Scalable Synthesis of 5-Nitro-6-Methylnicotinic Acid Ethyl Ester for PARP1 Inhibitors
Introduction to Advanced Pyridine Intermediate Synthesis
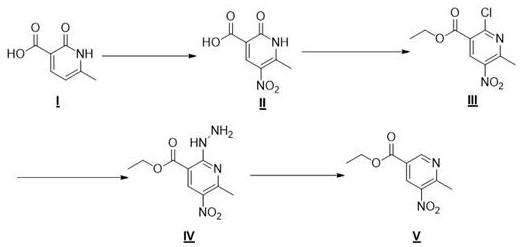
The pharmaceutical landscape is continuously evolving with the demand for potent anticancer and antibacterial agents, driving the need for robust synthetic routes to complex heterocyclic intermediates. Patent CN114605320A discloses a groundbreaking methodology for the synthesis of 5-nitro-6-methylnicotinic acid ethyl ester, a pivotal building block for PARP1 inhibitors and fourth-generation quinolone antibiotics. This innovation addresses the historical lack of published synthetic methods for this specific scaffold, offering a streamlined four-step pathway that begins with the commercially accessible 2-hydroxy-6-methylnicotinic acid. By leveraging classical organic transformations such as electrophilic aromatic substitution and nucleophilic displacement, coupled with a unique oxidative deamination finale, this process ensures high purity and operational simplicity. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable supply chain for high-value oncology intermediates while mitigating the risks associated with proprietary or obscure chemical routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of pyridine rings, particularly at the 5-position with a nitro group while maintaining a 6-methyl substituent, presents formidable challenges due to the electron-deficient nature of the heterocycle. Conventional approaches often suffer from poor regioselectivity, requiring harsh conditions that can lead to the degradation of sensitive functional groups or the formation of intractable impurity profiles. Furthermore, existing routes for similar nicotinic acid derivatives frequently rely on multi-step sequences involving expensive protecting groups or precious metal catalysts, which drastically inflate the cost of goods sold (COGS). The absence of a direct, published method for this specific intermediate has historically forced manufacturers to rely on inefficient custom syntheses that lack scalability and reproducibility. These legacy methods often result in low overall yields and necessitate complex purification protocols, creating bottlenecks in the supply of critical active pharmaceutical ingredient (API) precursors.
The Novel Approach
The disclosed invention introduces a highly efficient strategy that circumvents these traditional hurdles by utilizing a strategic sequence of nitration, chlorination-esterification, hydrazinolysis, and oxidative deamination. This route capitalizes on the activating effect of the hydroxyl group in the starting material to facilitate regioselective nitration, followed by a tandem chlorination and esterification step that installs both the leaving group and the ethyl ester moiety simultaneously. The subsequent displacement with hydrazine hydrate sets the stage for the final, elegant removal of the amino functionality using a copper sulfate system, restoring the aromaticity without compromising the nitro or methyl groups. This logical progression not only simplifies the operational workflow but also utilizes inexpensive, commodity-grade reagents, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The holistic view of this synthesis demonstrates a clear path toward industrial viability.
Mechanistic Insights into Copper-Mediated Oxidative Deamination
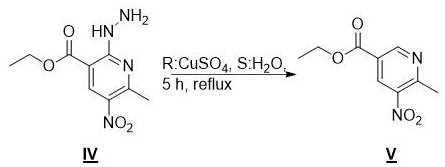
The cornerstone of this synthetic innovation lies in the final transformation, where the 2-hydrazino intermediate is converted into the target pyridine derivative through a copper-mediated oxidative process. In this step, the hydrazino group acts as a masked hydrogen equivalent, which is subsequently excised under oxidative conditions provided by the copper(II) sulfate species in an aqueous environment. The mechanism likely involves the coordination of the hydrazine nitrogen to the copper center, facilitating electron transfer that leads to the extrusion of nitrogen gas and the regeneration of the C-H bond at the 2-position of the pyridine ring. This approach is superior to direct reduction methods which might inadvertently reduce the sensitive 5-nitro group, thereby preserving the oxidation state required for downstream coupling reactions. The use of water as a co-solvent in this step further enhances the green chemistry profile of the process by reducing the reliance on volatile organic compounds during the critical final purification stage.
Furthermore, the preceding steps are meticulously optimized to control impurity generation, particularly during the initial nitration and the chlorination phases. The nitration is conducted at controlled low temperatures initially to prevent over-nitration or oxidative degradation, followed by a heating phase to drive the reaction to completion, ensuring a high-purity nitro-acid intermediate. Similarly, the use of phosphorus oxychloride with a catalytic amount of DMF generates a reactive Vilsmeier-Haack type complex that efficiently converts the hydroxyl group to a chloro group while simultaneously esterifying the carboxylic acid. This dual-functionality step minimizes unit operations and reduces the accumulation of partially reacted species, resulting in a cleaner crude profile that simplifies downstream isolation. Such precise control over reaction parameters is essential for meeting the stringent purity specifications required for clinical-grade materials.
