Scalable Synthesis of Eliglustat Intermediates via Diastereoselective Evans Aldol Chemistry
Scalable Synthesis of Eliglustat Intermediates via Diastereoselective Evans Aldol Chemistry
The pharmaceutical landscape for treating Type I Gaucher disease has been significantly advanced by the development of Eliglustat (Cerdelga), a potent glucosylceramide synthase inhibitor. As demand for this orphan drug grows, the efficiency of its supply chain becomes paramount for global health security. Chinese patent CN108822072B, published in January 2021, introduces a robust and highly efficient synthetic methodology for producing Eliglustat and its key intermediates. This patent represents a critical technological leap, moving away from complex, low-yielding legacy processes toward a streamlined route anchored by diastereoselective Aldol chemistry. For R&D directors and procurement strategists, understanding this shift is essential for securing a reliable API intermediate supplier capable of meeting stringent purity specifications while optimizing production costs.
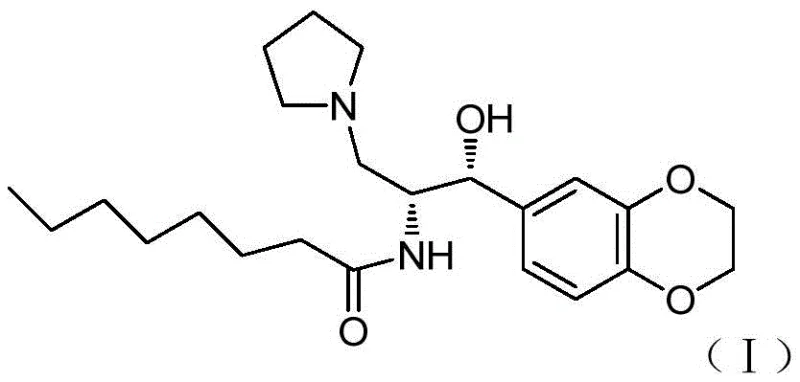
The core innovation lies in the strategic application of chiral auxiliaries to control stereochemistry early in the synthesis. Unlike traditional approaches that may require late-stage resolution or hazardous reagents, this method leverages the predictable stereoselectivity of Evans oxazolidinones or thiazolidinones. By reacting 1,4-benzodioxane-6-carbaldehyde with a chiral imide derivative, the process establishes the critical (1R,2R) configuration with high fidelity. This technical refinement not only enhances the optical purity of the final active pharmaceutical ingredient but also drastically simplifies the downstream processing requirements, making it an ideal candidate for commercial scale-up of complex chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eliglustat has been plagued by operational complexities and supply chain vulnerabilities. Early international patents, such as WO03008399 and WO201559679, describe routes initiating from (L)-glycidol or similar chiral pool materials. While chemically valid, these pathways often suffer from low yields in the initial coupling steps and require multiple protection-deprotection sequences that inflate the overall cost of goods. Furthermore, alternative methods reported in literature, such as those utilizing Henry reactions or Grignard additions to Garner aldehydes, frequently encounter challenges with regioselectivity and the handling of sensitive intermediates. Some reported strategies even rely on microwave-assisted heating at elevated temperatures, a technique that poses significant engineering hurdles when transitioning from gram-scale laboratory experiments to multi-ton industrial reactors.
These conventional limitations translate directly into commercial risks. Low overall yields mean higher raw material consumption and increased waste generation, conflicting with modern green chemistry mandates. Moreover, the reliance on specialized chiral starting materials that are not commodity chemicals creates bottlenecks in the supply chain. If a specific chiral building block faces a shortage, the entire production line for the API can be halted. Therefore, the industry has been actively seeking a route that utilizes broadly available feedstocks and robust, scalable reaction conditions to ensure reducing lead time for high-purity API intermediates.
