Advanced Cobalt-Catalyzed Synthesis of Chiral 3-Substituted-3-Aryl Oxindoles for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and cost-effective pathways to access complex chiral scaffolds, particularly those containing all-carbon quaternary centers which are prevalent in bioactive molecules. Patent CN113880750A introduces a groundbreaking methodology for the synthesis of chiral 3-substituted-3-aryl oxindole compounds, a structural motif found in numerous drugs exhibiting anti-inflammatory, antiviral, and antitumor activities. This innovation leverages an asymmetric Friedel-Crafts reaction between racemic 3-substituted-3-bromooxindoles and N,N-disubstituted anilines, catalyzed by an inexpensive cobalt salt system rather than traditional noble metals. For R&D directors and procurement specialists, this represents a paradigm shift towards sustainable chemistry that does not compromise on stereocontrol or yield. By utilizing earth-abundant cobalt catalysts in conjunction with specialized chiral ligands, the process achieves exceptional enantioselectivity under mild conditions, offering a reliable pharmaceutical intermediate supplier pathway that aligns with modern green chemistry principles while maintaining the rigorous purity standards required for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the challenging 3,3-disubstituted oxindole core has relied heavily on transition metal catalysis involving precious metals, which presents significant economic and logistical hurdles for large-scale production. Prominent prior art includes the palladium-catalyzed intramolecular asymmetric Heck reaction developed by Overman et al., which, while effective, often suffers from relatively low enantioselectivity and requires complex substrate preparation that limits its general applicability. Similarly, methods employing rhodium catalysts for the asymmetric addition of arylboronic acids to isatins, such as those reported by Hayashi et al., incur prohibitive costs due to the price of rhodium and the necessity for rigorous metal scavenging steps to meet regulatory limits. Furthermore, oxidative rearrangement strategies, like those by Movassaghi, typically demand pre-installed chiral centers on the starting materials, severely restricting the diversity of accessible analogs. These conventional routes often involve harsh reaction conditions, expensive reagents, and multi-step sequences that inflate the cost of goods and extend lead times, making them less attractive for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN113880750A utilizes a readily available cobalt salt, such as cobalt(II) acetylacetonate, paired with a chiral ligand to drive the asymmetric transformation with high efficiency. This novel approach eliminates the dependency on scarce noble metals, thereby offering substantial cost reduction in API manufacturing without sacrificing performance. The reaction proceeds through an asymmetric Friedel-Crafts mechanism where the racemic bromooxindole forms a reactive aza-diene intermediate in situ, which is then intercepted by the aniline nucleophile with excellent stereocontrol. As illustrated in the general reaction scheme below, this strategy accommodates a broad range of substituents on both the oxindole and aniline components, demonstrating superior substrate scope compared to previous methodologies.
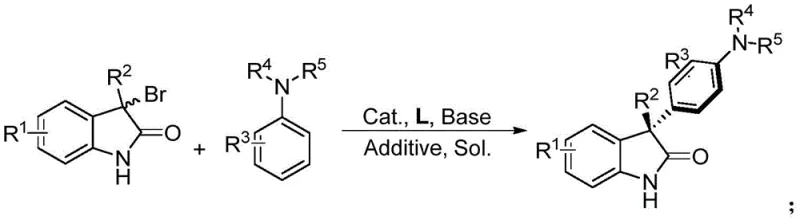
The operational simplicity is another key advantage; reactions typically run at temperatures between -20°C and 30°C in common solvents like dichloromethane, avoiding the need for cryogenic conditions or high-pressure reactors. This accessibility makes the technology highly adaptable for existing manufacturing infrastructure, ensuring a smoother transition from laboratory discovery to pilot plant operations.
Mechanistic Insights into Cobalt-Catalyzed Asymmetric Friedel-Crafts Reaction
The success of this transformation hinges on the precise interplay between the cobalt center and the chiral ligand environment, which dictates the stereochemical outcome of the C-C bond formation. Mechanistically, the base, typically cesium carbonate, facilitates the elimination of hydrogen bromide from the 3-bromooxindole precursor to generate a transient aza-diene intermediate. This highly reactive species is then coordinated by the chiral cobalt complex, creating a rigid chiral pocket that directs the approach of the N,N-disubstituted aniline. The use of additives such as 4Å molecular sieves plays a critical role in sequestering water generated during the process or present in the reagents, preventing hydrolysis of the sensitive intermediate and maintaining catalyst activity. This careful management of the reaction microenvironment ensures that the nucleophilic attack occurs exclusively from one face of the planar intermediate, resulting in the formation of the all-carbon quaternary center with high enantiomeric excess.
Furthermore, the versatility of this catalytic system extends beyond the primary oxindole product, enabling efficient access to complex fused ring systems valuable in medicinal chemistry. For instance, the synthesized chiral oxindoles can serve as pivotal precursors for the construction of furoindoline and pyrroloindoline skeletons through straightforward downstream derivatization. As demonstrated in the synthetic route for compound 20, the initial oxindole product can undergo N-methylation followed by reduction to form the fused furan ring system with retention of optical purity.
