Advanced Synthetic Route for 4,6-Dichloro-2-Methylthio-5-Nitropyrimidine: Technical and Commercial Analysis
The chemical landscape for pyrimidine derivatives has long been challenged by the lack of efficient, scalable total synthesis methods for specific halogenated intermediates. Patent CN102952084A introduces a groundbreaking four-step synthetic methodology for producing 4,6-dichloro-2-methylthio-5-nitropyrimidine, a critical building block in the development of advanced pharmaceutical and agrochemical agents. This technical disclosure addresses a significant gap in the prior art, where no ready-made total synthesis methods were previously available for this specific compound class. By leveraging diethyl malonate as a foundational starting material, the patented process establishes a logical and operationally simple pathway that transforms basic commodity chemicals into high-value heterocyclic structures. For R&D Directors and Procurement Managers evaluating new supply sources, this patent represents a validated route that balances chemical complexity with operational feasibility, ensuring that the target molecule can be produced with high purity and structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation detailed in CN102952084A, the synthesis of 4,6-dichloro-2-methylthio-5-nitropyrimidine was hindered by the absence of a defined, reliable production protocol in scientific literature. Conventional approaches to similar pyrimidine scaffolds often suffered from unpredictable regioselectivity, low overall yields, or the requirement for exotic, cost-prohibitive reagents that complicated supply chain logistics. Many existing methods relied on multi-step sequences that generated excessive waste or required harsh conditions incompatible with large-scale manufacturing environments. The lack of a standardized method meant that batch-to-batch consistency was difficult to achieve, posing significant risks for downstream drug development projects where impurity profiles must be tightly controlled. Furthermore, without a clear synthetic roadmap, procurement teams faced uncertainty regarding raw material availability and long-term cost stability, as custom synthesis often carried premium pricing due to process inefficiencies and optimization requirements.
The Novel Approach
The novel approach presented in this patent overcomes these historical barriers by establishing a streamlined four-step sequence that prioritizes reagent accessibility and reaction controllability. Starting from diethyl malonate, the process systematically builds the pyrimidine core through nitration, cyclization, methylation, and chlorination, each step optimized for maximum conversion and minimal byproduct formation. This methodology eliminates the need for obscure precursors, relying instead on widely available industrial chemicals such as thiourea, dimethyl sulfate, and phosphorus oxychloride. The strategic selection of reaction conditions, such as the use of sodium alkoxide for cyclization and N,N-dimethylaniline as a chlorination catalyst, ensures that the reaction kinetics favor the desired product. For supply chain heads, this translates to a robust manufacturing process that reduces dependency on single-source specialty vendors and allows for flexible sourcing of raw materials, thereby enhancing overall supply security and reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into the Four-Step Pyrimidine Construction
The mechanistic elegance of this synthesis lies in its precise control over functional group transformations, beginning with the nitration of diethyl malonate. In this initial phase, the substrate is treated with concentrated or fuming nitric acid at a carefully maintained temperature range of 0°C to 30°C. This thermal regulation is critical to prevent over-nitration or decomposition, ensuring the selective formation of diethyl 2-nitromalonate. Following isolation, the nitro-diester undergoes cyclization with thiourea in the presence of sodium alkoxide within an alcoholic solvent system at 40°C to 80°C. This step constructs the heterocyclic ring, generating 4,6-dihydroxy-2-mercapto-5-nitropyrimidine through a condensation mechanism that effectively locks the nitrogen and sulfur atoms into the pyrimidine framework. The subsequent methylation step utilizes dimethyl sulfate under alkaline conditions to selectively protect the thiol group, forming the methylthio moiety which is essential for the biological activity of the final derivative.
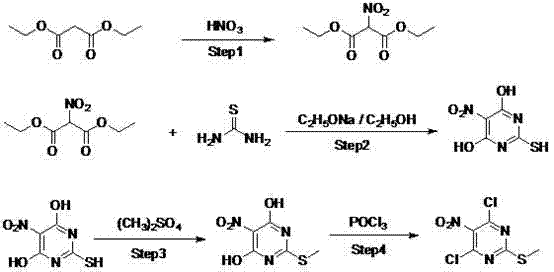
The final transformation involves the chlorination of the dihydroxy intermediate using phosphorus oxychloride (POCl3), which serves dual roles as both the chlorinating agent and the reaction solvent. The addition of a catalytic amount of N,N-dimethylaniline significantly accelerates the substitution of the hydroxyl groups at the 4 and 6 positions with chlorine atoms, yielding the target 4,6-dichloro-2-methylthio-5-nitropyrimidine. This step typically proceeds at reflux temperatures between 100°C and 110°C, driving the reaction to completion while maintaining the integrity of the nitro and methylthio groups. From an impurity control perspective, the sequential nature of these reactions allows for intermediate purification, such as extraction and distillation, which removes unreacted starting materials and side products before they can propagate through the synthesis. This layered approach to purification ensures that the final API intermediate meets stringent quality specifications required by regulatory bodies.
