Scalable Acetone-Induced Fluoroalkylation for Advanced Pharmaceutical Intermediates Manufacturing
The landscape of organofluorine chemistry is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective synthetic methodologies. As detailed in the recent patent publication CN110642831A, a groundbreaking approach has been established for the fluoroalkylation of aromatic hydrocarbons and heteroaromatic hydrocarbons under the induction of acetone. This innovation addresses critical bottlenecks in the production of fluorine-containing organic compounds, which are indispensable building blocks in life sciences, biomedicine, and material science. By leveraging visible light irradiation and utilizing inexpensive, green acetone as both a solvent and an electron donor, this method achieves high yields of diverse fluoroalkyl-substituted products. For global procurement leaders and R&D directors, this represents a pivotal shift away from reliance on scarce precious metals toward a more robust, scalable, and economically viable manufacturing paradigm for high-purity pharmaceutical intermediates.
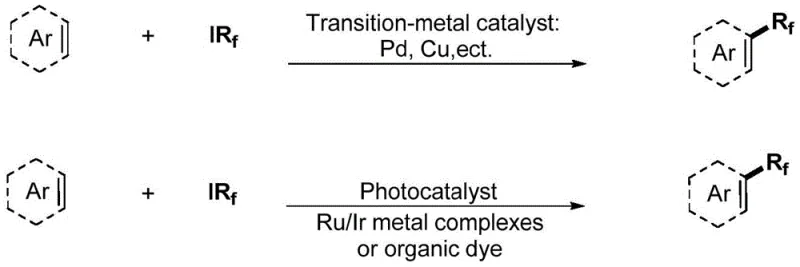
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluoroalkylated aromatic structures has been dominated by strategies relying heavily on transition-metal catalysts such as Palladium (Pd), Copper (Cu), Ruthenium (Ru), or Iridium (Ir). As illustrated in the upper portion of the reaction scheme, these conventional pathways often necessitate the use of sophisticated and costly ligand systems to stabilize the metal centers and facilitate the catalytic cycle. Furthermore, many of these catalytic systems exhibit high sensitivity to environmental factors; they are frequently intolerant to moisture and oxygen, mandating rigorous exclusion of air and water. This operational complexity often forces manufacturers to utilize glovebox techniques or elaborate Schlenk line setups, which drastically increases the capital expenditure and operational overhead associated with cost reduction in API manufacturing. The accumulation of heavy metal residues in the final product also poses significant regulatory hurdles, requiring extensive and expensive purification steps to meet stringent pharmacopeial standards.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN110642831A introduces a streamlined, metal-free alternative that fundamentally simplifies the reaction architecture. By employing acetone not merely as a passive medium but as an active electron donor compound under visible light irradiation, the process eliminates the need for exogenous photocatalysts or transition metals entirely. The reaction proceeds efficiently in a binary solvent system comprising acetone and a co-solvent such as DMF (N,N-dimethylformamide) or DMA (N,N-dimethylacetamide), facilitated by simple inorganic bases like sodium carbonate. This approach offers exceptional atom economy and functional group tolerance, allowing for the direct functionalization of complex molecular scaffolds without the protective group manipulations often required in metal-catalyzed variants. The result is a cleaner reaction profile that significantly lowers the barrier to entry for producing commercial scale-up of complex polymer additives and drug candidates.
Mechanistic Insights into Acetone-Induced Photocatalytic Fluoroalkylation
The core mechanistic advantage of this technology lies in the unique photochemical properties of acetone when exposed to visible light, specifically blue LEDs. Upon irradiation, acetone molecules enter an excited state capable of engaging in single-electron transfer (SET) processes with the iodo-fluoroalkyl reagents (such as ethyl iododifluoroacetate or perfluoroalkyl iodides). This interaction generates reactive fluoroalkyl radicals in situ, which subsequently attack the electron-rich aromatic or heteroaromatic rings. The presence of a mild base, typically sodium carbonate, plays a crucial role in neutralizing the acidic byproducts (such as HI) generated during the radical substitution, thereby driving the equilibrium toward the desired product. This mechanism avoids the formation of stable metal-complex intermediates that can lead to catalyst deactivation or difficult-to-remove impurities, ensuring a cleaner impurity profile essential for reducing lead time for high-purity electronic chemical precursors.
Furthermore, the substrate scope of this reaction is remarkably broad, encompassing a wide array of structurally diverse compounds as depicted in the generalized reaction scheme. The system effectively tolerates various substituents including alkoxy, alkyl, and amino groups on the aromatic rings. It successfully functionalizes five-membered heterocycles like thiophenes and furans, as well as fused bicyclic systems such as coumarins and quinolinones. The versatility extends to the fluoroalkyl source, accommodating both ester-functionalized groups (Rf = CF2COOEt) and perfluoroalkyl chains (Rf = CnF2n+1). This breadth of compatibility ensures that the method can be adapted for the synthesis of a vast library of fluorinated building blocks, providing a reliable foundation for developing new reliable agrochemical intermediate supplier portfolios without the need for method re-optimization for each new substrate class.
