Optimizing Baclofen Hydrochloride Production: A Technical Analysis of the Ultrafine Carbonate Alkylation Route
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for producing critical central nervous system agents. Patent CN102351726A introduces a transformative methodology for the synthesis of Baclofen Hydrochloride, a selective GABAβ receptor agonist widely used for treating muscle spasticity. Unlike traditional routes that rely on complex condensation reactions under harsh conditions, this innovation leverages a solid-liquid two-phase alkylation strategy using ultrafine potassium carbonate. This technical breakthrough addresses long-standing inefficiencies in the supply chain of this vital neurological medication. By shifting the synthetic starting point to 4-chlorophenylacetonitrile, the process circumvents the need for cryogenic reactors and toxic aromatic solvents. For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is essential for evaluating potential technology transfers or licensing opportunities. The following analysis dissects the chemical engineering principles behind this high-yield route, demonstrating its viability for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
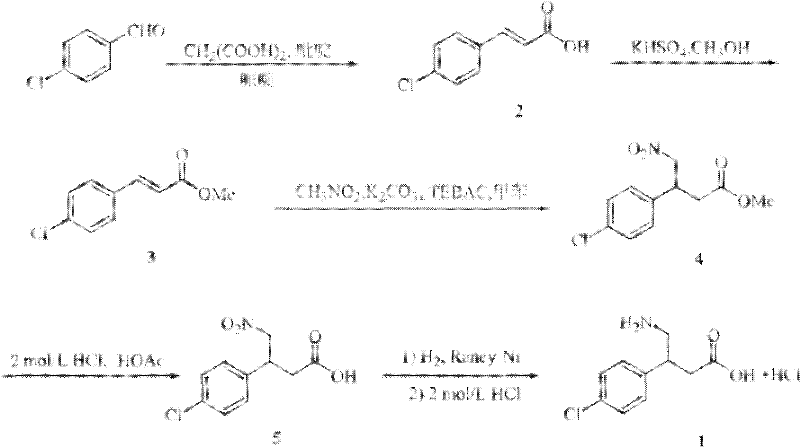
The Limitations of Conventional Methods
Historically, the industrial production of Baclofen Hydrochloride has relied heavily on the p-chlorobenzaldehyde pathway, a method fraught with significant operational and economic drawbacks. As illustrated in the reaction scheme below, this legacy process initiates with a Knoevenagel-Doebner condensation followed by esterification to generate 3-p-chlorophenyl acrylate. The critical bottleneck occurs during the subsequent Michael addition with nitromethane, which necessitates rigorous temperature control below 0°C to manage exothermicity and selectivity. Furthermore, this step typically requires the use of toluene, a solvent with substantial toxicity profiles and stringent environmental disposal regulations. The reliance on expensive phase transfer catalysts further complicates the downstream processing, as removing these catalysts to meet residual metal specifications adds costly purification steps. These factors collectively inflate the cost of goods sold (COGS) and introduce supply chain vulnerabilities related to solvent availability and waste management compliance.
The Novel Approach
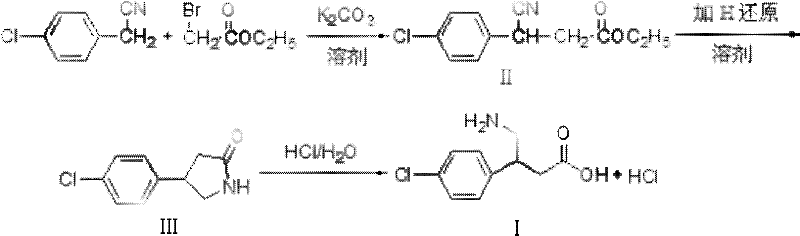
In stark contrast, the novel approach detailed in patent CN102351726A reimagines the carbon skeleton construction through a direct alkylation mechanism. By utilizing 4-chlorophenylacetonitrile and ethyl bromoacetate as primary building blocks, the synthesis bypasses the unstable nitro-alkene intermediates entirely. The core innovation lies in the employment of mechanically activated ultrafine potassium carbonate, which acts as both a base and a pseudo-phase transfer agent due to its nanoscale particle size (100-1000nm). This allows the reaction to proceed efficiently in benign C1-C4 low carbon alcohols at moderate temperatures ranging from 30°C to 70°C. The elimination of cryogenic requirements drastically reduces energy consumption and capital expenditure on specialized cooling equipment. Moreover, the inorganic byproducts formed are insoluble in the alcoholic medium, allowing for their removal via simple filtration rather than energy-intensive aqueous workups. This streamlined workflow not only enhances safety but also significantly shortens the batch cycle time, offering a compelling value proposition for large-scale manufacturers.
