Advanced Synthesis of Phthalimide-Containing Naphthalimides for Scalable Oncology Intermediate Production
The pharmaceutical industry is constantly seeking novel molecular scaffolds that can overcome the limitations of existing chemotherapeutic agents, particularly regarding toxicity and selectivity. Patent CN110804039B discloses a significant advancement in the field of bio-organic synthesis by introducing a class of phthalimide-containing 1,8-naphthalic anhydride derivatives. These compounds are designed to function as potent DNA intercalators, leveraging the planar structure of the naphthalimide core to insert between DNA base pairs, thereby disrupting replication in rapidly dividing tumor cells. The innovation lies in the strategic modification of the naphthalimide parent ring with a phthalimide pharmacophore, which has known anti-angiogenic and TNF-α inhibitory properties. This dual-mechanism approach aims to enhance antitumor activity while mitigating the severe side effects often associated with first-generation DNA intercalators like Amonafide. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of these intermediates is crucial for developing next-generation oncology pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional naphthalimide-based antitumor agents, such as Mitonafide and Amonafide, have demonstrated efficacy in clinical trials but are severely hampered by their high toxicity profiles and limited therapeutic windows. The primary chemical limitation of these conventional structures is their lack of selectivity; they often interact indiscriminately with DNA in both malignant and healthy tissues, leading to systemic toxicity that restricts dosage and clinical utility. Furthermore, the synthetic routes for many early-generation intercalators often involve harsh reaction conditions or expensive catalysts that are difficult to scale economically. The structural rigidity of the parent naphthalimide ring also limits the ability to fine-tune pharmacokinetic properties through simple derivatization. Consequently, there is a pressing need in the fine chemical industry for modified scaffolds that retain DNA-binding affinity but offer improved safety margins and more robust, cost-effective manufacturing pathways.
The Novel Approach
The methodology outlined in patent CN110804039B presents a transformative solution by hybridizing the naphthalimide core with a phthalimide moiety via a flexible alcohol amine bridge chain. This structural innovation allows for significant modulation of the molecule's electronic and steric properties, enhancing its ability to intercalate into DNA while reducing off-target toxicity. The synthetic strategy is remarkably efficient, starting from readily available 4-bromo-1,8-naphthalic anhydride and utilizing a nucleophilic substitution to introduce a piperidinyl group, which significantly boosts biological activity. The subsequent esterification step links the two pharmacophores without requiring transition metal catalysts, simplifying the purification process and reducing heavy metal contamination risks. 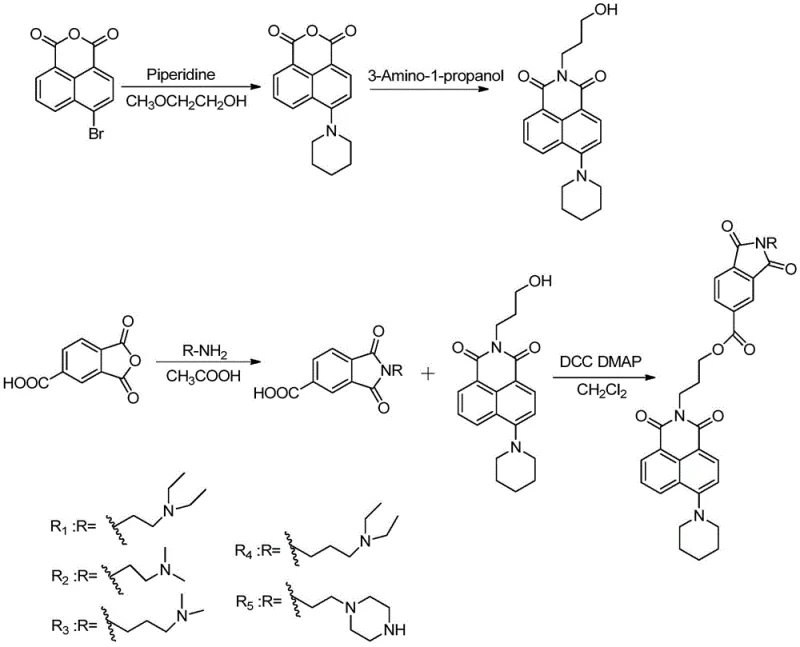
Mechanistic Insights into DCC-Mediated Esterification and DNA Intercalation
The core chemical transformation in this synthesis is the final coupling step, which utilizes N,N'-dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) to form an ester bond between the hydroxy-propyl-naphthalimide intermediate and the carboxy-phthalimide derivative. This mechanism is critical for maintaining the integrity of the sensitive imide rings while ensuring high conversion rates. The DCC activates the carboxylic acid group on the phthalimide intermediate to form an O-acylisourea, which is then attacked by the hydroxyl group of the naphthalimide side chain. DMAP acts as a nucleophilic catalyst, accelerating the acyl transfer and preventing side reactions such as N-acylurea formation. This mild, room-temperature reaction condition is particularly advantageous for preserving the stereochemical integrity of the molecule and avoiding thermal degradation of the complex heterocyclic system.
