Advanced Aqueous Phase Synthesis of 3-Hydroxy-2-Indolone Derivatives for Commercial Scale Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for high-value intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN108409630B, which discloses a novel preparation method for 3-hydroxy-2-indolone derivatives conducted entirely in an aqueous phase. This technology addresses critical pain points in modern organic synthesis by eliminating the need for hazardous organic solvents and expensive noble metal catalysts. 3-Hydroxy-2-indolone scaffolds are ubiquitous in medicinal chemistry, serving as core structures for numerous bioactive natural products and therapeutic agents. The ability to access these motifs efficiently is paramount for drug discovery pipelines. The patented methodology leverages a radical-mediated C-H hydroxylation strategy using tert-butyl peroxide as a dual-function reagent, offering a streamlined alternative to traditional multi-step sequences. For global procurement teams and R&D directors, this represents a shift towards greener manufacturing paradigms that align with increasingly stringent environmental regulations while maintaining high chemical fidelity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxy-2-indolone compounds has been fraught with operational complexities and economic inefficiencies. Prior art methodologies often rely on transition-metal catalysis, utilizing precious metals such as palladium or ruthenium, which not only inflate raw material costs but also introduce significant challenges regarding metal residue removal in pharmaceutical applications. Furthermore, many established protocols require cryogenic conditions, sometimes as low as -78°C, necessitating specialized equipment and substantial energy consumption for cooling. The use of strong bases like butyl lithium or potassium tert-butoxide adds another layer of safety risk and handling difficulty, particularly when scaling to multi-kilogram batches. Additionally, some existing routes involve ring-closure reactions or additions to isatin that may suffer from limited substrate scope or poor atom economy. These factors collectively contribute to extended lead times and higher production costs, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to deliver high-purity materials consistently.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the invention disclosed in CN108409630B introduces a remarkably simple yet powerful catalytic system. The core innovation lies in the direct C(sp3)-H hydroxylation of 3-substituted-2-indolones using tert-butyl peroxide (t-BuOOH) or its analogues as the hydroxyl source. This transformation proceeds efficiently in water, a non-toxic and inexpensive solvent, facilitated by sodium dodecyl sulfate (SDS) as a surfactant to enhance solubility and reaction kinetics. The reaction operates under mild thermal conditions, typically between 40°C and 100°C, with an optimal temperature of 60°C, and can be conducted under a standard air atmosphere. This eliminates the need for inert gas lines and complex temperature control systems. As illustrated in the reaction scheme below, the process converts readily available 3-substituted-2-indolones directly into the desired 3-hydroxy derivatives with excellent yields.
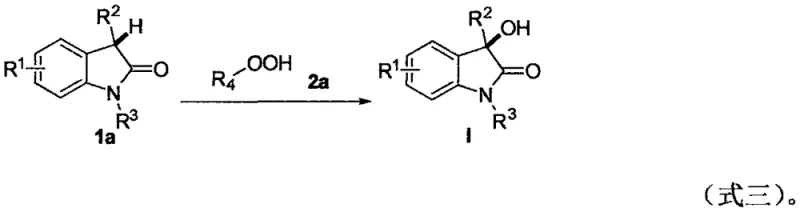
The versatility of this approach is demonstrated by its tolerance to a wide range of functional groups, including halogens, nitro groups, and various alkyl and aryl substituents, making it a universal solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Radical-Mediated C-H Hydroxylation
Understanding the underlying mechanism is crucial for R&D directors evaluating the robustness of this synthetic route. The reaction proceeds via a free-radical pathway initiated by the thermal decomposition of tert-butyl peroxide. Upon heating, the peroxide bond cleaves homolytically to generate tert-butoxyl radicals and hydroxyl radicals. The tert-butoxyl radical abstracts a hydrogen atom from the C-3 position of the 3-substituted-2-indolone substrate, generating a stabilized carbon-centered radical intermediate at the alpha-position to the carbonyl group. This key intermediate is then trapped by a hydroxyl radical, leading to the formation of the C-O bond and the final 3-hydroxy-2-indolone product. Control experiments utilizing radical scavengers such as BHT or hydroquinone significantly suppressed the reaction yield, confirming the radical nature of the transformation. Furthermore, isotopic labeling studies using H2(18)O confirmed that the oxygen atom in the hydroxyl group originates from the peroxide reagent rather than the aqueous solvent, highlighting the specific role of the oxidant.
