Advanced Synthesis of Chiral Salan Ligands for Asymmetric Catalysis and Commercial Scale-up
The landscape of asymmetric catalysis relies heavily on the availability of high-performance chiral ligands, and the recent disclosure in patent CN102786466A presents a significant advancement in the preparation of chiral Salan ligands. These specific ligands, identified as (1R,2R)-N1,N2-bis(2-(2-pyridinemethoxy)benzyl)cyclohexane-1,2-diamine (Salan I) and its diphenyl analogue (Salan II), represent a class of compounds with exceptional catalytic activity in metal-mediated transformations. The patent details a robust, three-step synthetic pathway that overcomes historical challenges associated with regioselectivity and purification. For R&D directors and procurement specialists seeking reliable sources for complex pharmaceutical intermediates, this methodology offers a compelling alternative to legacy processes, promising enhanced purity profiles and operational simplicity. By strategically sequencing the etherification prior to diamine coupling, the inventors have effectively eliminated the formation of difficult-to-separate byproducts that have long plagued this chemical space.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar Salan-type structures has been fraught with chemoselectivity issues, particularly when attempting to introduce multiple functional groups onto a diamine backbone simultaneously. Conventional literature often describes routes where the pyridine-methyl moiety is attached directly to the nitrogen atoms of the diamine, or where competitive alkylation occurs between the amine and phenolic hydroxyl groups. As illustrated in the prior art analysis within the patent, attempting to control these competing reactions through experimental conditions alone often results in inevitable byproduct formation. 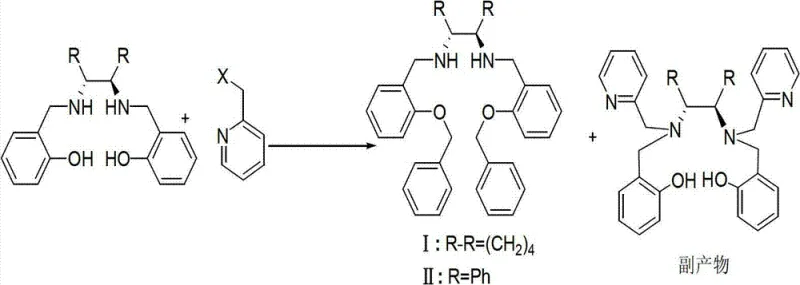 . This lack of control drastically reduces the overall reaction yield and significantly increases the difficulty of downstream purification. For a supply chain manager, this translates to higher costs due to material loss and extended processing times required to isolate the target molecule from a complex mixture of isomers and oligomers.
. This lack of control drastically reduces the overall reaction yield and significantly increases the difficulty of downstream purification. For a supply chain manager, this translates to higher costs due to material loss and extended processing times required to isolate the target molecule from a complex mixture of isomers and oligomers.
