Advanced Synthetic Route for Tivozanib Intermediates: Scalable Manufacturing Solutions
Advanced Synthetic Route for Tivozanib Intermediates: Scalable Manufacturing Solutions
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with targeted therapies like Tivozanib (AV-951) representing a critical frontier in inhibiting vascular endothelial growth factor receptors. The patent CN102532116A discloses a robust and highly efficient synthetic methodology for producing this potent anti-tumor agent, addressing the growing global demand for reliable tivozanib intermediate supplier capabilities. This comprehensive technical report analyzes the seven-step linear synthesis outlined in the patent, which transforms simple starting materials into complex quinoline derivatives through a series of optimized reactions including Friedel-Crafts acylation, nitration, and catalytic reduction. By leveraging this specific intellectual property, manufacturers can achieve significant improvements in process stability and cost-efficiency, ensuring a consistent supply of high-purity active pharmaceutical ingredients for clinical and commercial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for complex quinoline-based kinase inhibitors often suffer from prohibitive costs associated with expensive transition metal catalysts and harsh reaction conditions that compromise safety and scalability. Many legacy routes rely on precious metal-catalyzed cross-couplings or multi-step protections that introduce unnecessary complexity and reduce the overall atom economy of the process. Furthermore, conventional methods frequently struggle with regioselectivity issues during the functionalization of the aromatic ring, leading to difficult-to-separate impurities that require extensive downstream purification. These inefficiencies not only inflate the cost of goods sold but also create bottlenecks in the supply chain, making it challenging to meet the rigorous quality standards required for oncology drugs without substantial time delays.
The Novel Approach
In stark contrast, the methodology presented in CN102532116A offers a streamlined approach that prioritizes the use of inexpensive, commodity-grade chemicals such as veratrole and acetyl chloride to construct the core molecular framework. This novel route eliminates the need for exotic reagents by utilizing classic organic transformations like iron-mediated reduction and phosphorus trichloride chlorination, which are well-understood and easily managed in standard chemical reactors. The process is designed to operate under mild conditions, typically ranging from room temperature to moderate reflux, which significantly reduces energy consumption and minimizes the risk of thermal runaway incidents. By focusing on high-yielding individual steps, the overall throughput of the manufacturing line is maximized, providing a distinct competitive advantage in cost reduction in API manufacturing for high-value cancer therapeutics.
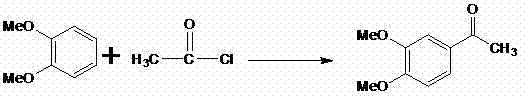
Mechanistic Insights into Friedel-Crafts Acylation and Quinoline Cyclization
The foundation of this synthetic strategy lies in the initial Friedel-Crafts acylation, where veratrole reacts with acetyl chloride in the presence of anhydrous aluminum trichloride to form 3,4-dimethoxyacetophenone with exceptional regioselectivity. As illustrated in the reaction scheme, the electron-donating methoxy groups direct the incoming acyl group to the preferred position, minimizing the formation of ortho-isomers and simplifying the purification workflow. Following this, the nitration step introduces a nitro group para to the acetyl moiety, setting the stage for the subsequent reduction. The use of iron powder as a reducing agent is particularly noteworthy from a mechanistic standpoint, as it provides a cost-effective alternative to catalytic hydrogenation while effectively converting the nitro group to an amine without affecting other sensitive functional groups on the molecule.
The construction of the quinoline ring system represents the most critical transformation in the sequence, achieved through an addition-elimination reaction between the amino-acetophenone derivative and ethyl formate under basic conditions. This cyclization step closes the heterocyclic ring to form 6,7-dimethoxy-4-quinolinone, a pivotal intermediate that defines the pharmacological activity of the final drug.  Subsequent chlorination with phosphorus trichloride activates the 4-position of the quinoline ring, enabling the final nucleophilic aromatic substitution with the phenolic component. This precise control over the electronic properties of the quinoline scaffold ensures that the final coupling reaction proceeds efficiently, yielding the target urea derivative with high structural fidelity and minimal side-product formation.
