Advanced Synthesis Of Cholesterol-Naphthalimide Hybrids For Oncology And Material Science Applications
Advanced Synthesis Of Cholesterol-Naphthalimide Hybrids For Oncology And Material Science Applications
The pharmaceutical and advanced materials sectors are constantly seeking novel molecular architectures that combine bioactivity with structural stability. Patent CN107698648B, published in August 2020, introduces a significant breakthrough in the field of bio-organic synthesis through the development of cholesterol-containing naphthalimide derivatives. These compounds represent a sophisticated fusion of two distinct functional domains: the planar, rigid naphthalimide core known for its DNA intercalating properties, and the lipophilic cholesterol moiety which enhances membrane permeability and cellular uptake. This dual-functionality design addresses critical challenges in targeted anticancer therapy while simultaneously opening avenues for applications in liquid crystal display technologies. For R&D directors and procurement specialists evaluating new pipeline candidates, understanding the synthetic accessibility and structural versatility of these hybrids is paramount for strategic planning and resource allocation.
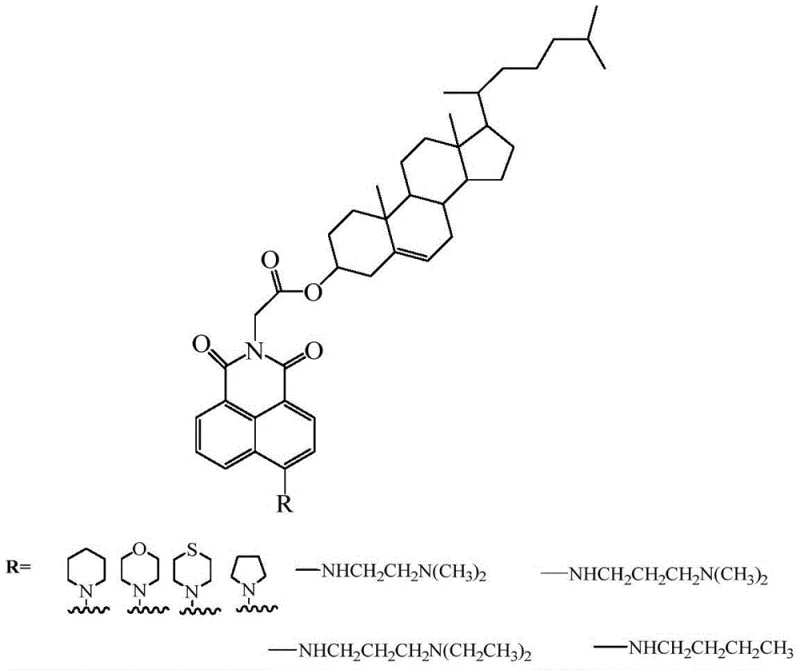
The core innovation lies in the modular construction of the molecule, where the naphthalimide parent body is systematically modified at the imide nitrogen and the aromatic ring positions. By substituting the bromine atom at the 4-position of the naphthalic anhydride precursor with various cyclic or aliphatic amines, researchers can finely tune the electronic and steric properties of the final drug candidate. Furthermore, the use of glycine as a bridging chain allows for the efficient conjugation of cholesterol via a robust ester linkage. This strategic molecular design not only aims to improve the therapeutic index against resistant tumor strains but also leverages the inherent mesogenic properties of cholesterol to create materials with specific liquid crystalline phases, demonstrating the versatile utility of this chemical platform across different high-value industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex naphthalimide-drug conjugates has been plagued by multi-step sequences that suffer from low overall yields and harsh reaction conditions. Traditional approaches often require the protection and deprotection of sensitive functional groups, leading to increased waste generation and higher production costs. Moreover, direct esterification of sterols with bulky aromatic acids frequently encounters steric hindrance issues, necessitating the use of expensive coupling reagents or forcing conditions that can degrade the thermally sensitive cholesterol backbone. In many legacy processes, the lack of a flexible linker results in poor solubility profiles, complicating downstream formulation and biological testing. These inefficiencies create significant bottlenecks for supply chain managers who struggle to secure consistent quantities of high-purity intermediates required for preclinical and clinical trials.