The Novel Approach
The innovative strategy outlined in CN102786466A fundamentally reorders the synthetic logic to prioritize selectivity. Instead of struggling with competing nucleophiles in a single pot, the process begins with the selective etherification of salicylaldehyde. This creates a stable aldehyde intermediate, 2-(2-pyridinemethoxy)benzaldehyde, which is then condensed with the chiral diamine. Finally, a mild reduction step yields the target Salan ligand. 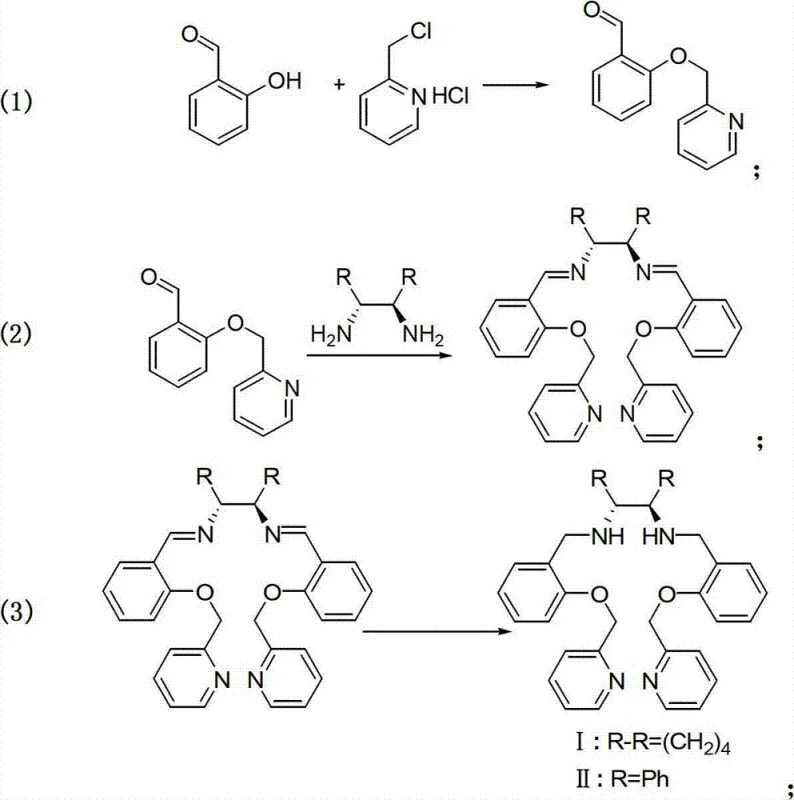 . This stepwise approach ensures that the phenolic oxygen is protected as an ether before the amine functionality is engaged, thereby eliminating the possibility of O-alkylation side reactions during the final assembly. The result is a process characterized by high reaction selectivity, mild conditions, and simple, controllable operations that are ideal for cost reduction in pharmaceutical intermediate manufacturing.
. This stepwise approach ensures that the phenolic oxygen is protected as an ether before the amine functionality is engaged, thereby eliminating the possibility of O-alkylation side reactions during the final assembly. The result is a process characterized by high reaction selectivity, mild conditions, and simple, controllable operations that are ideal for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Stepwise Ligand Assembly
The success of this synthesis lies in the precise control of nucleophilic substitution and condensation equilibria. In the first stage, the phenolic hydroxyl group of salicylaldehyde acts as a nucleophile, attacking the chloromethyl group of 2-chloromethylpyridine hydrochloride. The use of a strong base, specifically sodium hydride in dry DMF, facilitates the deprotonation of the phenol, driving the formation of the ether linkage at 80°C. This step is critical because it installs the pyridine coordinating arm without disturbing the aldehyde functionality needed for the subsequent step. Following isolation, the aldehyde intermediate undergoes a condensation reaction with chiral diamines such as (1R,2R)-1,2-cyclohexanediamine or (1R,2R)-1,2-diphenylethylenediamine. The addition of anhydrous magnesium sulfate or sodium sulfate serves a dual purpose: it acts as a drying agent to shift the equilibrium toward imine formation by removing water, and it helps maintain the integrity of the reaction medium in ethanol solvent.
The final mechanistic transformation involves the stereospecific reduction of the bis-imine intermediate to the secondary amine. Utilizing sodium borohydride in methanol allows for a controlled reduction at temperatures ranging from 0°C to 65°C. This mild reducing environment is crucial for preserving the stereochemical integrity of the chiral backbone, ensuring that the (1R,2R) configuration is maintained throughout the synthesis. The mechanism avoids harsh conditions that could lead to racemization, a common pitfall in chiral ligand synthesis. Furthermore, the choice of methanol as a solvent for the reduction step ensures good solubility of the polar imine intermediate while allowing for easy workup and isolation of the final viscous solid product. This mechanistic clarity provides R&D teams with confidence in the reproducibility of the process across different batch sizes.