Mechanistic Insights into Solid-Liquid Two-Phase Alkylation
The efficacy of this synthesis hinges on the unique physicochemical properties of the ultrafine potassium carbonate catalyst. In conventional organic synthesis, heterogeneous bases often suffer from poor mass transfer limitations, leading to incomplete conversions or prolonged reaction times. However, by reducing the particle size of potassium carbonate to the sub-micron range (specifically 100-500nm in optimized embodiments), the surface area available for reaction increases exponentially. This creates a highly reactive interface where the liquid reactants, 4-chlorophenylacetonitrile and ethyl bromoacetate, can interact directly with the solid base. The mechanism likely involves the deprotonation of the active methylene group in the nitrile at the solid-liquid interface, generating a nucleophilic carbanion that immediately attacks the electrophilic carbon of the bromoacetate. Because the base is insoluble, the equilibrium is driven forward without the complications of homogeneous base catalysis, such as ester hydrolysis or polymerization side reactions. This precise control over reactivity ensures high selectivity for the desired 3-(4-chlorophenyl)-3-cyanopropionic acid ethyl ester intermediate.
Following the alkylation, the subsequent transformation involves a tandem reduction-cyclization sequence to form the pyrrolidone ring. The patent specifies the use of hydrogenation catalysts such as Raney Nickel or Palladium on Carbon (Pd/C) under hydrogen pressure. Mechanistically, the nitrile group is first reduced to a primary amine, which then undergoes an intramolecular nucleophilic attack on the adjacent ester carbonyl. This cyclization releases ethanol and forms the stable five-membered lactam ring, 4-(4-chlorophenyl)-2-pyrrolidone. The final step is a straightforward acid-catalyzed hydrolysis of the lactam ring in aqueous hydrochloric acid. This step cleaves the amide bond to reveal the free carboxylic acid and the protonated amine, simultaneously forming the hydrochloride salt. The elegance of this final step is that it combines hydrolysis and salt formation into a single unit operation, avoiding the need for separate neutralization and acidification stages that typically generate large volumes of saline wastewater.
How to Synthesize Baclofen Hydrochloride Efficiently
Implementing this patented methodology requires careful attention to the preparation of the ultrafine base and the control of hydrogenation parameters. The process begins with the mechanical activation of potassium carbonate in anhydrous ethanol to achieve the critical particle size distribution. Once the alkylation is complete, the reaction mixture is filtered hot to remove potassium salts, and the filtrate is concentrated to isolate the cyano-ester intermediate. The subsequent hydrogenation must be monitored to ensure complete reduction of the nitrile without over-reduction of the aromatic ring. Finally, the hydrolysis step requires reflux conditions to drive the ring opening to completion. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare ultrafine potassium carbonate (100-1000nm) by grinding in ethanol, then react with 4-chlorophenylacetonitrile and ethyl bromoacetate at 30-70°C to form the cyano-ester intermediate.
- Subject the cyano-ester intermediate to catalytic hydrogenation using Raney Nickel or Pd/C in alcohol solvent to induce cyclization, yielding 4-(4-chlorophenyl)-2-pyrrolidone.
