Advanced Ru-Catalyzed Synthesis of Ortho-Hydroxy-Nitrogen Silanes for Commercial Scale-Up
Advanced Ru-Catalyzed Synthesis of Ortho-Hydroxy-Nitrogen Silanes for Commercial Scale-Up
The landscape of organosilicon chemistry is witnessing a significant transformation driven by the need for more efficient and economically viable synthetic routes for high-value intermediates. Patent CN111995636A introduces a groundbreaking methodology for the preparation of ortho-hydroxy-nitrogen silane compounds, utilizing a robust ruthenium-catalyzed hydrosilylation protocol. This technology addresses critical bottlenecks in the manufacturing of specialized silicon-containing scaffolds that are increasingly demanded in pharmaceutical development and electronic material science. By leveraging a specific class of ruthenium complexes, the process achieves exceptional conversion rates while maintaining operational simplicity, positioning it as a superior alternative to traditional palladium-mediated pathways. For industry leaders seeking a reliable pharmaceutical intermediate supplier, this innovation represents a pivotal shift towards sustainable and cost-effective production capabilities.
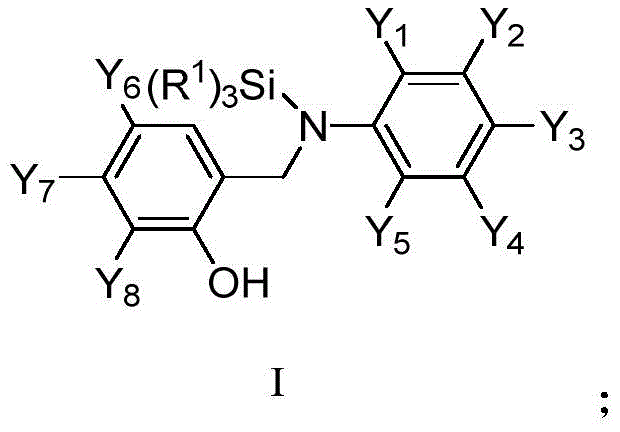
The strategic importance of this patent lies in its ability to streamline the synthesis of complex molecular architectures that serve as key building blocks for bioactive molecules. Organosilicon compounds have garnered immense attention for their potential therapeutic applications, ranging from antihypertensive to antitumor agents, due to the unique metabolic stability imparted by the silicon-carbon bond. The disclosed method facilitates the direct functionalization of salicylidene aniline derivatives, a versatile class of Schiff bases, into stable silylated amines. This transformation is not merely a laboratory curiosity but a scalable industrial process that promises to enhance the supply chain reliability for high-purity organosilicon compounds. As global demand for specialized fine chemicals grows, the adoption of such efficient catalytic systems becomes a cornerstone for maintaining competitive advantage in the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organosilicon frameworks has relied heavily on precious metal catalysts, particularly those based on palladium, which present substantial economic and logistical challenges for large-scale manufacturing. Conventional palladium-catalyzed C-H silylation strategies often necessitate the use of expensive ligands, stringent anhydrous conditions, and multiple synthetic steps to achieve the desired regioselectivity, thereby inflating the overall cost of goods. Furthermore, these traditional methods frequently suffer from limited substrate scope, failing to tolerate sensitive functional groups such as free hydroxyl moieties without extensive protection-deprotection sequences. The generation of stoichiometric halogenated by-products in older methodologies also imposes a heavy burden on waste management and environmental compliance, complicating the regulatory approval process for pharmaceutical intermediates. Consequently, manufacturers face significant hurdles in scaling these processes to meet the rigorous purity and volume requirements of modern drug development pipelines.
The Novel Approach
In stark contrast to legacy technologies, the novel approach detailed in CN111995636A employs a highly efficient ruthenium-catalyzed hydrosilylation of imine bonds, offering a direct and atom-economical route to the target structures. This method utilizes readily available alkylsilanes and salicylidene anilines as starting materials, reacting them in a single pot under relatively mild thermal conditions to yield the desired ortho-hydroxy-nitrogen silanes with impressive efficiency. The use of ruthenium complexes, such as tris(triphenylphosphine)carbonylhydrochlororuthenium, eliminates the need for costly palladium sources and simplifies the reaction setup by tolerating the presence of free phenolic hydroxyl groups without protection. This streamlined process not only reduces the number of unit operations but also minimizes the formation of hazardous waste, aligning perfectly with green chemistry principles. The result is a robust manufacturing platform that significantly lowers the barrier to entry for producing high-value silicon-containing intermediates.
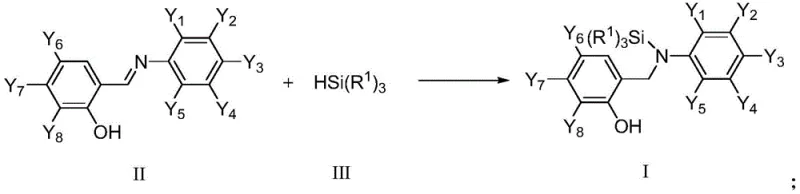
Mechanistic Insights into Ruthenium-Catalyzed Hydrosilylation
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the ruthenium catalyst, which activates the silicon-hydrogen bond of the alkylsilane reagent with remarkable specificity. Unlike radical-based silylation methods that often lead to indiscriminate functionalization and complex impurity profiles, the ruthenium-mediated process proceeds through a well-defined coordination-insertion mechanism that ensures high chemoselectivity for the imine functionality. The catalyst effectively coordinates to the nitrogen lone pair of the Schiff base, polarizing the carbon-nitrogen double bond and facilitating the nucleophilic attack by the hydridic silicon species. This precise control over the reaction trajectory prevents unwanted side reactions such as the reduction of the aromatic rings or the cleavage of the carbon-oxygen bond in the phenolic moiety, which are common pitfalls in less selective catalytic systems. Understanding this mechanism is crucial for R&D directors aiming to optimize reaction parameters for diverse substrate libraries.