The Novel Approach
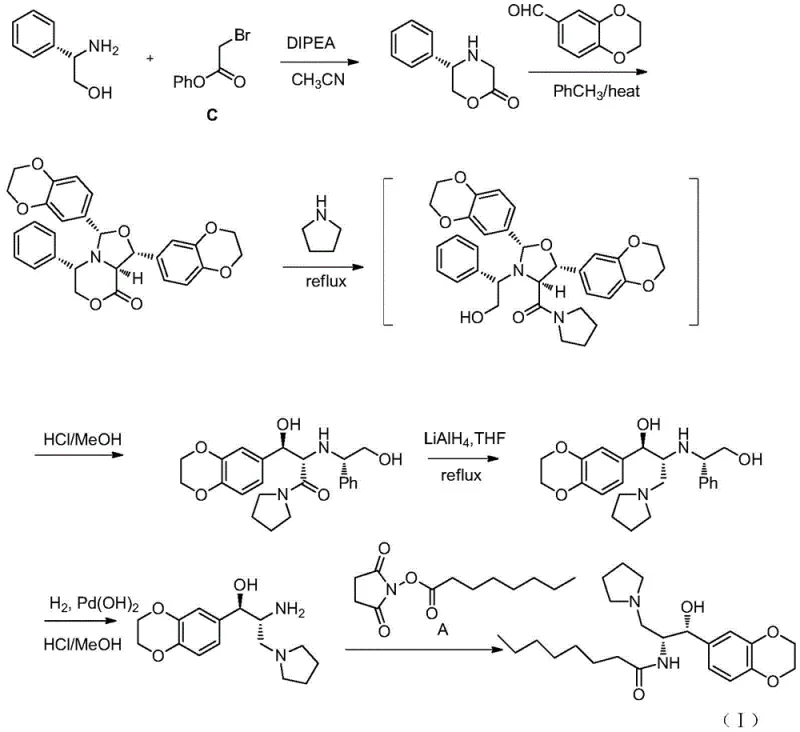
The methodology disclosed in CN108822072B offers a compelling solution by decoupling the chirality source from the main carbon skeleton assembly until the optimal moment. The process begins with the condensation of a readily available aldehyde and a recyclable chiral auxiliary. This diastereoselective Aldol reaction is the cornerstone of the new strategy, effectively setting two contiguous stereocenters in a single transformation. Following the aldol addition, the auxiliary is cleaved, and the resulting intermediate undergoes a sequence of reduction, activation, and substitution reactions that are chemically straightforward and high-yielding. This linear progression minimizes the number of unit operations and avoids the need for chromatographic purifications at every stage, which is a major driver for cost reduction in pharmaceutical manufacturing.
Furthermore, the versatility of this approach allows for the use of either oxazolidinone or thiazolidinone auxiliaries, providing process chemists with flexibility to optimize for cost or reactivity profiles. The final steps involve a Staudinger reduction or catalytic hydrogenation to convert the azide functionality into the requisite primary amine, followed by a simple amidation with octanoyl chloride (or equivalent activated ester). This logical flow ensures that the final product meets rigorous quality standards with minimal impurity carryover, addressing the critical needs of regulatory compliance and patient safety.
