Advanced Grignard Synthesis of 2-Benzoyl-1,10-Phenanthroline for High-Performance Polyolefin Catalyst Manufacturing
The global demand for linear alpha-olefins and advanced polyolefin materials has necessitated a rigorous re-evaluation of the supply chains supporting their key catalytic precursors. Patent CN116082332A introduces a transformative methodology for the preparation of 2-benzoyl-1,10-phenanthroline, a critical ligand intermediate used extensively in ethylene oligomerization catalysts. This intellectual property represents a significant departure from legacy synthetic routes, addressing long-standing inefficiencies in yield, safety, and environmental compliance that have plagued the fine chemical sector. By leveraging a direct nucleophilic addition strategy, this technology bypasses the cumbersome multi-step transformations previously required, offering a streamlined pathway that aligns with modern green chemistry principles while enhancing economic viability for large-scale manufacturers.
Strategic adoption of this patented process allows industrial partners to secure a more resilient supply of high-purity intermediates essential for next-generation polymerization technologies. The shift from traditional acylation methods to a Grignard-based approach not only simplifies the operational workflow but also mitigates the risks associated with handling highly toxic reagents such as thionyl chloride. For R&D directors and procurement specialists, understanding the mechanistic advantages of this innovation is crucial for optimizing catalyst performance and reducing total cost of ownership. As the polyolefin industry continues to expand, the ability to source 2-benzoyl-1,10-phenanthroline through a more efficient, safer, and scalable route becomes a distinct competitive advantage in the global marketplace.
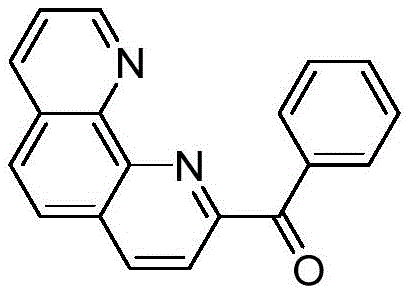
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-benzoyl-1,10-phenanthroline has relied on a convoluted sequence involving the hydrolysis of 2-cyano-1,10-phenanthroline to its corresponding carboxylic acid, followed by conversion to an acid chloride and subsequent Friedel-Crafts acylation. This traditional pathway, as documented in prior art such as Organometallics (2006), suffers from inherent structural inefficiencies that severely impact production economics and safety profiles. The requirement for thionyl chloride introduces significant hazards due to its corrosive nature and the generation of sulfur dioxide byproducts, necessitating expensive scrubbing systems and specialized containment infrastructure. Furthermore, the use of benzene as a solvent in the acylation step poses severe occupational health risks and environmental liabilities, complicating regulatory compliance in stringent jurisdictions.
Beyond safety concerns, the conventional method is characterized by poor atom economy and suboptimal yield performance, typically capping at approximately 48% with purity levels struggling to exceed 90%. The multi-step nature of the process amplifies material losses at each stage, from filtration of the carboxylic acid intermediate to the final purification of the ketone product. These cumulative losses translate directly into higher raw material consumption and increased waste disposal costs, eroding profit margins for manufacturers. The lengthy reaction times, often requiring extended reflux periods of up to 6 hours for multiple steps, further constrain production throughput, making it difficult to respond agilely to fluctuating market demands for polyolefin catalyst intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN116082332A revolutionizes the synthesis by employing a direct addition reaction between 2-cyano-1,10-phenanthroline and a phenyl Grignard reagent. This innovative route collapses the multi-step sequence into a single, highly efficient transformation, effectively eliminating the need for isolating unstable acid chloride intermediates. By utilizing organometallic chemistry, the process achieves a direct carbon-carbon bond formation that is both kinetically favorable and thermodynamically stable under controlled conditions. The elimination of thionyl chloride and benzene from the reagent list drastically reduces the toxicological footprint of the manufacturing process, aligning with increasingly rigorous global environmental standards and corporate sustainability goals.