How to Synthesize Chiral Salan Ligands Efficiently
Implementing this synthesis requires strict adherence to the sequential addition of reagents and moisture control, particularly during the imine formation stage. The patent provides specific molar ratios and solvent volumes that optimize the yield, such as using a 2:1 to 4:1 molar ratio of aldehyde to diamine to drive the double condensation to completion. While the laboratory examples demonstrate the feasibility on a 100mL scale, the principles of operation remain consistent for larger vessels. For detailed standard operating procedures regarding exact reagent quantities, stirring rates, and quenching protocols, please refer to the standardized synthesis guide below.
- Prepare 2-(2-pyridinemethoxy)benzaldehyde by reacting salicylaldehyde with 2-chloromethylpyridine hydrochloride using sodium hydride in DMF at 80°C.
- Condense the resulting aldehyde with chiral diamines (cyclohexanediamine or diphenylethylenediamine) in ethanol with a drying agent to form the imine intermediate.
- Reduce the imine intermediate using sodium borohydride in methanol at temperatures between 0°C and 65°C to obtain the final Salan ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial advantages for organizations looking to secure a stable supply of high-purity chiral ligands. The primary benefit stems from the drastic simplification of the purification workflow. By eliminating the formation of regioisomeric byproducts, the need for extensive chromatographic separation is reduced, which directly correlates to lower solvent consumption and reduced waste generation. This efficiency is a key driver for cost reduction in fine chemical manufacturing, as it minimizes the loss of valuable chiral starting materials that are often expensive to procure. Additionally, the use of commodity chemicals like salicylaldehyde and 2-chloromethylpyridine hydrochloride ensures that the raw material supply chain is robust and less susceptible to market volatility compared to routes requiring exotic precursors.
- Cost Reduction in Manufacturing: The streamlined three-step process eliminates the need for complex protection-deprotection strategies that typically add multiple unit operations to a synthesis. By avoiding the generation of hard-to-separate impurities, the overall material throughput is improved, leading to significant operational savings. The ability to use crude imine intermediates directly in the reduction step without prior purification further accelerates the production timeline and reduces labor costs associated with intermediate isolation.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials and standard solvents such as ethanol, methanol, and DMF means that production is not dependent on single-source suppliers for niche reagents. This diversification of the supply base enhances continuity and reduces the risk of production stoppages. Furthermore, the mild reaction temperatures (ranging from ambient to 80°C) allow the process to be run in standard glass-lined or stainless steel reactors without requiring specialized cryogenic or high-pressure equipment.
- Scalability and Environmental Compliance: The process is inherently scalable due to its linear nature and the absence of exothermic runaway risks associated with the reagents used. The reduction step using sodium borohydride is well-understood in industrial settings and generates manageable boron waste streams that can be treated using standard effluent protocols. This aligns with modern green chemistry principles by maximizing atom economy in the condensation step and minimizing the use of hazardous heavy metal catalysts often found in alternative coupling methods.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this patented technology. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating these ligands into their own catalytic processes. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation.
Q: How does this new synthesis method improve selectivity compared to traditional routes?
A: Traditional methods often suffer from competitive reactions between amine and phenolic hydroxyl groups. This patented method avoids this by introducing the pyridine-methyl group first via etherification, ensuring high regioselectivity before the diamine coupling step.
Q: What are the typical yields for Salan I and Salan II using this protocol?
A: According to the patent data, the process achieves robust yields. For example, Salan I was obtained with a two-step yield of 63%, while Salan II achieved a two-step yield of 71.7%, demonstrating high efficiency suitable for production.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the route utilizes common solvents like ethanol, methanol, and DMF, and operates at moderate temperatures (0°C to 80°C). The avoidance of complex purification steps for intermediates makes it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Salan Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands play in the development of next-generation pharmaceuticals and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify enantiomeric excess and chemical purity. Our capability to adapt the synthesis described in CN102786466A allows us to offer a reliable supply of these complex intermediates to global partners.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, we are ready to provide the data-driven insights you need. Contact us today to explore how our advanced manufacturing capabilities can accelerate your R&D timelines and optimize your production costs.