Mechanistic Insights into Diastereoselective Evans Aldol Reaction
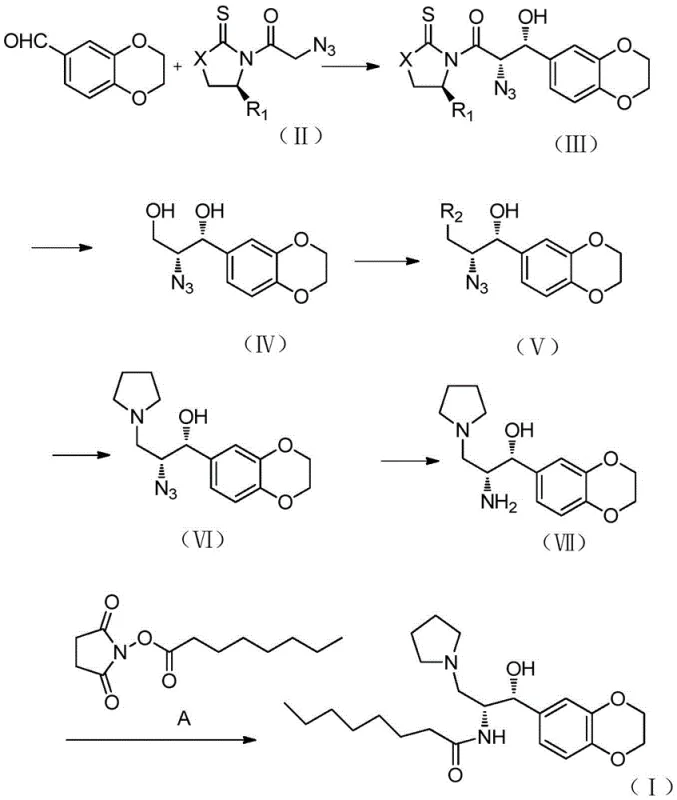
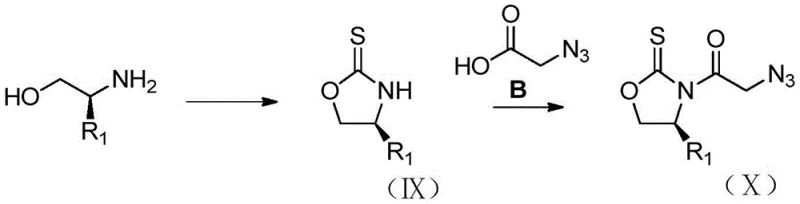
The heart of this synthetic breakthrough is the titanium-mediated Aldol reaction between the chiral N-acylated auxiliary (Formula II) and 1,4-benzodioxane-6-carbaldehyde. In this mechanism, a Lewis acid such as titanium tetrachloride (TiCl4) coordinates with the carbonyl oxygen of the imide and the enolate oxygen, forming a rigid Z-enolate complex. The presence of organic bases like DIPEA and polar aprotic solvents like NMP facilitates the formation of this reactive species at low temperatures, typically around -78°C to -30°C. The bulky substituent on the chiral auxiliary effectively shields one face of the enolate, forcing the incoming aldehyde to approach from the less hindered trajectory. This steric control results in the preferential formation of the 'cis' or syn-aldol adduct (Formula III) with excellent diastereomeric excess.
Following the carbon-carbon bond formation, the process moves to functional group manipulation. The ketone moiety in the aldol adduct is selectively reduced to a secondary alcohol using mild hydride sources like sodium borohydride, preserving the azide group introduced via the auxiliary side chain. Subsequent activation of the primary hydroxyl group—via mesylation or tosylation—creates a superior leaving group. This enables a clean SN2 displacement by pyrrolidine, installing the basic nitrogen center required for the final drug structure. The stereochemical integrity established in the first step is maintained throughout these transformations, ensuring that the final amine (Formula VII) possesses the correct (1R,2R) absolute configuration necessary for biological activity.
Impurity control is inherently built into this mechanism. The use of a covalently bound chiral auxiliary prevents racemization during the enolization step, a common pitfall in direct asymmetric catalysis. Additionally, the crystalline nature of many Evans auxiliary intermediates allows for purification via recrystallization rather than column chromatography, which is vital for removing trace metal contaminants and diastereomeric impurities. The final Staudinger reduction using triphenylphosphine or catalytic hydrogenation over Pd/C provides a clean conversion of the azide to the amine without affecting the sensitive benzodioxane ring or the newly formed secondary alcohol, resulting in a high-purity intermediate ready for the final acylation.