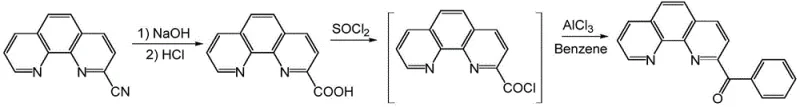
The operational simplicity of this novel approach is matched by its superior performance metrics, with experimental data demonstrating yields reaching up to 73% and purity levels exceeding 98% under optimized conditions. The reaction proceeds rapidly, often completing within 0.5 to 2 hours, which significantly enhances equipment utilization rates and overall plant productivity. Moreover, the flexibility in solvent selection, allowing for the use of tetrahydrofuran, 2-methyltetrahydrofuran, or anhydrous ether, provides process engineers with the ability to tune the reaction environment for optimal solubility and heat transfer. This adaptability is critical for scaling the process from laboratory benchtop to industrial reactor volumes without compromising product quality or safety.
Mechanistic Insights into Grignard Addition to Nitrile Group
The core chemical innovation lies in the nucleophilic attack of the phenyl Grignard reagent on the electrophilic carbon of the nitrile group attached to the phenanthroline ring system. This mechanism bypasses the need for activating the carboxyl group via chlorination, instead relying on the high reactivity of the organomagnesium species to form an imine magnesium salt intermediate directly. Upon acidic workup, this intermediate hydrolyzes cleanly to the desired ketone functionality, preserving the integrity of the sensitive phenanthroline nitrogen heterocycles. The precision of this reaction minimizes side reactions such as over-addition or degradation of the aromatic system, which are common pitfalls in less selective acylation chemistries. Understanding this mechanistic pathway is essential for R&D teams aiming to replicate or further optimize the process for specific catalyst applications.
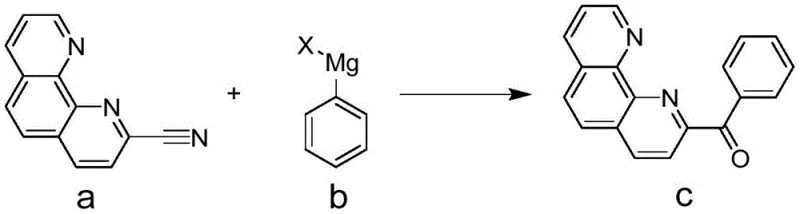
Impurity control is inherently superior in this Grignard-based route due to the absence of harsh acidic or chlorinating agents that often generate chlorinated byproducts or sulfonated impurities. The reaction conditions, typically maintained between -20°C and 5°C during reagent addition, ensure precise thermal management that suppresses exothermic runaway and prevents the formation of polymeric tars. The subsequent quenching with dilute hydrochloric acid is a mild procedure that effectively decomposes the magnesium complexes without attacking the product ketone. This gentle workup protocol facilitates easier downstream purification, often requiring only simple crystallization to achieve pharmaceutical-grade purity, thereby reducing the burden on analytical quality control laboratories and accelerating batch release times.
How to Synthesize 2-Benzoyl-1,10-Phenanthroline Efficiently
Implementing this synthesis requires strict adherence to anhydrous conditions and inert atmosphere protocols to maintain the activity of the Grignard reagent. The process begins with the in-situ or separate preparation of phenylmagnesium bromide or chloride, followed by its controlled addition to the nitrile substrate. Detailed standard operating procedures regarding temperature ramping, stoichiometry ratios, and quenching rates are critical for maximizing yield and ensuring operator safety. While the fundamental chemistry is robust, the specific engineering controls for heat removal and mixing efficiency become paramount as the scale increases. For a comprehensive guide on the exact standardized synthesis steps, please refer to the technical protocol section below.
- Preparation of Phenyl Grignard Reagent by reacting halobenzene with magnesium in anhydrous ether or THF under inert atmosphere.
