Advanced Hypervalent Iodine Chemistry for Scalable Naproxen Derivative Manufacturing
Introduction to Next-Generation Naproxen Functionalization
The pharmaceutical industry continuously seeks efficient pathways to diversify non-steroidal anti-inflammatory drug (NSAID) libraries, and patent CN111943874A introduces a transformative approach using aryl naproxen derivative hypervalent iodine compounds. This technology addresses the critical bottleneck in modifying the naproxen scaffold by replacing harsh transition metal-catalyzed C-H activation with a precise hypervalent iodine-mediated strategy. By leveraging the unique reactivity of lambda-3-iodanes, this method enables nucleophilic substitution at specific sites on the naproxen aryl ring, facilitating the rapid construction of diverse drug-like molecules. For R&D teams focused on lead optimization, this represents a significant leap forward, allowing for the exploration of chemical space that was previously inaccessible due to compatibility issues with traditional catalytic systems. The ability to introduce various nucleophiles cleanly and efficiently opens new avenues for developing next-generation anti-inflammatory agents with improved pharmacokinetic profiles.
From a commercial manufacturing perspective, the implications of this patent are profound, particularly for a reliable pharmaceutical intermediates supplier aiming to streamline production workflows. The synthesis avoids the use of expensive and toxic palladium or copper catalysts, which not only reduces raw material costs but also eliminates the regulatory burden associated with residual heavy metal limits in final drug substances. This aligns perfectly with the industry's push towards greener chemistry and more sustainable manufacturing practices. Furthermore, the operational simplicity of the reaction conditions—typically proceeding at 0°C to room temperature within one hour—suggests that this process can be easily integrated into existing multipurpose reactors without requiring specialized high-pressure or high-temperature equipment. This accessibility makes it an attractive candidate for cost reduction in API manufacturing, ensuring that supply chains remain robust and responsive to market demands for novel NSAID derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the derivatization of naproxen and similar arylpropionic acids has relied heavily on modifying the aliphatic carboxylic acid side chain or employing direct C-H activation on the aromatic ring. While functionalizing the side chain is straightforward, it often fails to impact the core pharmacophore's interaction with biological targets in meaningful ways. More critically, direct C-H activation of the naproxen aryl system typically necessitates the use of transition metal catalysts such as palladium or rhodium complexes. These conventional methods are plagued by inherent drawbacks, including poor regioselectivity, which leads to complex mixtures of isomers that are difficult and expensive to separate. Additionally, the requirement for stoichiometric or excess amounts of oxidants and metal salts generates significant hazardous waste, complicating environmental compliance and increasing disposal costs. The narrow substrate scope of many metal-catalyzed protocols further restricts the diversity of analogs that can be synthesized, limiting the ability of medicinal chemists to fully explore structure-activity relationships (SAR).
The Novel Approach
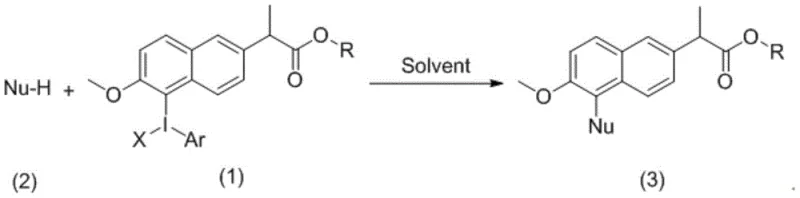
In stark contrast, the novel approach detailed in the patent utilizes hypervalent iodine chemistry to achieve site-specific modification of the naproxen skeleton with exceptional precision. By first converting a naproxen derivative into a hypervalent iodine species, the molecule becomes a potent electrophile capable of undergoing smooth nucleophilic substitution reactions. This strategy bypasses the need for transition metals entirely, thereby eliminating the risk of metal contamination and the associated purification challenges. The reaction conditions are remarkably mild, often proceeding in common halogenated solvents like dichloromethane at temperatures ranging from 0°C to room temperature. This gentleness preserves sensitive functional groups that might otherwise degrade under the harsh conditions required for metal catalysis. Moreover, the versatility of the hypervalent iodine intermediate allows for the introduction of a wide array of nucleophiles (Nu), including carbon, nitrogen, oxygen, phosphorus, and sulfur species, vastly expanding the chemical diversity accessible from a single naproxen precursor. This flexibility is instrumental for rapidly constructing large compound libraries to accelerate the discovery of active leads.
