Scalable Manufacturing of High-Purity Cyclohexyl Fused-Ring Spiro Bisphosphine Ligands
Scalable Manufacturing of High-Purity Cyclohexyl Fused-Ring Spiro Bisphosphine Ligands
The landscape of asymmetric catalysis is constantly evolving, driven by the demand for more efficient and stereoselective chiral ligands. Patent CN111848673A introduces a significant advancement in this field with the development of a cyclohexyl-fused-ring spirodihydroindane bisphosphine ligand. This compound represents a class of privileged structures known for their rigidity and ability to induce high enantioselectivity in various transformations. Unlike traditional methods that often rely on the tedious resolution of racemic mixtures, this patented technology outlines a streamlined synthetic route starting from optically pure cyclohexyl-fused-ring spirodiphenols. The process leverages modern organometallic techniques, specifically palladium-catalyzed cross-coupling followed by silane reduction, to install phosphine groups with precision. For R&D directors and procurement specialists in the fine chemical sector, this innovation offers a pathway to access high-performance chiral catalysts with improved cost-efficiency and supply chain reliability.
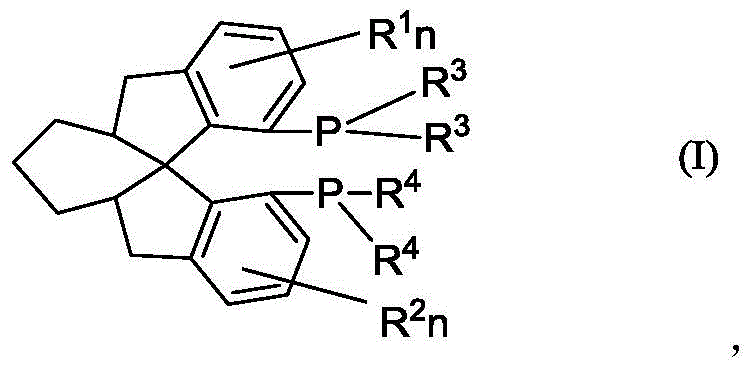
The structural integrity of these ligands is paramount for their performance in catalytic cycles. The spiro-center creates a rigid backbone that restricts conformational freedom, thereby providing a well-defined chiral environment around the metal center. This rigidity is crucial for achieving the high levels of stereocontrol observed in asymmetric hydrogenation reactions, where enantiomeric excess values exceeding 99% have been reported. The versatility of the structure allows for further functionalization on the aromatic rings, enabling fine-tuning of electronic and steric properties to suit specific substrate requirements. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this molecular architecture is essential for delivering products that meet the stringent purity specifications demanded by the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral spiro compounds has been fraught with challenges, primarily stemming from the difficulty in obtaining optically pure starting materials. Conventional approaches often involve the synthesis of racemic spiro skeletons followed by resolution using chiral resolving agents or chromatographic separation. These methods are inherently inefficient, as they theoretically discard at least 50% of the material, leading to significant waste and increased production costs. Furthermore, the resolution processes themselves can be operationally complex, requiring multiple crystallization steps or specialized chiral columns that are difficult to scale. The reliance on stoichiometric amounts of chiral auxiliaries not only drives up the raw material costs but also generates substantial chemical waste, posing environmental compliance issues for large-scale manufacturing facilities. Additionally, the harsh conditions sometimes required for resolution can lead to partial racemization, compromising the optical purity of the final ligand and consequently the performance of the catalyst in downstream applications.