From a biological perspective, the resulting structure functions through a dual-mode mechanism of action. The planar naphthalimide system inserts itself between DNA base pairs, causing unwinding and lengthening of the DNA helix, which inhibits topoisomerase activity and blocks replication. Simultaneously, the appended phthalimide group contributes anti-angiogenic effects by inhibiting TNF-α production, thereby starving the tumor of its blood supply. Experimental data from the patent indicates that compounds like N3 exhibit strong selectivity, with IC50 values against Hela cells as low as 23.3 μM, while showing significantly reduced toxicity against normal liver cells (HL7702). 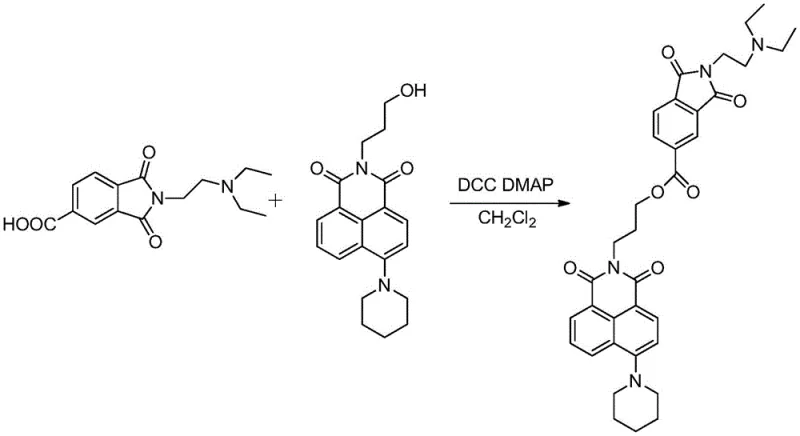
How to Synthesize Phthalimide-Containing Naphthalimides Efficiently
The synthesis of these high-value pharmaceutical intermediates follows a convergent linear sequence that is well-suited for process optimization. The initial step involves the nucleophilic displacement of the bromine atom on the naphthalic anhydride core using piperidine in ethylene glycol monomethyl ether at elevated temperatures. This step is high-yielding and establishes the critical 4-position substitution required for biological activity. Subsequent reaction with 3-amino-1-propanol introduces the necessary linker arm, creating a versatile handle for further functionalization. The parallel synthesis of the phthalimide acid component allows for rapid library generation by varying the amine substituents, enabling structure-activity relationship (SAR) studies without altering the core process. Detailed standardized synthesis steps see the guide below.
- Perform nucleophilic substitution on 4-bromo-1,8-naphthalic anhydride using piperidine in ethylene glycol monomethyl ether at 125°C.
- Condense the resulting intermediate with 3-amino-1-propanol to introduce a hydroxy-propyl bridge chain.
- Execute final esterification using DCC and DMAP to couple the naphthalimide core with the phthalimide pharmacophore.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the commercial viability of a new chemical entity is often determined by the robustness of its synthesis and the availability of raw materials. The route described in CN110804039B offers substantial advantages in terms of cost reduction in pharmaceutical intermediates manufacturing. By avoiding the use of precious metal catalysts such as palladium or platinum, which are common in cross-coupling reactions, the process eliminates the need for expensive metal scavenging steps and reduces the risk of heavy metal residues in the final API. This not only lowers the direct material costs but also simplifies the regulatory compliance burden associated with elemental impurity limits. Furthermore, the starting materials, including 4-bromo-1,8-naphthalic anhydride and various aliphatic amines, are commodity chemicals with stable global supply chains, ensuring consistent availability.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to maximize atom economy and minimize waste generation. The high yield observed in the initial substitution step (over 90%) ensures that expensive starting materials are efficiently converted into intermediates, reducing the overall cost per kilogram of the final product. Additionally, the use of standard organic solvents like dichloromethane and ethanol facilitates solvent recovery and recycling, further driving down operational expenses. The elimination of transition metals also means that purification can be achieved through standard silica gel chromatography or crystallization, avoiding the need for specialized preparative HPLC or complex extraction protocols.
- Enhanced Supply Chain Reliability: The modularity of this synthesis allows for flexible production scheduling. Since the two main fragments (the naphthalimide core and the phthalimide acid) are synthesized independently before the final coupling, manufacturers can stockpile intermediates to buffer against supply disruptions. This decoupling of the synthesis stream enhances the reliability of high-purity pharmaceutical intermediates supply, as a bottleneck in one branch does not necessarily halt the entire production line. The use of common reagents also means that secondary sourcing is readily available, mitigating the risk of single-supplier dependency for critical raw materials.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at atmospheric pressure, making the process inherently safer and easier to scale from laboratory to commercial production. The absence of high-pressure hydrogenation or cryogenic conditions reduces the capital expenditure required for specialized reactor equipment. From an environmental perspective, the process generates manageable waste streams, primarily organic solvents and urea byproducts from the DCC coupling, which can be treated using standard industrial waste management protocols. This aligns with modern green chemistry principles and facilitates smoother environmental impact assessments for new facility approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these naphthalimide derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating these intermediates into your existing drug development or manufacturing workflows.
Q: How does the phthalimide modification improve antitumor selectivity compared to traditional naphthalimides?
A: The introduction of the phthalimide pharmacophore enhances DNA intercalation capabilities while reducing toxicity to normal cells, as evidenced by higher IC50 values in normal hepatocyte lines compared to cancer cell lines.
Q: What are the critical reaction conditions for the final esterification step?
A: The final coupling requires anhydrous dichloromethane as a solvent, with DCC as the dehydrating agent and DMAP as the catalyst, stirred at room temperature for 24 hours to ensure high purity.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the route utilizes readily available starting materials like 4-bromo-1,8-naphthalic anhydride and avoids expensive transition metal catalysts, facilitating easier scale-up and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phthalimide-Containing Naphthalimide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent chemistry into reliable commercial supply. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and selectivity demonstrated in the laboratory are maintained at an industrial scale. We possess stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and impurity profiles of complex heterocyclic intermediates. Our commitment to quality ensures that every batch meets the exacting standards required for oncology drug development, providing you with a secure foundation for your clinical and commercial programs.
We invite you to collaborate with us to optimize your supply chain for these advanced antitumor intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating these high-purity pharmaceutical intermediates into your pipeline. Let us handle the complexities of synthesis and scale-up so you can focus on delivering life-saving therapies to patients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
