Sustainable Manufacturing of 1H-Benzothiazole Derivatives via Carbon Dioxide Fixation and Borohydride Reduction
The chemical industry is currently undergoing a paradigm shift towards sustainable manufacturing, driven by the urgent need to utilize renewable C1 resources like carbon dioxide. Patent CN114560827A introduces a groundbreaking methodology for the synthesis of 1H-benzothiazole derivatives, a critical scaffold in pharmaceutical and agrochemical applications. This technology leverages stable o-aminodisulfides as starting materials, reacting them with borohydrides under a carbon dioxide atmosphere to construct the benzothiazole ring efficiently. Unlike traditional methods that rely on unstable thiols or toxic reagents, this approach transforms a greenhouse gas into a valuable building block while ensuring high atomic economy. The process operates under moderate conditions, typically between 80-140°C and 0.1-5 MPa pressure, demonstrating remarkable versatility across various substituted substrates. For procurement managers and R&D directors seeking a reliable 1H-benzothiazole supplier, this patent outlines a pathway that combines environmental responsibility with robust chemical performance.

The limitations of conventional synthetic routes for benzothiazoles have long plagued the fine chemical sector, primarily due to raw material instability and harsh reaction conditions. Traditional methods often employ o-aminothiophenol, a compound notorious for its rapid oxidation to disulfides upon exposure to air, necessitating complex stabilization protocols and increasing inventory costs. Alternative routes involving o-haloanilines require additional sulfur sources and often rely on transition metal catalysts, introducing risks of heavy metal contamination that are unacceptable for high-purity pharmaceutical intermediates. Furthermore, condensation reactions with carbonyl compounds can suffer from poor atom economy and the generation of significant waste streams. In stark contrast, the novel approach detailed in this patent utilizes the oxidized disulfide form directly, turning a stability liability into a synthetic asset. By employing borohydrides to simultaneously activate CO2 and cleave the disulfide bond, the method achieves a streamlined, one-pot cyclization that eliminates the need for external sulfur donors or precious metal catalysts.
Mechanistic Insights into Borohydride-Mediated CO2 Activation and Cyclization
The mechanistic elegance of this synthesis lies in the dual role of the borohydride species, which acts as both a reducing agent for the disulfide bond and an activator for the inert carbon dioxide molecule. Under the specified reaction conditions, the borohydride facilitates the reduction of the o-aminodisulfide to a reactive thiolate intermediate in situ, bypassing the isolation of the unstable free thiol. Simultaneously, the boron species coordinates with CO2, lowering the activation energy barrier for its insertion into the organic framework. This cooperative activation allows for the formation of the C-N and C-S bonds required to close the heterocyclic ring at temperatures as low as 110-130°C. The reaction proceeds through a dehydration step that is intrinsic to the mechanism, avoiding the need for外加 dehydrating agents and further enhancing the green chemistry profile of the process. This metal-free catalytic cycle ensures that the final product is free from transition metal residues, a critical quality attribute for API intermediates.
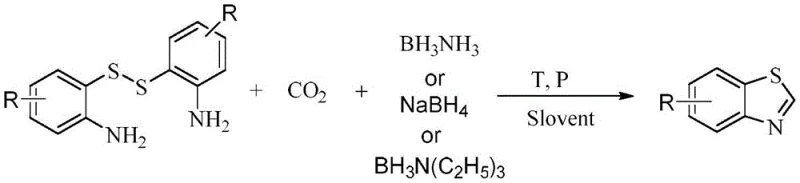
The substrate scope of this reaction is exceptionally broad, accommodating a wide range of electronic and steric environments on the aromatic ring. As demonstrated in the patent data, substituents such as halogens (F, Cl, Br), trifluoromethyl groups, alkoxy chains, and even fused ring systems like naphthalene are well-tolerated. This versatility is crucial for medicinal chemists who require diverse libraries of benzothiazole derivatives for structure-activity relationship (SAR) studies. The ability to synthesize complex structures like 6-trifluoromethylbenzothiazole or chloro-substituted variants with high yields underscores the robustness of the methodology. The reaction conditions are adaptable, allowing for optimization of solvent systems (NMP, DMF, DMSO) and pressure parameters to maximize conversion for specific difficult substrates. This flexibility ensures that the process can be tailored for the commercial scale-up of complex pharmaceutical intermediates without compromising on yield or purity.