How to Synthesize 4,6-Dichloro-2-Methylthio-5-Nitropyrimidine Efficiently
Executing this synthesis requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent embodiments to ensure reproducibility and safety. The process begins with the careful addition of nitric acid to diethyl malonate under cooling, followed by workup procedures involving organic solvent extraction and vacuum distillation to isolate the nitro-ester intermediate. Subsequent steps involve precise pH adjustments and temperature controls during the cyclization and methylation phases to optimize yield and minimize the formation of isomeric impurities. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions regarding the handling of corrosive reagents like phosphorus oxychloride and dimethyl sulfate.
- Nitration of diethyl malonate using concentrated nitric acid at 0-30°C to form diethyl 2-nitromalonate.
- Cyclization with thiourea and sodium alkoxide in alcohol solvent at 40-80°C to form the pyrimidine ring.
- Methylation using dimethyl sulfate in alkaline solution followed by chlorination with phosphorus oxychloride and DMA catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic advantages rooted in raw material economics and process scalability. The reliance on diethyl malonate and thiourea as primary feedstocks leverages a global supply chain of commodity chemicals that are produced in massive volumes, ensuring consistent availability and price stability compared to specialized custom building blocks. This fundamental shift in raw material strategy drastically simplifies the sourcing process and mitigates the risk of supply disruptions that often plague niche chemical markets. Furthermore, the operational simplicity of the four-step sequence reduces the requirement for highly specialized equipment or extreme reaction conditions, allowing for production in standard multipurpose chemical reactors. This flexibility enables manufacturers to respond rapidly to fluctuations in market demand without incurring significant capital expenditure on new infrastructure.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step protecting group strategies and the use of cost-effective reagents like phosphorus oxychloride significantly lowers the direct material costs associated with production. By optimizing the molar equivalents of reagents, such as using a slight excess of nitric acid to drive the initial nitration to completion, the process minimizes waste and maximizes atom economy. The ability to recover and recycle solvents like ethyl acetate and ethanol further contributes to substantial cost savings over the lifecycle of the product. Additionally, the high yields reported in the patent embodiments for the final chlorination step indicate a highly efficient conversion of intermediates, reducing the volume of waste disposal required and lowering environmental compliance costs.
- Enhanced Supply Chain Reliability: Utilizing a synthesis path based on widely available industrial chemicals ensures that the supply chain is resilient against geopolitical or logistical shocks that might affect specialty reagent availability. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without extensive re-validation. This portability is crucial for maintaining continuous supply to downstream pharmaceutical customers who require uninterrupted access to key intermediates for their own production schedules. The simplified workflow also reduces the lead time for batch production, allowing for faster turnaround from order placement to delivery of the final purified compound.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction exotherms that can be managed through standard cooling systems and workup procedures that utilize conventional separation techniques. The use of aqueous workups and common organic solvents facilitates waste stream management, making it easier to implement effective wastewater treatment protocols that meet strict environmental regulations. By avoiding the use of heavy metal catalysts or persistent organic pollutants, the synthesis aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this pyrimidine intermediate, based on the specific data and claims found within the patent documentation. These answers are designed to provide clarity on process capabilities, quality control measures, and the strategic benefits of this synthetic methodology for potential partners. Understanding these details is essential for making informed decisions about integrating this intermediate into broader drug discovery or agricultural chemical development pipelines.
Q: What are the critical reaction conditions for the nitration step?
A: The nitration of diethyl malonate requires strict temperature control between 0°C and 30°C using concentrated or fuming nitric acid to ensure safety and maximize the yield of diethyl 2-nitromalonate.
Q: Which catalyst is essential for the final chlorination step?
A: A small amount of N,N-dimethylaniline (DMA) is used as a catalyst during the chlorination with phosphorus oxychloride to facilitate the substitution of hydroxyl groups with chlorine atoms efficiently.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route utilizes common industrial reagents like diethyl malonate, thiourea, and phosphorus oxychloride, making it highly suitable for commercial scale-up and consistent supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Dichloro-2-Methylthio-5-Nitropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex fine chemical intermediates like 4,6-dichloro-2-methylthio-5-nitropyrimidine. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch delivered meets the highest standards required for pharmaceutical and agrochemical applications. We understand that consistency is key to your success, and our dedicated technical team is committed to maintaining the integrity of the supply chain from raw material intake to final product shipment.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us help you optimize your supply chain and accelerate your time to market with our proven expertise in pyrimidine chemistry.