The Novel Approach
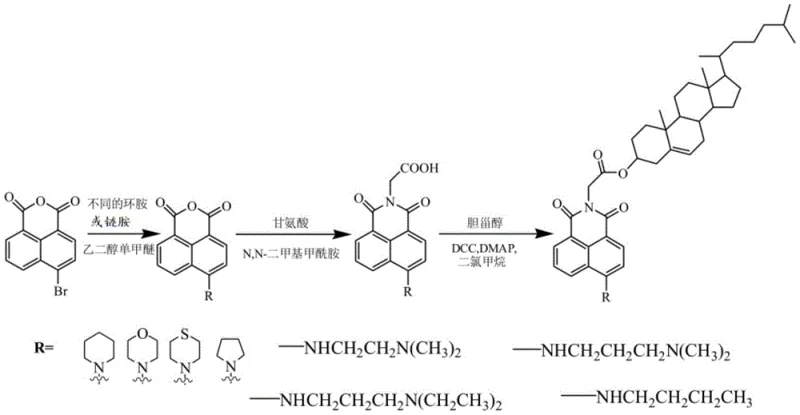
The methodology outlined in CN107698648B offers a streamlined, three-step synthetic pathway that circumvents these traditional hurdles by employing a convergent strategy. Instead of building the molecule linearly from scratch, the process utilizes readily available 4-bromo-1,8-naphthalic anhydride as a central hub for diversification. The initial nucleophilic aromatic substitution with amines proceeds under relatively mild conditions in ethylene glycol monomethyl ether, providing high conversion rates without the need for transition metal catalysts. Subsequent condensation with glycine introduces a carboxylic acid handle in a single pot, effectively setting up the molecule for the final conjugation step. This logical progression minimizes purification operations between stages, thereby reducing solvent consumption and processing time. The final esterification utilizes the well-established DCC/DMAP coupling system, which is highly effective for forming ester bonds between sterically hindered partners, ensuring that the cholesterol unit is attached efficiently without compromising its stereochemical integrity.
Mechanistic Insights into Nucleophilic Substitution and DCC-Mediated Esterification
The success of this synthetic route relies heavily on the precise control of reaction mechanisms at each stage. The first step involves a nucleophilic aromatic substitution where the electron-rich amine attacks the electron-deficient carbon at the 4-position of the naphthalic anhydride ring. The presence of the two flanking carbonyl groups activates the ring towards nucleophilic attack, facilitating the displacement of the bromide leaving group. This reaction is typically conducted at reflux temperatures to overcome the activation energy barrier, yet the choice of ethylene glycol monomethyl ether as a solvent ensures that the reactants remain in solution while stabilizing the transition state. The result is the formation of a stable 4-amino-1,8-naphthalic anhydride intermediate, which serves as the foundational scaffold for all subsequent derivatization. The versatility of this step allows for the rapid generation of a library of analogues by simply swapping the amine source, a feature highly valued in medicinal chemistry for structure-activity relationship studies.
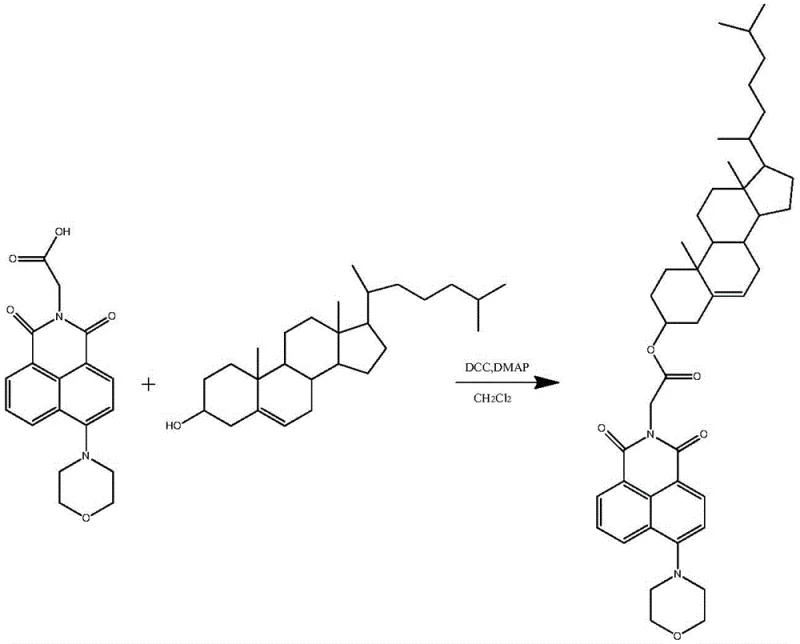
In the final conjugation stage, the mechanism shifts to carbodiimide-mediated esterification, a cornerstone reaction in peptide and natural product synthesis. The carboxylic acid group on the glycine-linked naphthalimide reacts with dicyclohexylcarbodiimide (DCC) to form an reactive O-acylisourea intermediate. This activated species is highly susceptible to nucleophilic attack by the hydroxyl group of cholesterol. The addition of 4-dimethylaminopyridine (DMAP) acts as a nucleophilic catalyst, accelerating the acyl transfer process and suppressing side reactions such as N-acylurea formation. This mechanistic pathway is crucial for achieving high yields in the coupling of bulky molecules, as it lowers the energy barrier for ester bond formation. By operating at room temperature in dichloromethane, the process preserves the delicate double bond within the cholesterol steroid nucleus, preventing isomerization or degradation that could occur under acidic or high-thermal conditions typical of Fischer esterification.