How to Synthesize 1H-Benzothiazole Efficiently
The operational protocol for this synthesis is designed for scalability and safety, utilizing standard high-pressure reactor equipment commonly found in fine chemical manufacturing facilities. The process begins with the precise charging of the o-aminodisulfide substrate and the selected borohydride reducing agent into the reactor, followed by the addition of a polar aprotic solvent such as NMP or DMF. Once the system is sealed, it is pressurized with carbon dioxide to the optimal range of 1-4 MPa and heated to the target temperature, typically around 120°C, for a duration of 15 to 36 hours. Post-reaction workup involves standard extraction techniques using ethyl acetate and brine, followed by purification via column chromatography or recrystallization to achieve purity levels exceeding 99%. For detailed standardized operating procedures and specific molar ratios optimized for different substrates, please refer to the technical guide below.
- Load o-aminodisulfide, borohydride reducing agent, and solvent into a high-pressure reactor.
- Pressurize the system with CO2 to 0.1-5 MPa and heat to 80-140°C for 6-36 hours.
- Cool the reaction, extract with ethyl acetate, and purify the crude product via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this technology offers transformative advantages by fundamentally altering the raw material landscape for benzothiazole production. The shift from unstable o-aminothiophenols to stable o-aminodisulfides drastically simplifies logistics, as the starting materials can be stored for extended periods without degradation, ensuring continuous production capability and reducing the risk of batch failures due to原料 quality issues. This stability translates directly into enhanced supply chain reliability, allowing manufacturers to maintain leaner inventories while guaranteeing on-time delivery for downstream clients. Furthermore, the elimination of transition metal catalysts removes the need for expensive and time-consuming metal scavenging steps, which are often bottlenecks in the purification of pharmaceutical intermediates. This simplification of the downstream processing workflow leads to substantial cost savings in terms of both consumables and labor hours.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and abundant carbon dioxide as a C1 building block, replacing costly formylation reagents or cyanide sources used in traditional methods. Additionally, the absence of precious metal catalysts significantly lowers the bill of materials, while the high atom economy minimizes waste disposal costs. The direct dehydration mechanism avoids the purchase of specialized dehydrating agents, further streamlining the cost structure. These factors combine to create a highly competitive manufacturing cost base, enabling significant margin improvement for producers of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing commercially available and shelf-stable o-aminodisulfides, manufacturers can mitigate the risks associated with the supply of sensitive thiol reagents. The robustness of the reaction conditions allows for consistent batch-to-batch reproducibility, which is essential for maintaining long-term contracts with global pharmaceutical companies. The process tolerance to various functional groups means that a single production line can be adapted to manufacture a wide portfolio of derivatives, increasing asset utilization and reducing the lead time for high-purity specialty chemicals. This flexibility ensures that supply disruptions are minimized even when market demand shifts between different benzothiazole variants.
- Scalability and Environmental Compliance: The green chemistry credentials of this method align perfectly with increasingly stringent environmental regulations governing the fine chemical industry. The utilization of CO2 not only reduces the carbon footprint of the manufacturing process but also positions the product favorably in markets that prioritize sustainability. The absence of heavy metals simplifies wastewater treatment and reduces the environmental liability associated with hazardous waste disposal. Moreover, the reaction parameters are conducive to scale-up in standard autoclaves, facilitating the transition from laboratory grams to multi-ton commercial production without the need for exotic equipment or extreme safety measures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CO2 fixation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits and chemical scope for potential partners. Understanding these nuances is critical for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is o-aminodisulfide preferred over o-aminothiophenol in this synthesis?
A: o-Aminothiophenol is highly unstable and prone to oxidation in air, complicating storage and handling. This patent utilizes the stable o-aminodisulfide directly, which is reduced in situ, significantly improving raw material stability and supply chain reliability.
Q: Does this process require expensive transition metal catalysts?
A: No. A key advantage of this method is that it operates without transition metal catalysts. It relies on borohydrides (such as ammonia borane or sodium borohydride) to activate CO2 and cleave the disulfide bond, eliminating the need for costly metal removal steps.
Q: What represents the primary cost driver in this CO2 fixation methodology?
A: The primary cost drivers are the borohydride reducing agents and the energy required for maintaining pressure and temperature. However, the use of cheap, abundant CO2 as a C1 source and the elimination of complex catalyst systems offer substantial overall cost optimization compared to traditional condensation routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-Benzothiazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the one described in CN114560827A to enhance our service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the exacting standards required by the global pharmaceutical industry. Our infrastructure is designed to handle complex heterocyclic syntheses safely and effectively, making us an ideal partner for your benzothiazole sourcing needs.
We invite you to engage with our technical procurement team to discuss how this advanced CO2 utilization technology can be applied to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient synthesis route. We are prepared to provide specific COA data and route feasibility assessments to support your regulatory filings and process validation efforts, ensuring a smooth and successful partnership.