How to Synthesize Eliglustat Intermediate Efficiently
The synthesis of the key chiral amine intermediate (Formula VII) described in this patent offers a practical roadmap for process development teams aiming to implement this technology. The procedure relies on standard laboratory equipment and reagents that are globally sourced, minimizing logistical friction. The initial Aldol condensation requires careful temperature control to maximize diastereoselectivity, but subsequent steps proceed under mild conditions compatible with large-scale reactor vessels. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures, please refer to the standardized synthesis guide below.
- Perform a diastereoselective Aldol reaction between 1,4-benzodioxane-6-carbaldehyde and a chiral oxazolidinone/thiazolidinone auxiliary (Formula II) using TiCl4 and DIPEA.
- Reduce the resulting beta-hydroxy ketone (Formula III) to the diol (Formula IV) using sodium borohydride, followed by selective mesylation or tosylation.
- Execute nucleophilic substitution with pyrrolidine to install the amine side chain, followed by Staudinger reduction or hydrogenation to yield the final amine (Formula VII).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for procurement managers and supply chain directors. The primary advantage stems from the utilization of 1,4-benzodioxane-6-carbaldehyde, a commodity chemical that is readily available from multiple global suppliers. This contrasts sharply with older routes that depend on proprietary or scarce chiral starting materials. By shifting the dependency to common feedstocks, manufacturers can mitigate the risk of supply disruptions and negotiate more favorable pricing terms, leading to substantial cost savings in the overall production budget.
- Cost Reduction in Manufacturing: The streamlined nature of this six-step sequence significantly lowers the operational expenditure associated with Eliglustat production. By consolidating stereocontrol into a single high-yielding step, the process eliminates the need for expensive chiral chromatography or repetitive recrystallizations that characterize less efficient routes. The high atom economy of the Aldol reaction, combined with the recoverability of the chiral auxiliary, ensures that raw material costs are minimized. Furthermore, the avoidance of extreme conditions like high-pressure microwave heating reduces energy consumption and equipment maintenance costs, contributing to a leaner manufacturing profile.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical transformations involved. The reactions described, such as the borohydride reduction and nucleophilic substitution, are well-understood industrial processes with low failure rates. This reliability translates to consistent batch-to-batch quality and predictable production timelines. For procurement teams, this means a more stable inventory of critical intermediates, reducing the need for excessive safety stock and allowing for just-in-time manufacturing strategies that free up working capital.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial mass production, avoiding laboratory curiosities that fail upon scale-up. The use of standard organic solvents like dichloromethane, ethyl acetate, and THF facilitates easy solvent recovery and recycling, aligning with strict environmental regulations. The high purity of the intermediates reduces the burden on wastewater treatment facilities by minimizing the load of organic impurities. This environmental compatibility not only ensures regulatory compliance but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in CN108822072B, offering clarity on why this method represents a superior choice for modern pharmaceutical manufacturing.
Q: What are the primary limitations of previous Eliglustat synthesis routes?
A: Prior art methods, such as those described in WO03008399, often rely on difficult-to-prepare starting materials like (L)-glycidol derivatives or require harsh microwave conditions that hinder large-scale industrial production. Additionally, some routes suffer from low yields in the initial chirality induction steps.
Q: How does the Evans auxiliary method improve stereocontrol?
A: The novel process utilizes a mature diastereoselective Aldol reaction induced by an Evans prosthetic group. This allows for the simultaneous construction of two chiral centers in a single step with high cis-selectivity, significantly simplifying the purification process compared to multi-step resolution methods.
Q: Is this synthesis route suitable for commercial scale-up?
A: Yes, the patent explicitly highlights the use of easily obtainable industrial raw materials and standard reaction conditions (e.g., avoiding extreme microwave heating). The high product yield and purity reported in the examples indicate strong potential for cost reduction in pharmaceutical manufacturing and reliable supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eliglustat Intermediate Supplier
The technological advancements detailed in patent CN108822072B underscore the potential for highly efficient, cost-effective production of Eliglustat intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for FDA and EMA filings.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and economically viable strategy available in the market.
