Revolutionizing Symmetrical Imide Production: A Green, Metal-Free Route for Scalable Pharmaceutical Intermediates
Revolutionizing Symmetrical Imide Production: A Green, Metal-Free Route for Scalable Pharmaceutical Intermediates
In the rapidly evolving landscape of pharmaceutical process development, the demand for efficient, environmentally benign, and cost-effective synthetic routes for key structural motifs is paramount. The recent disclosure in patent CN110041220B introduces a transformative methodology for the synthesis of symmetrical imide compounds, a class of molecules ubiquitous in medicinal chemistry and agrochemical applications. This innovation addresses critical bottlenecks in traditional manufacturing by utilizing a novel base-mediated transformation of activated amides. For R&D directors and procurement strategists alike, this technology represents a significant leap forward, offering a pathway to high-purity intermediates without the burden of transition metal contamination or complex oxidative protocols. By leveraging simple N-acyl glutarimide precursors, this method streamlines the supply chain for complex diaryl imides, ensuring robust availability for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the imide functionality has relied heavily on classical acylation strategies or rearrangement reactions, both of which present substantial operational and economic challenges for large-scale manufacturing. The most prevalent traditional route involves the reaction of amides with highly reactive acylating agents such as acid chlorides or anhydrides. While chemically effective, this approach suffers from poor atom economy and generates significant amounts of corrosive acidic byproducts that require neutralization and disposal. Furthermore, the high reactivity of acid chlorides often limits substrate scope, particularly when sensitive functional groups are present on the aromatic rings, leading to side reactions and reduced yields. Alternatively, the Mumm rearrangement of isoimides offers a different trajectory but necessitates the prior preparation of unstable isoimide intermediates, adding extra synthetic steps and increasing overall production costs. More modern approaches involving metal-catalyzed carbonylation or oxidative decarboxylation introduce further complications, including the requirement for expensive transition metal catalysts like palladium or rhodium, and the use of hazardous oxidants or high-pressure carbon monoxide gas.
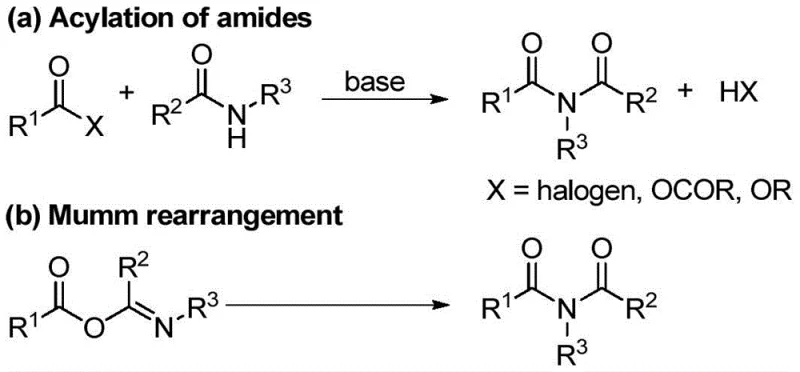
As illustrated in the comparison of conventional pathways, the reliance on metal catalysis and oxidative conditions creates a significant regulatory burden for pharmaceutical manufacturers. The presence of residual heavy metals in the final active pharmaceutical ingredient (API) is strictly regulated, necessitating additional purification steps such as scavenging or recrystallization, which drastically erode profit margins and extend lead times. Moreover, oxidative methods often produce complex mixtures of byproducts due to over-oxidation, complicating the isolation of the target symmetrical imide. These cumulative inefficiencies highlight the urgent need for a more direct, sustainable, and economically viable synthetic strategy that can deliver high-purity materials with minimal environmental impact.