Mechanistic Insights into Hypervalent Iodine Ligand Exchange
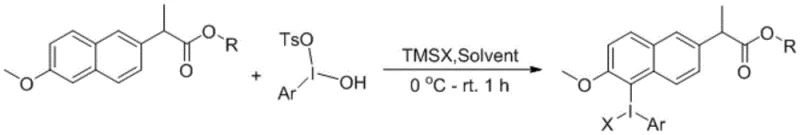
The core of this technology lies in the formation and reactivity of the aryl naproxen hypervalent iodine compound, which serves as a versatile synthon for further derivatization. The mechanism begins with the generation of a Koser reagent derivative (hydroxy(tosyloxy)iodoarene) from a simple aromatic precursor, iodine, and m-CPBA. This reactive iodine(III) species then engages in an anion ligand exchange reaction with the naproxen derivative. In the presence of a trimethylsilyl reagent (TMSX), the naproxen arene effectively displaces the leaving groups on the iodine center, forming the stable yet reactive hypervalent iodine-naproxen adduct. This step is crucial as it installs the hypervalent iodine moiety directly onto the naproxen framework, activating the adjacent aryl carbon for subsequent nucleophilic attack. The electronic properties of the hypervalent iodine group significantly increase the electrophilicity of the ipso-carbon, making it susceptible to displacement by various nucleophiles under mild conditions. This mechanistic pathway ensures high regioselectivity, as the reaction is directed by the pre-installed iodine group, avoiding the random substitution patterns often seen in electrophilic aromatic substitutions.
Understanding the impurity profile is essential for scaling this process to meet high-purity naproxen derivatives specifications. The primary byproducts in this sequence typically stem from the hydrolysis of the hypervalent iodine intermediate or incomplete ligand exchange. However, the patent describes a robust work-up procedure involving precipitation with weakly polar solvents like diethyl ether or hexane, which effectively separates the desired ionic or polar hypervalent species from neutral organic impurities. The use of specific counterions (X), such as triflate, chloride, or tosylate, allows for fine-tuning the solubility and stability of the intermediate, further aiding in purification. By controlling the stoichiometry of the trimethylsilyl reagent and maintaining strict temperature control during the addition (0°C), the formation of side products is minimized. This level of control over the reaction trajectory ensures that the final product possesses the stringent purity specifications required for pharmaceutical applications, reducing the need for resource-intensive chromatographic purification steps and enhancing the overall process mass intensity (PMI).
How to Synthesize Aryl Naproxen Hypervalent Iodine Efficiently
The synthesis of these valuable intermediates follows a logical and scalable two-stage protocol that balances reactivity with operational safety. Initially, the Koser reagent derivative is prepared by oxidizing an aryl iodide with m-chloroperoxybenzoic acid in the presence of p-toluenesulfonic acid, a standard procedure that yields the requisite iodine(III) precursor in high purity. Subsequently, this precursor is reacted with the naproxen derivative in a halogenated solvent system, often enhanced by fluorinated alcohols like hexafluoroisopropanol to stabilize the hypervalent state. The addition of a trimethylsilyl source drives the ligand exchange to completion, yielding the target hypervalent iodine compound as a precipitate upon concentration. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results.
- Prepare the Koser reagent derivative by reacting an aromatic compound with iodine, m-CPBA, and TsOH in DCM at room temperature, followed by precipitation in ether.
- Perform anion ligand exchange by mixing the Koser reagent with the naproxen derivative and trimethylsilyl reagent (TMSX) in a halogenated solvent at 0°C to room temperature.