The Novel Approach
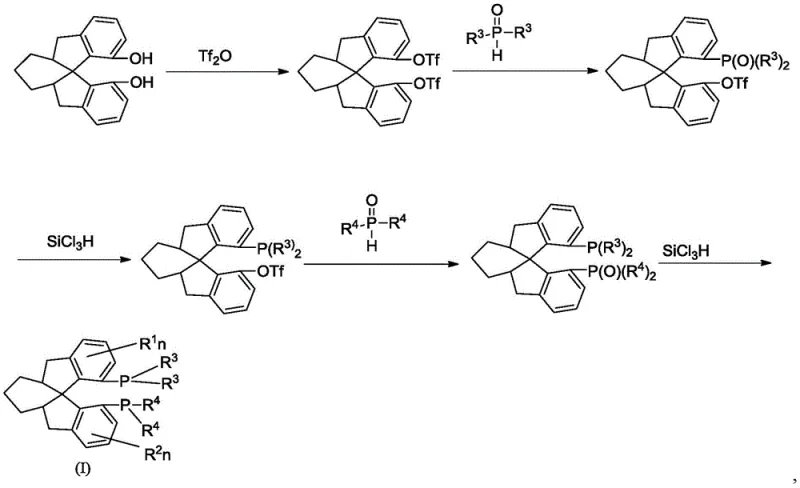
The methodology described in the patent circumvents these bottlenecks by utilizing a convergent synthesis strategy that preserves chirality from the outset. By starting with readily available optically pure cyclohexyl-fused-ring spirodiphenols, the need for resolution is entirely eliminated. The core transformation involves the activation of the phenolic hydroxyl groups into triflates, which serve as excellent leaving groups for subsequent nucleophilic substitution. This is followed by a palladium-catalyzed coupling with diarylphosphine oxides, a reaction known for its robustness and tolerance to various functional groups. The final reduction step using trichlorosilane efficiently converts the phosphine oxides into the desired phosphines without affecting the sensitive spiro scaffold. This sequence not only improves the overall atom economy but also simplifies the purification process, as the intermediates can be isolated with high purity using standard chromatographic techniques. The result is a scalable, environmentally friendlier process that delivers high-purity ligands suitable for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed C-P Bond Formation
The heart of this synthetic route lies in the palladium-catalyzed formation of the carbon-phosphorus bond, a critical step that determines the success of the ligand construction. The mechanism typically initiates with the oxidative addition of the palladium(0) species into the carbon-oxygen bond of the aryl triflate intermediate. This step generates an aryl-palladium(II) complex, which is highly reactive towards nucleophilic attack. The diarylphosphine oxide, activated by a base such as diisopropylethylamine, then coordinates to the palladium center. Subsequent reductive elimination releases the coupled product, regenerating the active palladium(0) catalyst to continue the cycle. The choice of ligand for the palladium catalyst, such as 1,4-bis(diphenylphosphino)butane (dppb), is crucial for stabilizing the active species and facilitating the reductive elimination step. The reaction conditions, typically involving polar aprotic solvents like DMSO and temperatures ranging from 50°C to 150°C, are optimized to ensure complete conversion while minimizing side reactions such as homocoupling or dehalogenation.
Following the coupling, the reduction of the phosphine oxide moiety is achieved using trichlorosilane in the presence of a base. This transformation proceeds through the formation of a silyl-phosphonium intermediate, which undergoes deoxygenation to yield the trivalent phosphine. The use of trichlorosilane is particularly advantageous due to its high reducing power and selectivity, allowing for the reduction of the P=O bond without reducing other sensitive functional groups that might be present on the aromatic rings. The reaction is typically conducted in toluene at reflux temperatures (100-110°C) to drive the equilibrium towards the product. Careful control of the reaction atmosphere is essential, as the resulting phosphines are air-sensitive and prone to oxidation. The rigorous exclusion of oxygen and moisture throughout the synthesis ensures that the final product retains its high chemical and optical purity, which is critical for its performance as a chiral ligand in asymmetric catalysis.
How to Synthesize Cyclohexyl Fused-Ring Spiro Bisphosphine Ligand Efficiently
The synthesis of these advanced ligands requires precise control over reaction parameters to ensure reproducibility and high yield. The process begins with the activation of the chiral diol, followed by iterative coupling and reduction cycles to install the phosphine arms. Each step must be monitored closely to prevent degradation of the chiral skeleton. The detailed standardized synthesis steps are provided in the guide below, offering a clear roadmap for laboratory and pilot-scale production.
- Convert chiral cyclohexyl-fused-ring spirodiphenol to bis-trifluoromethanesulfonate using trifluoromethanesulfonic anhydride and pyridine in dichloromethane.