The Novel Approach
The methodology described in patent CN110041220B fundamentally reimagines the disconnection of the imide bond by employing a base-mediated cleavage and recombination strategy using N-acyl glutarimides as privileged substrates. This innovative route bypasses the need for external acylating agents, metal catalysts, or oxidants entirely. Instead, it utilizes the inherent reactivity of the cyclic imide structure in the presence of a strong non-nucleophilic base, such as sodium hexamethyldisilazide (NaHMDS). The reaction proceeds smoothly in aprotic solvents like ethylene glycol dimethyl ether (DME) at moderate temperatures ranging from 80°C to 130°C. This one-step transformation directly converts the readily available N-acyl glutarimide into the desired symmetrical diaryl imide with exceptional efficiency. The simplicity of the reaction setup—requiring only mixing, heating, and a standard aqueous workup—makes it exceptionally attractive for process chemists aiming to reduce unit operations and minimize waste generation.
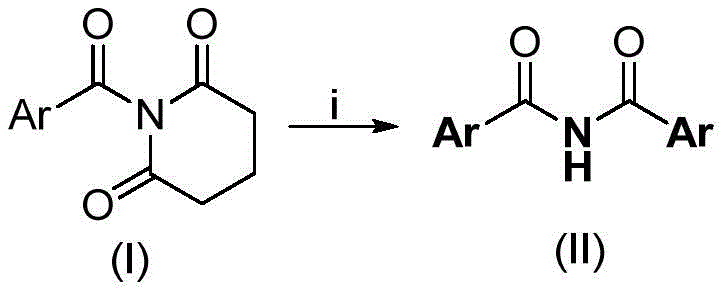
By shifting the paradigm from oxidative or metal-catalyzed coupling to a base-promoted rearrangement, this novel approach unlocks access to a diverse library of symmetrical imides with representative structures. The tolerance for various substituents on the aromatic ring, including halogens, alkyl groups, and electron-withdrawing functionalities like trifluoromethyl or cyano groups, demonstrates the robustness of the method. This versatility ensures that the process can be adapted for the synthesis of a wide array of pharmaceutical intermediates without the need for extensive re-optimization. The elimination of transition metals not only simplifies the purification process but also aligns perfectly with the principles of green chemistry, reducing the environmental footprint of the manufacturing process and lowering the total cost of ownership for the final product.
Mechanistic Insights into Base-Mediated Imide Formation
The mechanistic underpinning of this transformation relies on the unique electronic properties of the N-acyl glutarimide scaffold. Upon treatment with a strong base such as NaHMDS, the acidic proton at the nitrogen atom or the alpha-position is deprotonated, generating a reactive nucleophilic species. This intermediate undergoes a fragmentation or acyl transfer process where the glutarimide ring acts as a leaving group or a transient carrier of the acyl moiety. Unlike traditional nucleophilic acyl substitution which might struggle with steric hindrance, this intramolecular or pseudo-intramolecular pathway facilitates the formation of the new amide bond with high fidelity. The driving force for the reaction is likely the formation of the stable symmetrical imide product coupled with the release of the glutarimide byproduct, which can be easily separated. This mechanism avoids the high-energy transition states associated with direct coupling of two carboxylic acid derivatives, thereby allowing the reaction to proceed under relatively mild thermal conditions.
From an impurity control perspective, the absence of radical intermediates (common in oxidative methods) and metal complexes significantly reduces the complexity of the impurity profile. In metal-catalyzed reactions, trace metals can catalyze unforeseen side reactions such as homocoupling or dehalogenation, leading to difficult-to-remove impurities. In contrast, the base-mediated pathway described here is highly selective. The primary byproducts are derived from the glutarimide leaving group and the base salt, both of which are polar and easily removed during the aqueous workup or silica gel chromatography. This inherent selectivity ensures that the resulting symmetrical imides, such as the benzoyl derivatives shown in the patent examples, are obtained with high chemical purity, often exceeding 90% yield without the need for extensive recrystallization. This level of purity is critical for meeting the stringent specifications required for clinical trial materials and commercial API production.