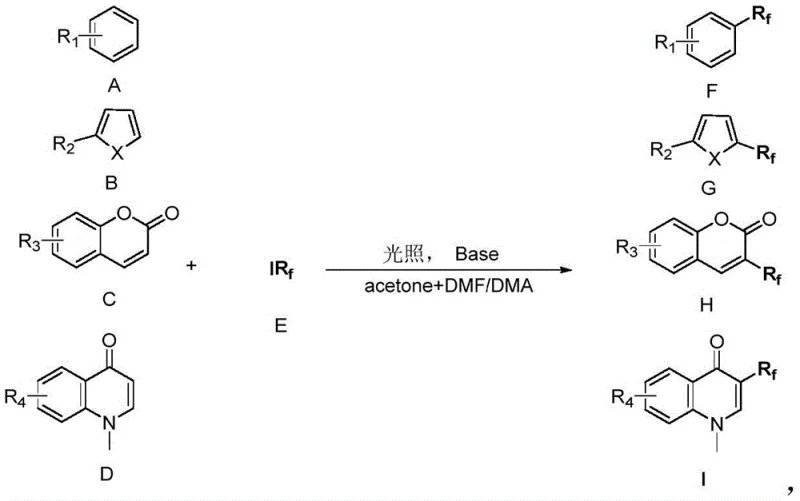
How to Synthesize Fluoroalkylated Aromatics Efficiently
Implementing this acetone-induced fluoroalkylation protocol requires precise control over reaction parameters to maximize yield and purity, particularly when transitioning from milligram-scale discovery to kilogram-scale production. The process involves mixing the aromatic substrate with the iodo-fluoroalkylating agent and a stoichiometric amount of base in the acetone/DMF solvent system, followed by irradiation with blue LEDs at ambient temperatures. While the general concept is straightforward, optimizing the molar ratios of the base and the fluoroalkyl source is critical; experimental data suggests that a slight excess of the fluoroalkyl reagent and base can significantly improve conversion rates. For detailed operational parameters, safety guidelines, and specific workup procedures tailored to your specific substrate, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining the aromatic or heteroaromatic substrate with the iodo-fluoroalkyl compound and a carbonate base in a solvent system of acetone and DMF.
- Purge the reaction vessel with inert gas such as argon to remove oxygen, ensuring a stable environment for the radical process.
- Irradiate the mixture with blue LEDs at room temperature for approximately 24 hours to drive the fluoroalkylation to completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acetone-induced methodology offers profound strategic advantages that extend beyond simple reaction yields. The most immediate impact is the drastic simplification of the raw material supply chain. By removing the dependency on volatile and geopolitically sensitive precious metals like Palladium and Iridium, manufacturers can insulate their production costs from the erratic fluctuations of the commodities market. Additionally, the use of commodity-grade solvents like acetone and DMF, coupled with inexpensive inorganic bases like sodium carbonate, ensures that the Bill of Materials (BOM) remains low and stable. This economic stability is crucial for long-term contract manufacturing agreements where price predictability is a key negotiation lever for securing reliable pharmaceutical intermediates supplier partnerships.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized ligands results in a substantial decrease in direct material costs. Furthermore, the removal of heavy metals from the process flow negates the need for costly scavenging resins or complex chromatographic purification steps typically required to meet residual metal specifications. This streamlining of the downstream processing phase reduces solvent consumption and waste disposal costs, contributing to a leaner and more profitable manufacturing operation. The ability to run reactions at room temperature also minimizes energy expenditures associated with heating or cryogenic cooling, further enhancing the overall cost-efficiency of the production line.
- Enhanced Supply Chain Reliability: Traditional metal-catalyzed reactions often suffer from batch-to-batch variability due to catalyst sensitivity to trace impurities or oxygen ingress. The robust nature of the acetone-induced radical mechanism provides superior process consistency, reducing the risk of failed batches and ensuring a steady flow of materials to downstream formulation units. Moreover, the reagents used in this protocol are widely available industrial chemicals with established global supply networks, mitigating the risk of supply disruptions that can occur with niche catalytic reagents. This reliability is essential for maintaining continuous production schedules and meeting tight delivery windows for critical high-purity OLED material precursors.
- Scalability and Environmental Compliance: The simplicity of the reaction setup—requiring only a light source and standard glassware or photoreactors—facilitates seamless scale-up from laboratory to pilot and commercial plant scales. The use of acetone, a green solvent with a favorable environmental profile, aligns with increasingly stringent global regulations regarding volatile organic compound (VOC) emissions and workplace safety. The absence of toxic heavy metals simplifies wastewater treatment protocols and reduces the environmental footprint of the manufacturing facility. These factors collectively position this technology as a future-proof solution for sustainable chemical manufacturing, appealing to stakeholders focused on ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel fluoroalkylation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, aiming to clarify the operational feasibility and strategic value of adopting this method for your specific application needs. Understanding these nuances is vital for making informed decisions about process integration and vendor selection.
Q: What is the primary advantage of using acetone in this fluoroalkylation method?
A: Acetone serves a dual role as both the main solvent and the electron donor compound, eliminating the need for expensive external photocatalysts or transition metal complexes typically required in traditional methods.
Q: Does this reaction require strict anhydrous conditions or glovebox operations?
A: No, unlike many transition-metal catalyzed processes that are sensitive to water and oxygen, this acetone-induced method operates efficiently under standard inert gas protection without the need for complex glovebox setups.
Q: What types of substrates are compatible with this synthesis route?
A: The method exhibits excellent functional group compatibility, successfully fluoroalkylating various aromatics, heteroaromatics like thiophenes and furans, as well as complex scaffolds such as coumarins and quinolinones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkylated Aromatics Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of acetone-induced photocatalysis in modernizing the synthesis of fluorinated building blocks. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical, industrial setting. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fluoroalkylated intermediate meets the highest quality standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with our technical team to evaluate the feasibility of adapting this green chemistry approach to your specific product portfolio. By leveraging our expertise in process optimization, we can help you achieve significant efficiency gains and cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project requirements. Let us help you secure a competitive edge through innovative and sustainable chemical manufacturing solutions.