Subsequent chlorination with phosphorus trichloride activates the 4-position of the quinoline ring, enabling the final nucleophilic aromatic substitution with the phenolic component. This precise control over the electronic properties of the quinoline scaffold ensures that the final coupling reaction proceeds efficiently, yielding the target urea derivative with high structural fidelity and minimal side-product formation.
How to Synthesize Tivozanib Efficiently
The synthesis of Tivozanib intermediates requires strict adherence to the optimized reaction parameters detailed in the patent to ensure maximum yield and purity at every stage of the process. Operators must carefully control the stoichiometry of reagents, particularly during the acylation and nitration steps, to prevent over-reaction or the generation of poly-substituted byproducts. The detailed standardized synthesis steps见下方的指南 outline the specific temperatures, solvent choices, and workup procedures necessary to replicate the high success rates reported in the experimental examples, serving as a critical reference for process engineers aiming to implement this technology.
- Initiate the synthesis via Friedel-Crafts acylation of veratrole with acetyl chloride using aluminum trichloride catalyst.
- Perform nitration followed by catalytic reduction using iron powder to generate the amino-acetophenone precursor.
- Execute cyclization with ethyl formate and subsequent chlorination to form the core quinoline scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial economic benefits by relying on a supply chain of raw materials that are globally available and priced competitively in the bulk chemical market. The elimination of precious metal catalysts removes a significant cost driver and mitigates the risk of supply disruptions associated with rare earth elements, thereby enhancing the overall resilience of the manufacturing operation. Additionally, the use of standard solvents and reagents simplifies the sourcing process, allowing procurement managers to negotiate better terms with multiple vendors and avoid single-source dependencies that could jeopardize production schedules.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing iron powder for reduction instead of expensive palladium or platinum catalysts, which drastically lowers the raw material expenditure per kilogram of product. Furthermore, the high yields observed across the synthetic sequence mean that less starting material is wasted, directly improving the mass balance and reducing the cost of waste disposal. The mild reaction conditions also translate to lower energy costs, as the process does not require extreme heating or cryogenic cooling, contributing to a leaner and more economical production model.
- Enhanced Supply Chain Reliability: By selecting starting materials like veratrole and acetyl chloride, which are produced on a massive industrial scale for various applications, the supply chain becomes inherently more stable and less prone to volatility. The robustness of the chemical steps ensures that production can continue consistently without frequent interruptions for catalyst regeneration or complex equipment maintenance. This reliability is crucial for maintaining continuous inventory levels and meeting the just-in-time delivery requirements of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing unit operations such as filtration and crystallization that are easily transferred from pilot plants to multi-ton commercial reactors. The avoidance of hazardous heavy metals simplifies the environmental compliance landscape, reducing the burden of wastewater treatment and facilitating easier regulatory approval for new manufacturing sites. This alignment with green chemistry principles not only lowers operational risks but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity on its feasibility and advantages for potential partners. These answers are derived directly from the experimental data and technical specifications contained within the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their supply chains.
Q: What are the key advantages of this Tivozanib synthesis route?
A: The process utilizes cheap and readily available raw materials like veratrole and employs mild reaction conditions, resulting in a stable process with high overall yields suitable for industrial scale-up.
Q: How is the purity of the quinoline intermediate controlled?
A: Purity is maintained through precise stoichiometric control during the Friedel-Crafts and nitration steps, alongside rigorous purification methods such as recrystallization and suction filtration described in the patent examples.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its low cost, operational simplicity, and the use of standard chemical reagents without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tivozanib Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of analyzing complex impurity profiles to guarantee that every batch of Tivozanib intermediate meets the highest international standards. We understand the critical nature of oncology supply chains and are committed to delivering consistent quality and reliability to support your clinical and commercial needs.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By engaging with us, you can access specific COA data and route feasibility assessments that will demonstrate the tangible value of partnering with a leader in fine chemical manufacturing. Let us collaborate to optimize your supply chain and accelerate the delivery of life-saving therapies to patients worldwide.