- Reflux the pyrrolidone intermediate in aqueous hydrochloric acid for hydrolysis, followed by concentration and recrystallization with isopropanol to obtain the final API salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical novelty. The primary driver for adoption is the substantial reduction in raw material complexity and hazard management costs. By replacing toxic toluene with lower alcohols like ethanol or methanol, facilities can leverage existing solvent recovery infrastructure and reduce regulatory burdens associated with volatile organic compound (VOC) emissions. Furthermore, the starting material, 4-chlorophenylacetonitrile, is a commodity chemical with a stable global supply chain, unlike some specialized intermediates required in older routes. This ensures supply continuity and mitigates the risk of price volatility caused by niche supplier bottlenecks. The simplified purification protocol, which relies on filtration and crystallization rather than chromatography or complex extractions, translates directly into higher throughput and lower labor costs per kilogram of produced API.
- Cost Reduction in Manufacturing: The elimination of expensive phase transfer catalysts and the avoidance of cryogenic cooling systems represent significant capital and operational expenditure savings. The process operates at near-ambient pressures and moderate temperatures, reducing energy loads on plant utilities. Additionally, the high atom economy of the alkylation step minimizes waste generation, lowering disposal costs. The ability to recover and recycle the alcoholic solvents further enhances the economic efficiency of the process, making it highly competitive in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Sourcing 4-chlorophenylacetonitrile and ethyl bromoacetate is straightforward, as these are bulk chemicals produced by multiple vendors worldwide. This diversification of supply sources reduces dependency on single-source suppliers and enhances negotiation leverage. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by minor fluctuations in utility performance, such as cooling water temperature variations. This reliability is crucial for maintaining consistent inventory levels and meeting just-in-time delivery commitments to downstream pharmaceutical formulators.
- Scalability and Environmental Compliance: The solid-liquid nature of the key alkylation step scales linearly from laboratory to pilot to commercial production without the heat transfer issues often seen in highly exothermic homogeneous reactions. The use of non-halogenated solvents in the initial steps aligns with green chemistry principles and simplifies environmental permitting. The final aqueous hydrolysis step generates minimal organic waste, and the inorganic salts produced are easily managed. This environmental profile facilitates faster regulatory approvals and supports corporate sustainability goals, which are increasingly important criteria for partnership selection in the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN102351726A, providing a reliable foundation for feasibility assessments. Understanding these details is critical for engineering teams planning the technology transfer and for quality assurance teams establishing specification limits.
Q: What are the primary advantages of using ultrafine potassium carbonate in this synthesis?
A: Using ultrafine potassium carbonate (100-1000nm) eliminates the need for expensive phase transfer catalysts and strong alkalis like sodium alkoxides. It facilitates a efficient solid-liquid two-phase reaction that avoids side reactions and simplifies the removal of inorganic salts through simple filtration.
Q: How does this new route compare to the traditional p-chlorobenzaldehyde method regarding safety?
A: The traditional method requires toxic toluene solvents and cryogenic conditions (below 0°C) for the Michael addition. The new route operates at mild temperatures (30-70°C) using safer C1-C4 low carbon alcohols, significantly reducing operational hazards and energy consumption for cooling.
Q: What is the overall yield and purity achievable with this patented process?
A: The patent reports a total yield of over 59% for the three-step sequence. The final product, after recrystallization with isopropanol, achieves a purity greater than 98.5%, meeting stringent pharmaceutical standards for API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baclofen Hydrochloride Supplier
The synthesis of Baclofen Hydrochloride via ultrafine potassium carbonate alkylation represents a mature, industrially viable technology that aligns perfectly with modern manufacturing standards. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of solid-liquid phase reactions and catalytic hydrogenations, supported by rigorous QC labs that enforce stringent purity specifications. We understand that in the pharmaceutical sector, batch-to-batch reproducibility is paramount, and our process engineering team is dedicated to optimizing every parameter to deliver high-quality intermediates and APIs.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your validation studies. Let us partner to bring this efficient, safe, and cost-effective production method to your commercial operations.
