Advanced SeO2-Catalyzed Synthesis of Nitroarylamines for Commercial Scale Production
Advanced SeO2-Catalyzed Synthesis of Nitroarylamines for Commercial Scale Production
The chemical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing critical intermediates, and patent CN101130499B presents a groundbreaking methodology for the synthesis of nitroarylamine compounds. This intellectual property details a novel catalytic system utilizing selenium dioxide (SeO2) in conjunction with carbon monoxide and water to achieve the selective reduction of aromatic dinitro compounds. Unlike traditional high-pressure hydrogenation or polluting metal reduction methods, this technology operates under remarkably mild conditions, specifically at normal atmospheric pressure and moderate temperatures ranging from 20°C to 120°C. For R&D directors and process chemists, this represents a significant leap forward in controlling reaction selectivity, achieving mono-reduction rates exceeding 99% while preserving sensitive functional groups on the aromatic ring. The ability to utilize inexpensive reagents like carbon monoxide and water as the reducing equivalents, rather than expensive hydrogen gas or stoichiometric metal powders, fundamentally shifts the economic and safety profile of producing these valuable pharmaceutical intermediates.
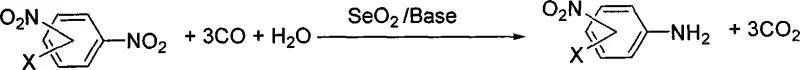
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nitroarylamines from dinitro precursors has relied heavily on methods that are increasingly untenable in the modern regulatory landscape. Traditional techniques such as iron powder reduction or sodium sulfide reduction, while technically simple, generate massive quantities of heavy metal sludge and sulfur-containing wastewater, creating severe environmental liabilities and high disposal costs. Furthermore, these stoichiometric reductions often suffer from poor selectivity, leading to over-reduction to diamines or incomplete conversion, which complicates downstream purification and lowers overall yield. Even catalytic hydrogenation methods, while cleaner, often require specialized high-pressure equipment and precious metal catalysts like palladium or platinum, which are susceptible to poisoning by impurities and represent a significant capital expenditure. The lack of chemoselectivity in many of these older processes means that sensitive substituents such as halogens, cyano groups, or carbonyls on the aromatic ring are frequently compromised, limiting the scope of substrates that can be effectively processed.
The Novel Approach
The methodology disclosed in CN101130499B offers a transformative alternative by leveraging a unique SeO2-catalyzed system that operates under normal pressure, thereby eliminating the need for costly high-pressure reactors and enhancing operational safety. By employing carbon monoxide and water as the terminal reductants, the process generates carbon dioxide as the primary byproduct, drastically simplifying the waste stream compared to metal-based reductions. The patent data demonstrates that this system exhibits exceptional chemoselectivity, successfully reducing one nitro group while leaving the second nitro group and other sensitive functionalities like fluoro, chloro, or trifluoromethyl groups completely untouched. This high degree of control allows for the synthesis of complex high-purity nitroarylamine derivatives that were previously difficult or expensive to access. Moreover, the catalyst loading can be optimized between 0.1% to 100% molar ratio, providing flexibility for cost-sensitive applications, and the reaction can proceed in common polar solvents like DMF or DMSO, ensuring ease of integration into existing manufacturing infrastructure.
Mechanistic Insights into SeO2-Catalyzed Selective Reduction
The core innovation of this technology lies in the specific interaction between selenium dioxide, carbon monoxide, and water within the organic phase. Mechanistically, the selenium dioxide acts as an oxygen transfer agent that facilitates the oxidation of carbon monoxide to carbon dioxide, simultaneously generating the active reduced selenium species in situ that effects the reduction of the nitro group. This catalytic cycle is distinct from direct hydrogenation as it avoids the formation of reactive radical species that typically lead to non-selective reduction of other functional groups. The presence of water is critical, serving not just as a proton source but potentially participating in the regeneration of the active catalytic species, ensuring the cycle continues efficiently. The patent examples illustrate that the reaction kinetics are highly dependent on the molar ratio of water to substrate, with optimal results observed when maintaining a balance that supports the catalytic turnover without diluting the reaction medium excessively. This precise mechanistic control is what enables the reported selectivity of over 99%, a figure that is exceptionally rare in the reduction of poly-nitro aromatics.
From an impurity control perspective, this mechanism offers profound advantages for the production of agrochemical intermediates and pharmaceutical building blocks. Because the reduction potential is tuned specifically for the nitro group via the selenium cycle, there is minimal risk of dehalogenation, a common side reaction in catalytic hydrogenation that can ruin batch quality. The data shows successful conversion of substrates containing fluorine, chlorine, and even aldehyde groups without degradation, indicating a very gentle reduction environment. Furthermore, the reaction byproduct is elemental selenium or soluble selenium species which can be easily separated from the organic product through simple filtration or distillation steps described in the embodiments. This ease of separation means that the final product requires less rigorous purification, reducing solvent consumption and energy usage during the workup phase. The robustness of the catalyst system against various electronic environments on the aromatic ring, whether electron-donating or electron-withdrawing, ensures consistent performance across a diverse library of substrates.