Furthermore, the impurity control mechanism inherent in this catalytic system is a major advantage for producing high-purity OLED material or pharmaceutical precursors. The reaction conditions, typically involving heating at 100-120°C in solvents like toluene, are optimized to drive the equilibrium towards the product while suppressing the formation of oligomeric siloxanes or disilanes that often plague silane chemistry. The absence of strong bases or acids in the catalytic cycle preserves the integrity of acid- or base-sensitive functional groups on the aromatic rings, allowing for a broader scope of substituents including halogens and esters. This level of precision ensures that the final product stream requires minimal downstream purification, directly translating to higher overall yields and reduced processing time. For quality assurance teams, this means a more consistent impurity profile and easier validation of the manufacturing process.
How to Synthesize Ortho-Hydroxy-Nitrogen Silane Efficiently
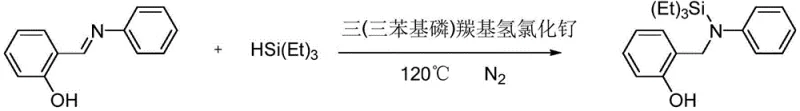
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory equipment and commercially sourced reagents to achieve reproducible results. The process begins with the charging of the salicylidene aniline substrate and the alkylsilane reagent into a reaction vessel under an inert atmosphere, followed by the addition of the ruthenium catalyst in a controlled molar ratio. The mixture is then heated to the specified temperature range, where the hydrosilylation proceeds smoothly over a period of 8 to 36 hours depending on the steric bulk of the substrates involved. Upon completion, the reaction mixture is subjected to simple solvent removal and purification via column chromatography, yielding the target compound as a stable oil or solid. This straightforward protocol minimizes the need for specialized handling techniques, making it accessible for both pilot-scale trials and full commercial production.
- Prepare the reaction mixture by combining salicylidene aniline compounds and alkylsilanes (e.g., triethylsilane) in a suitable solvent like toluene under nitrogen atmosphere.
- Add a ruthenium catalyst such as tris(triphenylphosphine)carbonylhydrochlororuthenium to the mixture with a molar ratio of catalyst to substrate between 0.01 and 0.10.
- Heat the reaction mixture to 100-120°C and stir for 8-36 hours, then remove solvent and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this ruthenium-catalyzed methodology offers profound advantages in terms of cost structure and supply chain resilience. The substitution of expensive palladium catalysts with more abundant and affordable ruthenium complexes directly impacts the raw material cost, allowing for significant margin improvement in the final product pricing. Moreover, the simplicity of the reaction workup, which avoids complex extraction or neutralization steps, reduces the consumption of auxiliary chemicals and solvents, further driving down the operational expenditure. For supply chain heads, the reliance on commodity chemicals like triethylsilane and simple Schiff bases ensures a stable and diversified supply base, mitigating the risk of shortages associated with specialized reagents. This robustness is essential for maintaining continuous production schedules and meeting the just-in-time delivery expectations of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of precious palladium catalysts and the reduction of synthetic steps lead to a substantial decrease in production costs without compromising quality. The high atom economy of the hydrosilylation reaction ensures that the majority of the starting material mass is incorporated into the final product, minimizing waste disposal fees and maximizing resource efficiency. Additionally, the ability to run the reaction at moderate temperatures reduces energy consumption compared to high-pressure or cryogenic alternatives, contributing to a lower carbon footprint and reduced utility costs. These cumulative savings create a compelling economic case for switching to this new technology, especially for high-volume manufacturing campaigns.
- Enhanced Supply Chain Reliability: The use of widely available starting materials and a robust catalytic system ensures that production is not vulnerable to the supply constraints often seen with exotic ligands or metals. The process tolerance to various functional groups allows for the sourcing of diverse substrates from multiple vendors, enhancing negotiation leverage and supply security. Furthermore, the stability of the intermediate and final products simplifies logistics and storage requirements, reducing the risk of degradation during transit. This reliability is critical for long-term supply agreements where consistency and on-time delivery are paramount contractual obligations.
- Scalability and Environmental Compliance: The one-pot nature of the reaction and the absence of hazardous by-products make this process inherently scalable from gram to ton quantities with minimal re-optimization. The reduced generation of halogenated waste aligns with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing facilities. The straightforward purification process also reduces the load on wastewater treatment plants, lowering the environmental compliance burden. This scalability ensures that the technology can grow with market demand, supporting the commercial scale-up of complex polymer additives or electronic chemicals without technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its potential. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of using Ruthenium catalysts over Palladium for this synthesis?
A: Ruthenium catalysts are significantly more cost-effective than Palladium systems and operate efficiently under milder conditions without requiring complex ligand systems, reducing overall production costs and simplifying downstream purification.
Q: Can this synthesis method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes simple raw materials and a one-pot reaction strategy with high yields (up to 88% in examples), making it highly suitable for commercial scale-up and consistent supply chain delivery.
Q: What is the purity profile of the resulting ortho-hydroxy-nitrogen silane compounds?
A: The process generates minimal by-products, and the final products are easily purified via standard column chromatography, ensuring high purity specifications required for sensitive applications like semiconductor photoresists and API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Hydroxy-Nitrogen Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ruthenium-catalyzed synthesis described in CN111995636A and are fully equipped to leverage this technology for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ortho-hydroxy-nitrogen silane meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to quality and technical excellence makes us the ideal partner for companies seeking to secure a stable supply of these critical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this ruthenium-catalyzed process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain. Let us collaborate to bring your next generation of silicon-containing molecules to market faster and more cost-effectively.