- Isolate the final hypervalent iodine product by removing the solvent under reduced pressure and precipitating the white solid using a weakly polar solvent like diethyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hypervalent iodine technology offers tangible strategic benefits that extend beyond mere chemical novelty. The elimination of transition metal catalysts fundamentally alters the cost structure of naproxen derivative manufacturing. Traditional palladium-catalyzed routes incur significant expenses not only from the catalyst itself but also from the specialized ligands and the extensive downstream processing required to remove trace metals to ppm levels. By switching to a metal-free hypervalent iodine protocol, manufacturers can drastically simplify their purification trains, potentially removing entire unit operations such as scavenger resin treatments or activated carbon filtrations. This simplification translates directly into substantial cost savings and reduced cycle times, allowing for faster turnaround on custom synthesis orders. Furthermore, the reagents involved, such as m-CPBA and trimethylsilyl salts, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that often plague specialty metal catalysts.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven by the removal of expensive noble metals and the simplification of the purification workflow. Without the need for rigorous heavy metal clearance steps, the consumption of auxiliary materials like scavengers and filtration media is significantly reduced. Additionally, the high yields reported in the patent examples, reaching up to 88% for certain derivatives, ensure that raw material utilization is optimized, minimizing waste and maximizing output per batch. The ability to isolate products via simple precipitation rather than column chromatography further lowers operational expenditures, making the commercial scale-up of complex pharmaceutical intermediates much more financially viable.
- Enhanced Supply Chain Reliability: Relying on transition metals often exposes supply chains to geopolitical volatility and mining constraints. In contrast, the reagents for this hypervalent iodine synthesis are derived from abundant feedstocks like iodine and benzene derivatives, ensuring a stable and continuous supply. The robustness of the reaction conditions, which tolerate ambient temperatures and pressures, means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. This reliability is critical for maintaining consistent delivery schedules to downstream API manufacturers, reducing lead time for high-purity drug candidates and ensuring that clinical trial timelines are met without delay.
- Scalability and Environmental Compliance: From an environmental standpoint, this method aligns well with modern green chemistry principles. The absence of toxic heavy metals reduces the hazardous waste burden, simplifying effluent treatment and lowering disposal costs. The solvents used, primarily dichloromethane and ethers, are well-understood and can be efficiently recovered and recycled in a closed-loop system. The high atom economy of the ligand exchange step, combined with the ease of product isolation, results in a process with a favorable environmental footprint. This compliance with stringent environmental regulations facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hypervalent iodine technology in naproxen derivative production. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is crucial for project managers evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the advantages of using hypervalent iodine for naproxen modification compared to traditional C-H activation?
A: Traditional methods often require transition metal catalysts which suffer from poor selectivity and necessitate costly heavy metal removal steps. The hypervalent iodine approach described in CN111943874A operates under mild conditions (0°C to room temperature) without transition metals, offering superior regioselectivity at the specific aryl site and simplifying downstream purification.
Q: What yields can be expected from this synthesis method?
A: According to the patent data, the process demonstrates robust efficiency. For instance, the synthesis of phenyl naproxen methyl ester trifluoromethanesulfonic acid hypervalent iodine achieved an isolated yield of 88%, while other derivatives like the 4-methylphenyl variant reached 82%, indicating high reproducibility suitable for production.
Q: Is this method scalable for commercial API intermediate production?
A: Yes, the methodology is highly amenable to scale-up. The reactions utilize common solvents like dichloromethane and proceed rapidly (1 hour reaction time) at near-ambient temperatures. The work-up involves simple precipitation and filtration, avoiding complex chromatographic separations, which significantly enhances operational feasibility for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Naproxen Hypervalent Iodine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of hypervalent iodine chemistry in accelerating the development of next-generation NSAIDs. Our team of expert chemists has extensively validated the protocols described in CN111943874A, ensuring that we can deliver these complex intermediates with the highest standards of quality and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to handle sensitive iodine chemistry safely and efficiently. Our rigorous QC labs employ advanced analytical techniques to verify stringent purity specifications, guaranteeing that every batch meets the exacting requirements of global pharmaceutical clients. We are committed to being a partner that not only supplies materials but also provides technical solutions to optimize your drug discovery pipeline.
We invite you to collaborate with us to explore the full potential of aryl naproxen hypervalent iodine compounds for your specific research needs. Whether you require custom synthesis of novel derivatives or scale-up of established routes, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project volume. We encourage you to reach out for specific COA data and route feasibility assessments to see how our expertise can drive efficiency and innovation in your naproxen derivative manufacturing programs.