How to Synthesize Nitroarylamine Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the key reagents to maximize yield and minimize catalyst loading. The patent outlines a straightforward procedure where the aromatic dinitro compound is dissolved in a polar solvent such as DMF, DMSO, or ethylene glycol diethyl ether, followed by the addition of the selenium dioxide catalyst and an optional base promoter. The base, which can range from inorganic salts like sodium acetate to organic amines like triethylamine or DBU, plays a crucial role in modulating the reaction rate and can be adjusted from 0% to 400% molar ratio depending on the specific substrate reactivity. Once the mixture is prepared, carbon monoxide is introduced, and the system is heated to a temperature between 20°C and 120°C, with reaction times varying from 1 to 20 hours based on the specific thermal profile chosen. Detailed standardized synthetic steps for optimizing these parameters are provided in the guide below.
- Charge an organic solvent (such as DMF or DMSO) with aromatic dinitro compound, selenium dioxide catalyst, and optional base promoter under normal pressure.
- Introduce carbon monoxide and water into the reaction mixture and maintain temperature between 20°C and 120°C for 1 to 20 hours.
- Cool the mixture, switch gas flow to oxygen or air to oxidize residual catalyst, then filter and distill to isolate the high-purity nitroarylamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this SeO2-catalyzed technology translates directly into tangible operational efficiencies and risk mitigation. The shift away from high-pressure hydrogenation equipment significantly reduces the capital expenditure required for facility upgrades, as the reaction proceeds safely under normal atmospheric pressure. This inherent safety feature also lowers insurance premiums and reduces the regulatory burden associated with handling high-pressure hydrogen gas, thereby streamlining the approval process for new production lines. Additionally, the use of carbon monoxide, which can be sourced from industrial tail gases as noted in the patent embodiments, offers a pathway to utilize lower-cost feedstock materials, further driving down the variable cost of goods sold. The simplicity of the workup procedure, involving basic filtration and distillation, minimizes the demand for specialized separation equipment and reduces the turnaround time between batches, enhancing overall plant throughput.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of expensive precious metal catalysts like palladium or platinum, replacing them with selenium dioxide which is significantly more affordable and available. Since the catalyst loading can be as low as 0.1% molar ratio in optimized scenarios, the direct material cost for catalysis is negligible compared to traditional hydrogenation methods. Furthermore, the high selectivity of the reaction minimizes the formation of byproducts like diamines or dehalogenated species, which drastically improves the mass balance and reduces the loss of valuable starting materials. By avoiding the generation of heavy metal sludge associated with iron powder reduction, the facility also saves substantially on hazardous waste disposal fees and environmental compliance costs, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents such as selenium dioxide, carbon monoxide, and common organic solvents ensures a robust supply chain that is less susceptible to geopolitical disruptions or market volatility. Unlike processes dependent on specialized hydrogenation catalysts that may have long lead times or single-source suppliers, the materials for this synthesis are commodity chemicals with multiple global vendors. The mild reaction conditions also mean that the process is less energy-intensive, reducing exposure to fluctuating utility costs and ensuring consistent production capability even during periods of energy constraint. This stability allows for more accurate forecasting and inventory planning, ensuring that critical nitroarylamine intermediates are available to meet downstream customer demand without interruption.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of high-pressure constraints, allowing for the use of standard glass-lined or stainless steel reactors commonly found in fine chemical plants. The environmental profile of the process aligns perfectly with modern green chemistry initiatives, as the primary emission is carbon dioxide and the selenium catalyst can potentially be recovered or managed more easily than heavy metal sludge. This cleaner production profile simplifies the permitting process for capacity expansion and enhances the corporate sustainability metrics of the manufacturing site. The ability to handle a wide range of substrates with the same fundamental protocol also provides supply chain flexibility, allowing a single production line to manufacture diverse products without extensive retooling or cleaning validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective reduction technology. These answers are derived directly from the experimental data and claims within patent CN101130499B, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this method into your current production portfolio.
Q: What is the selectivity of the SeO2 catalytic reduction method?
A: The method described in patent CN101130499B achieves reduction selectivity as high as 99% for mono-nitro reduction, leaving sensitive groups like halogens and acyl groups intact.
Q: Can this process be scaled for industrial production?
A: Yes, the process operates under normal pressure and mild temperatures (20-120°C), utilizing inexpensive catalysts and common solvents, making it highly suitable for large-scale commercial manufacturing.
Q: How does this method compare to traditional iron powder reduction?
A: Unlike traditional iron powder or sulfide reduction which generates significant heavy metal waste and pollution, this SeO2/CO system is environmentally friendlier with easier product separation and lower three-waste treatment burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitroarylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced catalytic technologies like the SeO2-mediated reduction described in CN101130499B to enhance our service offerings. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with the necessary infrastructure to handle atmospheric pressure reactions safely and efficiently, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of nitroarylamine meets the exacting standards required by the global pharmaceutical and agrochemical industries. We are committed to leveraging this innovative chemistry to provide our clients with superior quality intermediates at competitive price points.
We invite you to engage with our technical team to explore how this specific synthetic route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic system for your specific target molecules. We encourage you to contact our technical procurement team today to discuss your project requirements,索取 specific COA data for similar compounds, and review our comprehensive route feasibility assessments. Let us partner with you to bring your next generation of high-value chemical products to market faster and more sustainably.