How to Synthesize Cholesterol-Naphthalimide Derivatives Efficiently
The synthesis of these high-value intermediates requires strict adherence to stoichiometric ratios and temperature controls to maximize yield and purity. The process begins with the preparation of the amine-substituted anhydride, followed by ring-opening with glycine, and concludes with the cholesterol coupling. Each step generates specific byproducts, such as dicyclohexylurea in the final step, which must be removed via column chromatography or recrystallization to meet pharmaceutical grade standards. The detailed standardized synthesis steps for producing these compounds are provided in the guide below.
- Perform nucleophilic substitution of 4-bromo-1,8-naphthalic anhydride with cyclic or aliphatic amines in ethylene glycol monomethyl ether to form the 4-amino intermediate.
- Condense the resulting anhydride intermediate with glycine in DMF at elevated temperatures to introduce the carboxylic acid bridge chain.
- Execute the final esterification between the carboxylic acid intermediate and cholesterol using DCC and DMAP catalysts in dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents compelling economic and logistical benefits. The reliance on commodity chemicals such as glycine, cholesterol, and common organic solvents significantly reduces the risk of raw material shortages that often plague specialized fine chemical manufacturing. Unlike processes dependent on scarce precious metal catalysts or proprietary reagents, this method utilizes widely available reagents that can be sourced from multiple global suppliers, enhancing supply chain resilience. Furthermore, the elimination of transition metals from the synthetic sequence simplifies the purification workflow, removing the need for expensive metal scavenging resins and extensive analytical testing for residual metals, which directly translates to reduced operational expenditures and faster batch release times.
- Cost Reduction in Manufacturing: The streamlined three-step process minimizes the number of unit operations required, leading to substantial savings in labor, energy, and solvent usage. By avoiding the use of expensive transition metal catalysts and complex protecting group strategies, the overall cost of goods sold is significantly optimized. The high atom economy of the nucleophilic substitution step ensures that the majority of the starting material mass is incorporated into the final product, reducing waste disposal costs associated with hazardous byproducts. Additionally, the ability to perform reactions at ambient pressure and moderate temperatures lowers the capital expenditure requirements for reactor infrastructure, making this process economically viable for both pilot-scale and large-scale commercial production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, particularly 4-bromo-1,8-naphthalic anhydride and cholesterol, are established industrial chemicals with stable market availability. This reduces the lead time for high-purity pharmaceutical intermediates by mitigating the risks associated with custom synthesis of exotic precursors. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This reliability is critical for maintaining continuous production schedules and meeting the stringent delivery deadlines demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, as it avoids exothermic runaway risks and uses solvents that are easily recovered and recycled through distillation. The absence of heavy metals aligns with increasingly strict environmental regulations regarding effluent discharge and product safety, facilitating easier regulatory approval for new drug applications. The solid byproduct formed during the esterification step, dicyclohexylurea, can be filtered off simply, simplifying the work-up procedure and reducing the volume of liquid waste generated. This green chemistry profile supports corporate sustainability goals while ensuring that the manufacturing process remains compliant with international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these derivatives, based on the specific embodiments and data presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this chemistry into their existing pipelines.
Q: What is the primary advantage of using glycine as a linker in this synthesis?
A: Glycine serves as a simple, non-toxic, and metabolically compatible bridge chain that facilitates the esterification with cholesterol while maintaining the structural integrity required for DNA intercalation activity.
Q: Can this synthetic route be scaled for industrial production of liquid crystal materials?
A: Yes, the process utilizes standard organic solvents like dichloromethane and DMF, and reagents such as DCC which are commercially available in bulk, making the transition from lab scale to commercial manufacturing feasible without exotic equipment.
Q: How does the choice of amine substituent affect the biological activity?
A: The patent demonstrates that varying the amine group (e.g., morpholine vs. pyrrolidine) significantly alters the cytotoxicity profile against different cancer cell lines, allowing for structure-activity relationship optimization during drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholesterol-Naphthalimide Derivatives Supplier
As the demand for specialized oncology intermediates and advanced electronic materials continues to grow, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of cholesterol-naphthalimide derivatives meets the highest industry standards for both pharmaceutical and material science applications.
We invite you to collaborate with our technical team to explore how this innovative synthetic route can be optimized for your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your volume needs. Let us help you accelerate your development timeline with reliable, high-quality chemical solutions.