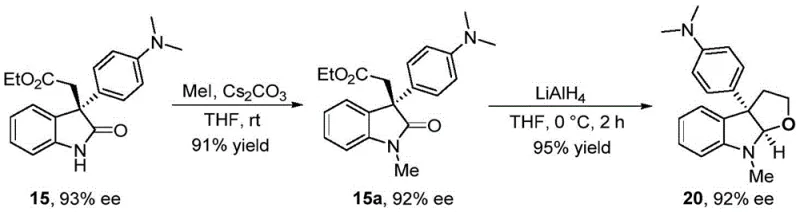
This capability highlights the strategic value of the method for R&D teams aiming to rapidly generate libraries of biologically active natural product analogs. The robustness of the catalyst system also minimizes the formation of side products, simplifying purification workflows and enhancing the overall impurity profile of the final active pharmaceutical ingredient. By controlling the reaction kinetics through ligand design and additive selection, the process effectively suppresses racemization pathways that often plague the synthesis of quaternary carbon centers, ensuring consistent batch-to-batch quality essential for regulatory approval.
How to Synthesize Chiral 3-Substituted-3-Aryl Oxindole Efficiently
Implementing this synthesis in a practical setting requires attention to detail regarding reagent quality and reaction atmosphere to maximize yield and enantioselectivity. The protocol generally involves the pre-formation of the catalyst complex in anhydrous solvent, followed by the sequential addition of substrates and base under inert conditions. Maintaining strict moisture control via molecular sieves is paramount, as water can deactivate the Lewis acidic cobalt species or hydrolyze the reactive intermediates. While the specific stoichiometry may vary depending on the electronic nature of the substrates, a typical molar ratio involves a slight excess of the aniline nucleophile relative to the bromooxindole to drive the reaction to completion. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical documentation below, ensuring reproducibility across different production sites.
- Prepare the catalyst system by stirring cobalt(II) acetylacetonate and chiral ligand L5 in ultra-dry dichloromethane at room temperature for 20 minutes to ensure complete complexation.
- Cool the reaction mixture to -20°C and sequentially add N,N-disubstituted aniline, racemic 3-substituted-3-bromooxindole, cesium carbonate, and activated 4Å molecular sieves.
- Maintain the reaction at -20°C for 12 hours, then filter off solids, concentrate the filtrate, and purify the crude product via column chromatography to isolate the target chiral oxindole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed methodology offers tangible benefits that directly impact the bottom line and operational resilience. The shift from noble metals to base metals like cobalt represents a fundamental change in the cost structure of raw materials, decoupling production expenses from the volatile pricing of palladium and rhodium markets. Moreover, the simplified workflow reduces the number of unit operations required, leading to faster cycle times and increased throughput capacity. The use of common organic solvents and standard laboratory equipment further lowers the barrier to entry for contract manufacturing organizations, facilitating a more competitive bidding landscape for production contracts.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with inexpensive cobalt salts results in a drastic reduction in direct material costs. Additionally, the absence of toxic heavy metals simplifies the downstream purification process, eliminating the need for specialized metal scavenger resins or extensive washing protocols, which translates to lower solvent consumption and waste disposal fees. This streamlined process flow significantly enhances the overall economic viability of producing high-value chiral intermediates, allowing for more aggressive pricing strategies in competitive tender processes.
- Enhanced Supply Chain Reliability: Cobalt salts and the requisite chiral ligands are commercially available from multiple global suppliers, mitigating the risk of single-source bottlenecks that often affect noble metal supply chains. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures or stringent environmental controls associated with high-pressure or cryogenic processes. This reliability ensures consistent delivery schedules for downstream API manufacturers, strengthening the overall resilience of the pharmaceutical supply network against external disruptions.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently safer and easier to scale from pilot plants to multi-ton commercial reactors. The reduced reliance on hazardous reagents and the generation of less toxic waste streams align with increasingly strict environmental regulations, reducing the compliance burden on manufacturing facilities. This eco-friendly profile not only minimizes regulatory risks but also enhances the corporate sustainability metrics of the supply chain, a factor of growing importance to end-user pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on the practical aspects of adopting this new method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using cobalt catalysts over palladium or rhodium for oxindole synthesis?
A: Cobalt salts are significantly cheaper and more environmentally friendly than noble metals like palladium or rhodium. This substitution drastically reduces raw material costs and simplifies the removal of heavy metal residues, which is critical for pharmaceutical compliance.
Q: What is the typical enantiomeric excess (ee) achieved with this method?
A: The patented method consistently achieves high enantioselectivity, with ee values ranging from 90% to 99% across a wide variety of substrates, often requiring only simple recrystallization to reach >99% ee.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the reaction operates under mild conditions (-20°C to 30°C) using common solvents like dichloromethane and stable reagents, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-Substituted-3-Aryl Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral building blocks for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify identity and enantiomeric excess. Our expertise in transition metal catalysis allows us to optimize this cobalt-mediated process for maximum yield and minimal impurity formation, guaranteeing a supply of high-purity chiral oxindoles that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your budget without compromising quality. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us partner with you to accelerate your drug development timeline.
