Advanced Synthesis of Biaryl Chalcones: Unlocking Potent Antitumor Activity via V-Shaped Conformation
The pharmaceutical landscape is constantly evolving, driven by the need for more potent anticancer agents with distinct mechanisms of action. Patent CN102344351B introduces a significant advancement in the field of drug design by disclosing a novel class of aryl-substituted chalcone compounds. Unlike traditional chalcones which often suffer from limited bioactivity, these new derivatives incorporate a biaryl structure that imposes a specific 'V'-shaped spatial conformation. This structural motif is critical because it mimics the geometry of powerful natural microtubule inhibitors such as Colchicine and Combretastatin A4 (CA4). By strategically introducing aromatic substituents onto the phenyl skeleton, the invention achieves a fusion-shaped molecule that demonstrates markedly superior inhibitory activity against various cancer cell lines, including lung, nasopharyngeal, colon, breast, and liver cancers. For R&D directors and procurement specialists seeking high-purity pharmaceutical intermediates, understanding the synthesis and commercial potential of these biaryl chalcones is essential for developing next-generation antitumor therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chalcone synthesis typically relies on the Claisen-Schmidt condensation of simple acetophenones and benzaldehydes. While this method is chemically straightforward, the resulting 1,3-diphenylpropenone scaffolds often exhibit a relatively planar configuration. This planarity limits their ability to effectively bind to the colchicine binding site on tubulin, resulting in modest antitumor efficacy that frequently fails to meet the rigorous standards of modern oncology drug development. Furthermore, conventional modifications often involve simple electron-donating or withdrawing groups which do not fundamentally alter the steric profile of the molecule. Consequently, many existing chalcone derivatives display IC50 values in the micromolar range that are insufficient for clinical progression without extensive structural optimization. The lack of three-dimensional complexity in standard chalcones restricts their interaction with biological targets, necessitating a paradigm shift in how these scaffolds are constructed to enhance potency and selectivity.
The Novel Approach
The methodology outlined in CN102344351B overcomes these geometric limitations by integrating a Suzuki-Miyaura cross-coupling reaction to install a second aromatic ring, thereby creating a biaryl axis. This modification forces the molecule into a twisted, 'V'-shaped conformation that closely resembles the bioactive geometry of established microtubule destabilizing agents. The patent details a flexible synthetic strategy where the biaryl bond can be formed either before or after the chalcone double bond formation. This dual-pathway approach provides significant process flexibility, allowing chemists to choose the route that offers the best solubility profile or the most accessible starting materials. By combining the robustness of palladium-catalyzed coupling with the simplicity of aldol condensation, this novel approach delivers compounds with significantly improved biological profiles, making them highly attractive candidates for further preclinical development and scale-up.
Mechanistic Insights into Pd-Catalyzed Biaryl Formation
The core of this synthetic innovation lies in the efficient construction of the biaryl bond using palladium catalysis. The reaction mechanism follows the standard Suzuki-Miyaura cycle, initiated by the oxidative addition of the aryl halide (typically a bromoacetophenone or bromochalcone intermediate) to the Pd(0) species generated in situ from precursors like PdCl2(dppf). This step is crucial as it activates the carbon-halogen bond for subsequent transmetallation. In the presence of a base such as potassium carbonate (K2CO3), the organoboron reagent (arylboronic acid) forms a reactive boronate complex which then transfers the aryl group to the palladium center. The final reductive elimination step releases the desired biaryl product and regenerates the active Pd(0) catalyst, allowing the cycle to continue. The use of microwave irradiation at 150°C significantly accelerates this cycle, overcoming the kinetic barriers often associated with sterically hindered biaryl formations. This rapid heating method ensures high conversion rates within just 15 minutes, minimizing the exposure of sensitive functional groups to prolonged thermal stress and reducing the formation of homocoupling byproducts.
Controlling impurities in this multi-step sequence is paramount for pharmaceutical grade intermediates. The patent specifies the use of dioxane as the preferred solvent, which provides excellent solubility for both organic substrates and the inorganic base, ensuring a homogeneous reaction environment that promotes consistent coupling efficiency. Following the coupling reaction, the workup involves extraction with ethyl acetate and purification via column chromatography. This rigorous purification protocol is designed to remove residual palladium species, unreacted boronic acids, and any dehalogenated side products. For the Claisen-Schmidt condensation step, the use of potassium hydroxide in ethanol at room temperature allows for mild conditions that preserve the integrity of the newly formed biaryl bond while driving the dehydration to form the alpha,beta-unsaturated ketone. The combination of these orthogonal reaction conditions ensures that the final aryl-substituted chalcones are obtained with high chemical purity, a critical requirement for reliable API intermediate suppliers aiming to support downstream drug substance manufacturing.
