Advanced Synthesis of Dibutyryl Adenosine Cyclophosphate for High-Purity Pharmaceutical Applications
The pharmaceutical landscape continuously demands more efficient synthetic routes for critical bioactive intermediates, particularly those involved in cardiovascular and metabolic therapies. Patent CN112321660A introduces a groundbreaking preparation method for dibutyryl adenosine cyclophosphate compounds and their metal salts, addressing long-standing inefficiencies in the production of this vital cAMP derivative. This technology represents a significant leap forward by streamlining the acylation process, eliminating the need for intermediate quaternary ammonium salt isolation, and achieving exceptional purity levels suitable for parenteral formulations. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition through reduced operational complexity and enhanced yield consistency. By leveraging a direct acylation strategy in aprotic solvents, the process minimizes solvent usage and shortens the overall production cycle, directly impacting the cost structure of manufacturing high-purity pharmaceutical intermediates.
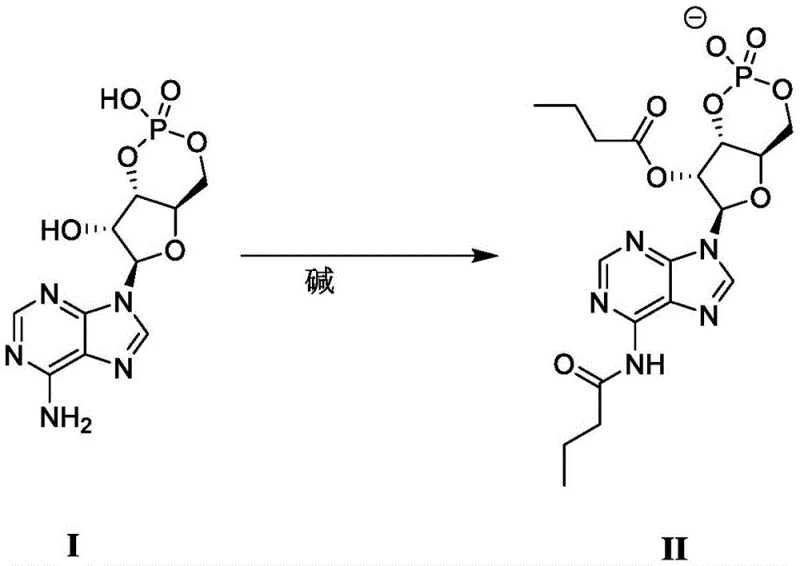
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of calcium dibutyryladenosine cyclophosphate has been plagued by multi-step inefficiencies that inflate both production time and material costs. The traditional chemical synthesis method typically necessitates a preliminary step to prepare a triethylamine salt of the starting material before any acylation can occur. This isolation of an intermediate quaternary ammonium salt not only adds a distinct unit operation to the manufacturing workflow but also introduces additional opportunities for yield loss and impurity generation. Furthermore, the requirement for separate salt formation often dictates higher solvent volumes to ensure adequate crystallization and purification of the intermediate, thereby increasing the environmental footprint and waste disposal costs associated with the process. These cumulative inefficiencies result in a prolonged production period that struggles to meet the agile demands of modern supply chains, creating bottlenecks that can delay the availability of critical API intermediates for downstream drug formulation.
The Novel Approach
In stark contrast to legacy protocols, the novel approach disclosed in the patent utilizes a direct acylation strategy that fundamentally simplifies the reaction architecture. By conducting the reaction in an aprotic solvent in the presence of an organic base, the process allows adenosine cyclophosphate (Formula I) to react directly with n-butyric anhydride without the prerequisite of isolating a specific amine salt. This telescoping of steps effectively merges the activation and acylation phases, leading to a drastic reduction in the total processing time and labor intensity required. The method employs a controlled volume-to-mass ratio of solvent to substrate, optimizing the reaction kinetics while minimizing excess solvent usage. As illustrated in the reaction scheme, the transformation proceeds smoothly to generate the dibutyryl adenosine cyclophosphate anion (Formula II), which can then be directly converted into the desired metal salt. This streamlined workflow not only enhances throughput but also aligns perfectly with green chemistry principles by reducing the overall solvent load and waste generation.
Mechanistic Insights into Organic Base-Mediated Acylation
The core of this technological advancement lies in the precise selection and utilization of organic bases to facilitate the nucleophilic attack of the hydroxyl groups on the ribose moiety. The patent specifies the use of bases such as diisopropylethylamine, triethylamine, or 4-dimethylaminopyridine, which serve dual roles as proton scavengers and potential nucleophilic catalysts. The molar ratio of the organic base to the adenosine cyclophosphate is carefully tuned between 0.5:1 and 4:1, ensuring sufficient basicity to drive the equilibrium towards the acylated product without promoting excessive degradation or side reactions. The reaction temperature is maintained between 50°C and 80°C, a range that provides the necessary activation energy for the acylation while preserving the integrity of the sensitive cyclic phosphate ring. This thermal control is critical, as excessive heat could lead to ring opening or hydrolysis, compromising the biological activity of the final cAMP derivative.