How to Synthesize Symmetrical Diaryl Imides Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the dissolution of the N-acyl glutarimide precursor in a dry aprotic solvent, followed by the controlled addition of the base under an inert atmosphere to prevent moisture interference. The reaction mixture is then heated to the optimal temperature, typically around 120°C, and maintained for a period of 8 to 12 hours to ensure complete conversion. Upon completion, the reaction is quenched with water, and the product is extracted into an organic phase. The crude material can be purified using standard flash column chromatography or recrystallization techniques to afford the pure symmetrical imide. For detailed operational parameters and safety considerations regarding specific substrates, please refer to the standardized synthesis guide below.
- Dissolve the N-acyl glutarimide precursor (Formula I) in an aprotic solvent such as ethylene glycol dimethyl ether (DME).
- Add a strong base, specifically sodium hexamethyldisilazide (NaHMDS), potassium hexamethyldisilazide, or lithium hexamethyldisilazide, under stirring.
- Heat the reaction mixture to a temperature between 80°C and 130°C for 8 to 12 hours, then quench with water and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The most significant advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for precious metal catalysts such as palladium or expensive oxidizing agents, the bill of materials (BOM) cost is significantly reduced. Furthermore, the removal of heavy metals from the process flow negates the need for specialized metal scavenging resins and the associated validation testing for residual metals, which are time-consuming and costly procedures. This streamlined workflow translates into faster batch turnover times and reduced inventory holding costs, allowing manufacturers to respond more agilely to market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of expensive catalytic systems and the reduction of waste disposal costs. Traditional metal-catalyzed processes often require stoichiometric or near-stoichiometric amounts of ligands and additives, which contribute heavily to the overall production cost. In contrast, the base-mediated method utilizes relatively inexpensive reagents like NaHMDS and common solvents. Additionally, the high yields reported in the patent examples, often reaching up to 95%, mean that less starting material is wasted, maximizing the output per kilogram of input. This efficiency gain is crucial for maintaining competitive pricing in the global market for fine chemicals and API intermediates, where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable starting materials. N-acyl glutarimides are robust solids that can be stored for extended periods without degradation, unlike sensitive acid chlorides or isoimides which may require cold storage or immediate use. This stability reduces the risk of supply disruptions due to material spoilage. Moreover, the reaction conditions do not require specialized high-pressure reactors or cryogenic cooling, meaning that the process can be manufactured in a wider range of facilities, including multi-purpose plants. This flexibility increases the available capacity for production, reducing lead times and ensuring a continuous supply of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this green synthesis route positions manufacturers favorably for future compliance. The absence of toxic heavy metals and hazardous oxidants minimizes the generation of dangerous waste streams, simplifying wastewater treatment and solid waste disposal. The process operates in closed systems with standard venting, reducing the risk of volatile organic compound (VOC) emissions compared to processes involving volatile acid chlorides. This environmental compatibility not only lowers regulatory compliance costs but also enhances the corporate social responsibility (CSR) profile of the manufacturing entity, making it a preferred partner for multinational corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this symmetrical imide synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the primary advantages of this metal-free synthesis over traditional acylation methods?
A: Unlike traditional acylation which often requires hazardous acid chlorides or anhydrides and generates stoichiometric waste, this base-mediated method utilizes stable N-acyl glutarimides. It eliminates the need for transition metal catalysts, thereby removing the costly and complex heavy metal removal steps required for pharmaceutical grade compliance.
Q: What is the typical yield and purity profile for these symmetrical imides?
A: The patent data demonstrates high efficiency, with isolated yields frequently exceeding 90% for various substrates including electron-rich and electron-deficient aryl groups. The absence of metal catalysts and oxidants inherently leads to a cleaner impurity profile, simplifying downstream purification.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process is highly scalable. It employs common organic solvents like DME and standard heating conditions (80-130°C) without requiring high-pressure equipment or specialized inert atmosphere techniques beyond standard nitrogen protection, making it ideal for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Symmetrical Imide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies to enhance the quality and cost-efficiency of our product portfolio. Our technical team has thoroughly evaluated the base-mediated synthesis route described in CN110041220B and confirmed its potential for delivering superior symmetrical imide intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of symmetrical imide meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of novel imide derivatives or scale-up of existing intermediates, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how our commitment to innovation can drive value and efficiency in your supply chain.