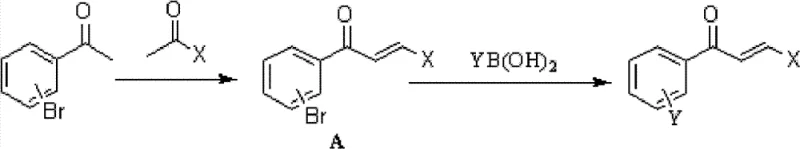
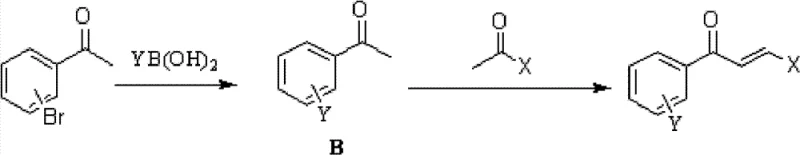
How to Synthesize Aryl-Substituted Chalcones Efficiently
The synthesis of these high-value intermediates can be achieved through two primary convergent routes, offering flexibility based on raw material availability. The process generally involves reacting a bromo-substituted acetophenone with a substituted benzaldehyde to form a bromo-chalcone intermediate, which is then coupled with an arylboronic acid. Alternatively, the bromo-acetophenone can first be coupled with the boronic acid to form a biaryl ketone, which subsequently undergoes condensation with the aldehyde. Both pathways utilize microwave-assisted heating to drive the palladium-catalyzed step to completion rapidly. Detailed standardized synthesis protocols, including specific molar ratios, solvent systems, and purification parameters, are essential for reproducibility. For a comprehensive guide on executing these reactions with optimal yield and purity, please refer to the technical documentation below.
- Perform Claisen-Schmidt condensation between bromoacetophenone and substituted benzaldehyde using KOH in ethanol to form the chalcone intermediate.
- Execute Suzuki-Miyaura coupling using PdCl2(dppf) catalyst, K2CO3 base, and dioxane solvent under microwave irradiation at 150°C for 15 minutes.
- Purify the final biaryl chalcone product via column chromatography to achieve high purity specifications suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the synthetic route described in CN102344351B offers several distinct advantages that translate into tangible operational benefits. The reliance on widely available commodity chemicals such as bromoacetophenones, benzaldehydes, and phenylboronic acids ensures a stable and resilient supply chain. These starting materials are produced on a large industrial scale globally, mitigating the risk of raw material shortages that often plague specialized fine chemical manufacturing. Furthermore, the modular nature of the synthesis allows for the easy substitution of different aromatic groups without altering the core process parameters. This flexibility enables manufacturers to rapidly generate diverse libraries of analogs for structure-activity relationship (SAR) studies without requiring significant retooling or process redevelopment, thereby accelerating the timeline from discovery to clinical candidate selection.
- Cost Reduction in Manufacturing: The implementation of microwave-assisted synthesis represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By reducing the reaction time for the critical cross-coupling step to merely 15 minutes, the process drastically increases throughput and reduces energy consumption per kilogram of product compared to conventional thermal heating methods which may require hours or days. Additionally, the elimination of exotic or highly sensitive reagents in favor of robust palladium catalysts and common bases simplifies the handling requirements and reduces waste disposal costs. The high yields reported in the examples suggest that material loss is minimized, further contributing to overall cost efficiency and making the commercial scale-up of complex biaryl chalcones economically viable.
- Enhanced Supply Chain Reliability: The use of standard solvents like ethanol and dioxane, along with inorganic bases like potassium carbonate, ensures that the supply chain is not dependent on single-source or highly regulated specialty reagents. This diversification of the supply base enhances reliability and reduces lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or moisture, leading to consistent batch-to-batch quality. For procurement managers, this consistency translates to fewer rejected batches and a more predictable inventory flow, which is critical for maintaining continuous production schedules in a GMP environment.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the simplicity of the unit operations involved, primarily mixing, heating, extraction, and chromatography. The short reaction times inherent to the microwave protocol can be adapted to continuous flow chemistry technologies, which offer even greater scalability and safety profiles for exothermic reactions. Moreover, the atom economy of the Suzuki coupling is generally favorable, and the use of ethanol as a solvent for the condensation step aligns with green chemistry principles by avoiding chlorinated solvents. This environmental compatibility simplifies regulatory compliance and waste treatment, supporting sustainable manufacturing practices that are increasingly demanded by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these aryl-substituted chalcones. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of incorporating these intermediates into your drug development pipeline.
Q: What is the structural advantage of aryl-substituted chalcones over traditional chalcones?
A: The introduction of an aromatic group creates a biaryl structure that forces the molecule into a 'V'-shaped spatial conformation. This geometry mimics natural products like Colchicine and CA4, significantly enhancing tubulin inhibition and antitumor activity compared to planar chalcones.
Q: Can the synthesis sequence be reversed to optimize yield?
A: Yes, the patent describes two viable pathways. One can either perform the condensation first followed by Suzuki coupling, or execute the Suzuki coupling on the acetophenone precursor first followed by condensation. This flexibility allows process chemists to select the route with the most cost-effective or available starting materials.
Q: What catalyst system is recommended for the cross-coupling step?
A: The preferred catalyst system involves PdCl2(dppf) with potassium carbonate (K2CO3) in dioxane. Microwave heating at 150°C for 15 minutes is specified to drive the reaction to completion efficiently, minimizing side reactions and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl-Substituted Chalcones Supplier
The technological breakthroughs detailed in CN102344351B highlight the immense potential of biaryl chalcones as potent antitumor agents, yet realizing this potential requires a manufacturing partner with deep technical expertise and scalable infrastructure. NINGBO INNO PHARMCHEM stands ready to support your development goals as a trusted CDMO partner. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from gram-scale research to industrial supply is seamless. Our facilities are equipped with advanced microwave reactors and dedicated purification suites capable of meeting stringent purity specifications required for oncology drug candidates. With our rigorous QC labs and commitment to quality, we guarantee that every batch of aryl-substituted chalcones meets the highest standards of identity, strength, and purity.
We invite you to collaborate with us to optimize this synthetic route for your specific needs. Whether you require custom analogs with varied aromatic substitutions or large-scale production of the lead compounds, our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your project. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your antitumor drug development with reliable, high-quality intermediates delivered on time and within budget.