Impurity control is rigorously managed through a sophisticated post-treatment extraction protocol that leverages the differential solubility of the product and by-products. Following the acylation, the reaction mixture is quenched with water, and the aprotic solvent is removed, leaving an aqueous phase containing the target anion. The purification strategy involves sequential washing with specific organic solvent systems, such as a mixture of ethyl acetate and petroleum ether, followed by dichloromethane. This multi-stage extraction effectively removes unreacted n-butyric anhydride, butyric acid by-products, and residual organic bases. The final product is isolated from the organic phase after drying and concentration, yielding a material with a liquid phase content consistently higher than 99%. This high level of purity is essential for pharmaceutical applications, ensuring that the final API meets stringent regulatory standards for safety and efficacy.
How to Synthesize Dibutyryl Adenosine Cyclophosphate Efficiently
Implementing this synthesis route requires careful attention to solvent selection and reaction monitoring to maximize yield and purity. The process begins by dissolving the starting adenosine cyclophosphate in a suitable aprotic solvent like DMF or THF, followed by the addition of n-butyric anhydride and the chosen organic base under an inert atmosphere. The detailed standardized synthesis steps below outline the precise conditions for reaction, workup, and salt formation that have been validated to produce high-quality material suitable for commercial scale-up.
- Dissolve adenosine cyclophosphate (Formula I) in an aprotic solvent such as DMF or THF under nitrogen protection.
- Add n-butyric anhydride and an organic base (e.g., DIPEA or triethylamine) to the solution and heat to 50-80°C for acylation.
- Perform workup by adding water, removing solvent, and extracting with specific organic solvent mixtures to isolate the pure anion.
- React the isolated dibutyryl adenosine cyclophosphate with a metal salt (e.g., calcium chloride) in alcohol/water to form the final metal salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates into tangible strategic advantages regarding cost stability and supply reliability. By eliminating the discrete step of preparing a separate quaternary ammonium salt, the process significantly reduces the consumption of raw materials and solvents, which are major cost drivers in fine chemical manufacturing. The reduction in unit operations also lowers the demand for reactor time and labor, allowing facilities to increase their batch turnover rate without requiring additional capital investment in equipment. This efficiency gain is particularly valuable in a volatile market where raw material prices fluctuate, as it provides a buffer against cost inflation by optimizing the usage of existing resources. Furthermore, the simplified workflow reduces the complexity of the manufacturing process, minimizing the risk of operational errors that can lead to batch failures and supply disruptions.
- Cost Reduction in Manufacturing: The elimination of the intermediate salt preparation step results in substantial cost savings by reducing both material consumption and processing time. Without the need to isolate and dry a quaternary ammonium salt intermediate, the process saves on filtration media, drying energy, and solvent recovery costs. Additionally, the high yield and purity achieved reduce the need for extensive reprocessing or recycling of off-spec material, further enhancing the overall economic efficiency of the production line. These cumulative savings contribute to a more competitive cost structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The streamlined nature of this synthesis route enhances supply chain resilience by shortening the lead time required to produce each batch. With fewer steps and simpler operations, the risk of bottlenecks is minimized, ensuring a more consistent and predictable flow of material to downstream customers. The use of commercially available and stable reagents, such as n-butyric anhydride and common organic bases, ensures that raw material sourcing remains robust and unaffected by niche supply constraints. This reliability is crucial for maintaining continuous production schedules for life-saving cardiovascular medications.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing standard solvents and reaction conditions that are easily transferred from pilot to commercial scale. The reduced solvent usage and simplified waste streams facilitate easier compliance with environmental regulations, lowering the burden of waste treatment and disposal. By generating fewer by-products and utilizing efficient extraction methods, the process aligns with sustainable manufacturing practices, which is increasingly becoming a key criterion for supplier selection by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and benefits of adopting this technology for large-scale production.
Q: How does this novel synthesis method improve upon traditional processes?
A: The traditional method requires a separate step to prepare a triethylamine salt before acylation. This novel approach omits that independent quaternary ammonium salt preparation step, significantly shortening the production period and reducing solvent consumption while maintaining high purity.
Q: What purity levels can be achieved with this acylation protocol?
A: The process is optimized to yield a final product with a liquid phase content exceeding 99%. This high purity is achieved through specific extraction and washing protocols using mixed organic solvents, making it suitable for stringent pharmaceutical applications.
Q: Which solvents and bases are compatible with this reaction?
A: The method utilizes conventional aprotic solvents such as tetrahydrofuran, acetonitrile, pyridine, or N,N-dimethylformamide. Compatible organic bases include diisopropylethylamine, triethylamine, or 4-dimethylaminopyridine, offering flexibility in process optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibutyryl Adenosine Cyclophosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and manufacture of effective pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of dibutyryl adenosine cyclophosphate meets the highest industry standards. Our capability to implement advanced synthesis routes like the one described in CN112321660A demonstrates our dedication to technological innovation and process excellence.
We invite you to collaborate with us to leverage these technical advantages for your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing goals, ensuring a seamless partnership that drives value and efficiency.