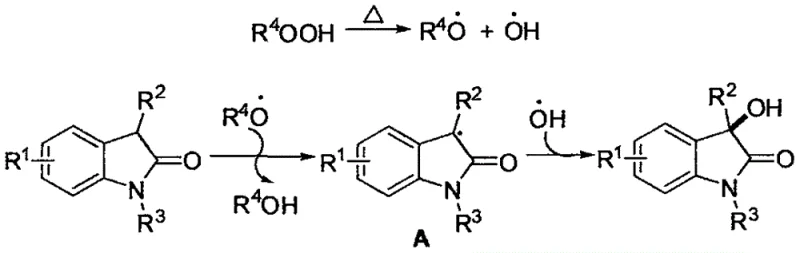
This mechanistic clarity ensures that impurity profiles are predictable and manageable. Since the reaction does not involve transition metals, there is no risk of metal-catalyzed side reactions or difficult-to-remove metal contaminants, which simplifies downstream purification and ensures the resulting high-purity pharmaceutical intermediates meet strict regulatory specifications for residual metals.
How to Synthesize 3-Hydroxy-2-Indolone Derivatives Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemists looking to implement rapid synthesis campaigns. The general procedure involves charging a reaction vessel with the starting indolone, the peroxide oxidant, a catalytic amount of SDS surfactant, and water. The mixture is heated with stirring until conversion is complete, typically monitored by TLC or GC. Workup is straightforward, involving extraction with ethyl acetate, drying, and standard chromatographic purification. This streamlined workflow minimizes unit operations and reduces the overall processing time compared to multi-step alternatives.
- Charge a Schlenk reaction flask with 3-substituted-2-indolone derivative, tert-butyl peroxide (t-BuOOH), sodium dodecyl sulfate (SDS), and water as the solvent.
- Stir the reaction mixture at a temperature between 40°C and 100°C (optimally 60°C) under an air atmosphere for approximately 24 hours until TLC or GC indicates complete consumption of starting material.
- Extract the reaction mixture with ethyl acetate, dry the organic phase over anhydrous sodium sulfate, concentrate under reduced pressure, and purify the residue via column chromatography to obtain the target 3-hydroxy-2-indolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous-phase synthesis technology offers tangible strategic benefits beyond mere chemical elegance. The elimination of noble metal catalysts removes a major cost driver and supply chain vulnerability associated with fluctuating prices of palladium and ruthenium. Moreover, the use of water as the primary reaction medium drastically reduces the volume of volatile organic compounds (VOCs) emitted during production, aligning with global sustainability goals and reducing waste disposal costs. The mild reaction conditions enhance operational safety, lowering insurance premiums and facility maintenance requirements associated with high-pressure or cryogenic processes.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts and ligands with inexpensive tert-butyl peroxide and SDS results in a substantial decrease in raw material expenditure. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification. By avoiding the need for specialized low-temperature equipment, capital expenditure for new production lines is also significantly minimized, allowing for more efficient allocation of resources towards capacity expansion.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including water, SDS, and common peroxides, are commodity chemicals with stable and abundant global supply chains. This reduces the risk of production delays caused by the scarcity of specialized reagents. The robustness of the reaction under air atmosphere further simplifies logistics, as there is no dependency on bulk inert gases like nitrogen or argon, ensuring continuous operation even in facilities with limited utility infrastructure.
- Scalability and Environmental Compliance: The aqueous nature of the reaction facilitates heat transfer and temperature control, which are critical factors for safe scale-up from laboratory to commercial tonnage. The absence of toxic heavy metals simplifies the regulatory approval process for new drug applications, as extensive testing for metal residues is not required. This green chemistry approach not only meets current environmental standards but also future-proofs the manufacturing process against tightening regulations on industrial emissions and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a reliable foundation for process development decisions.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN108409630B operates without transition metal catalysts such as palladium or ruthenium, utilizing tert-butyl peroxide as both the radical initiator and hydroxyl source, which significantly lowers raw material costs.
Q: What is the source of the hydroxyl group in the final product?
A: Isotopic labeling experiments confirm that the hydroxyl oxygen atom is derived from the tert-butyl peroxide reagent rather than the water solvent, ensuring high atom economy and predictable reaction pathways.
Q: Can this process be scaled up for industrial production?
A: Yes, the use of water as a green solvent, mild reaction temperatures around 60°C, and simple workup procedures involving standard extraction and chromatography make this route highly suitable for commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2-Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous-phase synthesis technology described in CN108409630B for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of batch size. Our state-of-the-art facilities are equipped to handle green chemistry processes safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee that every shipment meets the highest international standards. We are committed to leveraging innovative synthetic methods to drive value for our partners in the global pharmaceutical market.
We invite you to collaborate with us to explore how this advanced hydroxylation technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your development timelines and commercial goals with reliable, high-quality 3-hydroxy-2-indolone derivatives.
