacturers.
The Novel Approach
In stark contrast, the methodology described in CN111943885A streamlines the production of high-purity pharmaceutical intermediates through a concise three-step protocol that eliminates the need for transition metal catalysts and hazardous phosgene gas. The process initiates with a safe ring-closure reaction using a synergistic combination of bis(trichloromethyl) carbonate (BTC) and N,N'-carbonyldiimidazole (CDI), followed by a highly selective photocatalytic bromination using liquid bromine under UV irradiation. This innovative route bypasses the formation of difficult-to-remove succinimide impurities and avoids the use of heavy metals entirely, thereby simplifying the downstream processing workflow. The final hydrolysis step efficiently yields the target molecule with high conversion rates, demonstrating a clear pathway for the commercial scale-up of complex pharmaceutical intermediates. By reducing the step count and utilizing safer reagents, this approach not only enhances operator safety but also significantly lowers the cost of goods sold (COGS) through improved yield and reduced waste disposal requirements.
Mechanistic Insights into Photocatalytic Bromination and Safe Cyclization
The core innovation of this synthetic strategy lies in the mechanistic elegance of the ring-closure and bromination steps, which are designed to maximize selectivity while minimizing hazard. In the initial cyclization, the combination of BTC and CDI acts as a potent carbonyl source that activates the amino and hydroxyl groups of the starting pyridine derivative without releasing free phosgene gas into the reaction headspace. This dual-reagent system facilitates the formation of the oxazolopyridine core with exceptional molar yields ranging from 92% to 98%, ensuring that the crude product is of sufficient quality to proceed directly to the next stage without intermediate purification. This telescoping capability is crucial for maintaining throughput in a manufacturing environment, as it reduces solvent consumption and handling time. The subsequent bromination step leverages photochemical activation, where a specific photoinitiator, 2-hydroxy-2-methyl-1-phenyl-1-propanone, generates radical species under UV light to drive the substitution of hydrogen with bromine. This radical mechanism operates effectively at low temperatures between 0°C and 5°C, which suppresses side reactions and poly-bromination, ensuring high regioselectivity for the desired 5-bromo position.
Impurity control is inherently built into this mechanism through the choice of brominating agent and reaction conditions. Unlike thermal bromination methods that might require harsh Lewis acids or result in uncontrolled substitution patterns, the photocatalytic approach allows for precise control over the reaction kinetics via light intensity and temperature modulation. The use of liquid bromine instead of NBS means that the only byproduct generated is hydrogen bromide, which is easily neutralized during the workup, whereas NBS would leave behind succinimide solids that require extensive washing or chromatography to remove. This cleanliness of reaction translates directly to higher purity profiles in the final API intermediate, reducing the risk of genotoxic impurities carrying through to the final drug substance. Furthermore, the hydrolysis step is conducted under basic conditions followed by careful pH adjustment, which ensures the opening of the oxazole ring without degrading the sensitive amino functionality, resulting in a final product with liquid chromatography relative content exceeding 90% even before final recrystallization or column purification.

How to Synthesize 2-Amino-5-bromo-3-hydroxypyridine Efficiently
The execution of this synthesis requires careful attention to the photochemical parameters and reagent addition rates to ensure reproducibility and safety at scale. The process begins with the dissolution of the starting material in tetrahydrofuran, followed by the sequential addition of CDI and BTC under controlled cooling to manage the exotherm of the cyclization. Once the oxazolopyridine intermediate is secured, it is transferred to a quartz vessel equipped with a UV source for the critical bromination step, where the temperature must be strictly maintained near freezing to prevent thermal runaway or over-bromination. Detailed standardized operating procedures regarding light wavelength, stirring efficiency, and quenching protocols are essential for transferring this laboratory-scale success to pilot and production plants. For a comprehensive breakdown of the specific reagent quantities, reaction times, and workup procedures validated in the patent examples, please refer to the technical guide below.
- Perform ring closure of 2-amino-3-hydroxypyridine using a combination of BTC and CDI in THF to form 3H-oxazol[4,5-b]pyridin-2-one.
- Execute photocatalytic bromination using liquid bromine and a photoinitiator under UV light at 0-5°C to introduce the bromo group.
- Complete the synthesis via alkaline hydrolysis of the brominated intermediate to yield the final 2-amino-5-bromo-3-hydroxypyridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend beyond simple yield improvements. By eliminating the dependency on precious metal catalysts like palladium, which are subject to volatile market pricing and geopolitical supply risks, manufacturers can achieve a much more stable and predictable cost structure for their raw materials. The removal of NBS from the process inventory further reduces the burden of hazardous waste management, as the absence of succinimide byproducts simplifies the effluent treatment process and lowers disposal fees. Additionally, the use of readily available starting materials such as 2-amino-3-hydroxypyridine ensures that the supply chain is not bottlenecked by exotic or custom-synthesized precursors, thereby enhancing the overall resilience of the manufacturing network against global disruptions. These factors collectively contribute to a more agile and cost-effective production model that aligns with the strategic goals of modern pharmaceutical sourcing.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the avoidance of complex protection-deprotection sequences drastically reduce the direct material costs associated with producing this critical intermediate. Furthermore, the ability to telescope the first two steps without isolating the intermediate saves significant amounts of solvents and labor hours, leading to substantial operational expenditure savings. The reduction in waste generation, specifically the absence of succinimide solids, lowers the environmental compliance costs and waste treatment fees, contributing to a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of commodity chemicals like liquid bromine and BTC, which are produced at massive scales globally, ensuring consistent availability and price stability. The simplified process flow reduces the number of unit operations required, which in turn shortens the overall production cycle time and allows for faster response to fluctuating market demand. This agility is crucial for maintaining continuous supply to downstream API manufacturers, minimizing the risk of stockouts that could delay clinical trials or commercial drug launches.
- Scalability and Environmental Compliance: The photochemical step, while requiring specific equipment, is highly amenable to scale-up using modern flow chemistry reactors or large batch vessels with optimized lighting arrays, ensuring that production volumes can be increased to meet commercial needs without compromising quality. The greener profile of the synthesis, characterized by the avoidance of phosgene gas and heavy metals, aligns perfectly with increasingly strict environmental regulations and corporate sustainability mandates. This compliance advantage facilitates smoother regulatory approvals and enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on safety, efficiency, and scalability. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams negotiating supply contracts based on this technology.
Q: How does this method improve safety compared to traditional phosgene usage?
A: The novel process replaces highly toxic phosgene gas with a safer combination of bis(trichloromethyl) carbonate (BTC) and N,N'-carbonyldiimidazole (CDI), significantly reducing operational hazards and regulatory burdens.
Q: What are the advantages of using photocatalytic bromination over NBS?
A: Using liquid bromine with a photoinitiator eliminates the generation of succinimide byproducts associated with N-bromosuccinimide (NBS), simplifying downstream purification and reducing solid waste disposal costs.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available starting materials and avoids expensive transition metal catalysts, making it highly scalable and economically viable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-bromo-3-hydroxypyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for high-value oncology intermediates like 2-amino-5-bromo-3-hydroxypyridine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our facility is equipped to handle the specific requirements of photochemical reactions and hazardous reagent handling, guaranteeing a safe and compliant manufacturing environment for your most challenging projects.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and reliability for your organization. Let us be your partner in advancing the availability of life-saving medications through superior chemical engineering and supply chain excellence.