How to Synthesize 5-Nitro-6-Methylnicotinic Acid Ethyl Ester Efficiently
The execution of this synthesis requires careful attention to thermal management and stoichiometric ratios, particularly during the exothermic nitration and the high-temperature chlorination steps. Operators must adhere to the specified temperature gradients, such as maintaining the nitration mixture between -5°C and 0°C during acid addition before ramping to 90°C, to maximize yield and safety. The detailed standardized synthetic steps below outline the precise conditions for each transformation, ensuring reproducibility from the laboratory bench to the pilot plant scale.
- Nitration of 2-hydroxy-6-methylnicotinic acid using mixed acid (H2SO4/HNO3) at controlled temperatures to form the nitro-intermediate.
- Chlorination and simultaneous esterification using phosphorus oxychloride and ethanol to generate the ethyl ester chloride derivative.
- Displacement of the chloro group with hydrazine hydrate followed by copper sulfate-mediated oxidative deamination to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic route offers substantial strategic benefits for supply chain managers seeking to diversify their vendor base and reduce dependency on single-source suppliers for critical oncology intermediates. By utilizing a pathway that relies on abundant, non-proprietary starting materials like 2-hydroxy-6-methylnicotinic acid, the risk of raw material shortages is significantly mitigated, ensuring greater supply continuity even during market fluctuations. The elimination of exotic catalysts and the use of standard industrial solvents such as ethanol and chlorobenzene further streamline the procurement process, allowing for easier sourcing and inventory management across global manufacturing sites. Additionally, the robustness of the reaction conditions suggests a high tolerance for scale-up, reducing the technical risk associated with technology transfer from R&D to commercial production facilities.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive transition metal catalysts with economical copper sulfate and utilizing phosphorus oxychloride for dual-purpose functionalization. This consolidation of steps reduces the total number of reactor charges and workup procedures, leading to lower labor and utility costs per kilogram of produced intermediate. Furthermore, the high purity achieved in the early stages minimizes the need for resource-intensive chromatographic purifications, relying instead on crystallization and extraction which are far more cost-effective at scale. These factors collectively contribute to a leaner manufacturing model that enhances margin potential for the final API.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized reagents ensures that the supply chain remains resilient against geopolitical or logistical disruptions. Since the reagents such as concentrated sulfuric acid, nitric acid, and hydrazine hydrate are produced globally in massive quantities, lead times for raw material acquisition are inherently shorter and more predictable. This stability allows for more accurate production planning and inventory forecasting, enabling pharmaceutical companies to maintain optimal stock levels of the intermediate without the fear of sudden supply cutoffs. Consequently, this reliability translates directly into improved on-time delivery performance for the downstream drug manufacturers.
- Scalability and Environmental Compliance: The synthetic design inherently supports commercial scale-up of complex pharmaceutical intermediates by avoiding hazardous reagents that require specialized containment or waste treatment infrastructure. The aqueous workup in the final step and the ability to recycle solvents like chlorobenzene align with modern environmental, health, and safety (EHS) standards, reducing the regulatory burden on manufacturing sites. The process generates manageable waste streams that can be treated using conventional methods, facilitating easier permitting and operation in diverse regulatory jurisdictions. This environmental compatibility is increasingly becoming a decisive factor for multinational corporations aiming to meet their sustainability goals while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific nicotinic acid derivative. These insights are derived directly from the patented methodology to provide clarity on process capabilities and product specifications. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing drug development pipelines.
Q: What is the primary application of 5-nitro-6-methylnicotinic acid ethyl ester?
A: This compound serves as a critical intermediate in the synthesis of PARP1 inhibitor drugs, which are significant in oncology treatment, as well as in the broader class of quinolone antibacterial agents.
Q: How does the copper sulfate step function in this synthesis?
A: The copper sulfate aqueous solution facilitates an oxidative deamination reaction, effectively removing the hydrazino group introduced in the previous step to restore the aromatic pyridine ring structure with high purity.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the process utilizes readily available raw materials and standard solvents like ethanol and chlorobenzene, avoiding expensive transition metal catalysts, which makes it highly amenable to large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitro-6-Methylnicotinic Acid Ethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the timely development of life-saving therapies, which is why we have invested heavily in mastering complex heterocyclic syntheses like the one described in CN114605320A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in pre-clinical development or full-scale commercialization. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 5-nitro-6-methylnicotinic acid ethyl ester meets the exacting standards required for GMP manufacturing. Our commitment to quality assurance means that you can trust our materials to perform consistently in your downstream coupling reactions, minimizing the risk of batch failures.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs with a Customized Cost-Saving Analysis tailored to your volume forecasts. By partnering with us, you gain access to our deep technical expertise and flexible manufacturing capabilities, allowing you to optimize your supply chain for both cost and efficiency. Please reach out today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can accelerate your drug development timeline while reducing overall project costs.