- Dropwise addition of the Grignard reagent to a solution of 2-cyano-1,10-phenanthroline at controlled low temperatures (-20 to 5°C).
- Quenching the reaction with dilute hydrochloric acid, followed by extraction, concentration, and crystallization to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this patented synthesis route offers substantial strategic benefits that extend beyond mere technical specifications. The reduction in process steps directly correlates to a significant decrease in manufacturing cycle time, allowing for faster turnaround on orders and improved responsiveness to downstream catalyst production schedules. By eliminating the reliance on highly regulated and hazardous chemicals like thionyl chloride, companies can reduce their regulatory compliance burden and lower insurance premiums associated with chemical storage and handling. This simplification of the supply chain also mitigates the risk of production stoppages due to the unavailability of niche reagents, ensuring a more continuous and reliable flow of critical intermediates.
- Cost Reduction in Manufacturing: The streamlined one-step process drastically reduces utility consumption and labor hours associated with multiple isolation and purification stages. Eliminating the need for specialized corrosion-resistant equipment required for thionyl chloride handling lowers capital expenditure requirements for new production lines. Furthermore, the higher yield means less raw material is wasted per kilogram of finished product, directly improving the gross margin profile of the intermediate. The ability to use common ether solvents instead of benzene also reduces solvent procurement costs and waste disposal fees, contributing to a leaner and more cost-effective manufacturing operation overall.
- Enhanced Supply Chain Reliability: Sourcing 2-benzoyl-1,10-phenanthroline from suppliers utilizing this technology ensures a more robust supply chain less susceptible to environmental regulatory crackdowns on toxic emissions. The simplified process flow reduces the number of potential failure points in the manufacturing sequence, leading to higher batch success rates and consistent delivery performance. Suppliers can maintain larger safety stocks of stable starting materials rather than unstable intermediates, enhancing their ability to buffer against market volatility. This reliability is crucial for polyolefin producers who cannot afford interruptions in their catalyst supply chains due to the high cost of plant shutdowns.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, as evidenced by successful kilogram-scale trials that maintained high purity and yield without complex engineering modifications. The reduction in toxic waste generation simplifies effluent treatment requirements, making it easier for manufacturing sites to meet stringent local and international environmental discharge standards. This environmental friendliness enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize sustainable sourcing. The use of less hazardous solvents also improves workplace safety conditions, reducing the likelihood of accidents and associated operational downtime.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader chemical manufacturing strategies. The answers reflect the consensus on performance metrics and safety improvements observed during the development of this method.
Q: What is the primary advantage of the Grignard route over the traditional hydrolysis method?
A: The Grignard route eliminates the need for toxic thionyl chloride and hazardous Friedel-Crafts acylation conditions, significantly reducing reaction steps from multi-stage to a direct addition, thereby improving overall yield from 48% to over 70%.
Q: What purity levels can be achieved with this new synthesis method?
A: Experimental data indicates that optimizing the solvent system, particularly using anhydrous ether or specific ether mixtures, can achieve product purity levels as high as 98%, surpassing the 90% limit of conventional methods.
Q: Is this process scalable for industrial polyolefin catalyst production?
A: Yes, the patent demonstrates successful scale-up examples where kilogram-level batches maintained high yield and purity, confirming the thermal control and quenching protocols are robust enough for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Benzoyl-1,10-Phenanthroline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of advanced polymerization technologies. Our team of expert chemists has thoroughly analyzed the patented Grignard route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the global market. We are committed to delivering 2-benzoyl-1,10-phenanthroline with stringent purity specifications that meet the exacting demands of catalyst manufacturers. Our rigorous QC labs ensure that every batch conforms to the highest standards of quality, providing you with the consistency needed for reproducible catalyst performance.
We invite you to collaborate with us to optimize your supply chain for polyolefin catalyst precursors. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your production goals. Let us help you secure a sustainable and efficient source of this vital chemical building block.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