- Perform palladium-catalyzed coupling with diarylphosphine oxide using Pd(OAc)2 and dppb ligand in DMSO at elevated temperatures.
- Reduce the phosphine oxide intermediate to the final phosphine using trichlorosilane and base in toluene, repeating the coupling-reduction cycle for the second arm.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical performance. The elimination of chiral resolution steps translates directly into significant cost reduction in chiral ligand manufacturing. By avoiding the loss of material inherent in resolution processes, the overall yield of the process is substantially improved, leading to better utilization of raw materials. Furthermore, the reagents used, such as triflic anhydride and palladium catalysts, are commercially available and can be sourced from multiple suppliers, reducing the risk of supply chain disruptions. The robustness of the palladium-catalyzed coupling allows for flexibility in scaling, meaning that production volumes can be adjusted to meet market demand without extensive re-optimization of the process. This scalability is a key factor in ensuring supply continuity for downstream pharmaceutical manufacturers who rely on consistent quality and availability of chiral intermediates.
- Cost Reduction in Manufacturing: The streamlined synthesis avoids the expensive and wasteful resolution steps typical of traditional chiral ligand production. By utilizing a direct coupling strategy from optically pure starting materials, the process maximizes atom economy and minimizes the consumption of chiral resolving agents. This efficiency leads to a lower cost of goods sold (COGS), making high-performance chiral catalysts more accessible for large-scale industrial applications. Additionally, the use of standard organic solvents and common reagents simplifies the procurement process and reduces inventory holding costs.
- Enhanced Supply Chain Reliability: The synthetic route relies on widely available building blocks and catalysts, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for manufacturing in diverse geographical locations, enhancing the resilience of the supply chain against regional disruptions. Moreover, the high purity of the intermediates reduces the need for complex purification steps, shortening the overall production lead time. This reliability is crucial for pharmaceutical companies that require just-in-time delivery of critical intermediates to maintain their own production schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The avoidance of hazardous chiral resolving agents and the reduction of waste generation align with green chemistry principles, facilitating easier regulatory approval and environmental compliance. The ability to recycle solvents and catalysts further enhances the sustainability profile of the manufacturing process, appealing to environmentally conscious stakeholders and reducing the overall environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spiro bisphosphine ligands. The answers are derived from the detailed experimental data and mechanistic understanding provided in the patent literature, ensuring accuracy and relevance for industry professionals.
Q: What is the primary advantage of this spiro bisphosphine ligand synthesis?
A: The primary advantage is the avoidance of costly and wasteful chiral resolution steps. By starting from optically pure spirodiphenols and utilizing efficient Pd-catalyzed coupling, the process maintains high optical purity throughout without the need for separating racemates.
Q: Can this ligand be used for large-scale asymmetric hydrogenation?
A: Yes, the rigid spiro structure provides excellent stability and stereocontrol. The patent demonstrates successful application in the asymmetric hydrogenation of acetophenone with over 99% enantiomeric excess, indicating strong potential for industrial catalytic applications.
Q: What are the key reagents required for the reduction step?
A: The reduction of the phosphine oxide intermediate to the active phosphine ligand utilizes trichlorosilane (SiCl3H) as the reducing agent in the presence of a base such as diisopropylethylamine, typically conducted in toluene at temperatures between 100°C and 110°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexyl Fused-Ring Spiro Bisphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral ligands play in the development of next-generation pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both early-stage research and full-scale manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of ligand meets the highest standards of optical and chemical purity. Our state-of-the-art facilities are equipped to handle air-sensitive chemistry safely and efficiently, preserving the integrity of these valuable catalysts throughout the production process.
We invite you to collaborate with us to leverage this advanced technology for your specific catalytic needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements. Whether you need specific COA data for regulatory filings or route feasibility assessments for process optimization, we are here to support your success. Contact us today to discuss how our expertise in chiral ligand synthesis can accelerate your drug development timeline and enhance the efficiency of your catalytic processes.
