Advanced Selenium-Catalyzed Synthesis of Disulfide Compounds for Commercial Scale-up
Introduction to Next-Generation Disulfide Manufacturing
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable and cost-effective methodologies. A pivotal advancement in this domain is documented in Chinese Patent CN101613247B, which outlines a robust method for synthesizing disulfide compounds using a selenium-catalyzed system. This technology represents a significant departure from traditional oxidative coupling methods, offering a streamlined one-pot procedure that operates under atmospheric pressure. By utilizing readily available raw materials such as halogenated hydrocarbons and elemental sulfur, combined with carbon monoxide and water, this process achieves high selectivity and yield without the need for expensive transition metal catalysts. For R&D directors and procurement managers alike, understanding the nuances of this patent is crucial for optimizing supply chains and reducing the overall cost of goods sold for critical pharmaceutical and agrochemical intermediates.
The core innovation lies in the dual role of selenium, which acts not only as a catalyst but also facilitates a unique phase-transfer mechanism. Unlike homogeneous catalysts that remain dissolved and require complex downstream purification to meet stringent residual metal specifications, the selenium in this process precipitates out as a solid upon exposure to air or oxygen at the end of the reaction. This inherent separability drastically simplifies the workup procedure, reducing solvent consumption and processing time. Furthermore, the reaction conditions are remarkably mild, ranging from 20°C to 100°C, which minimizes energy consumption and enhances operational safety. As we delve deeper into the technical specifics, it becomes evident that this methodology provides a compelling value proposition for the commercial scale-up of complex organic sulfides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of disulfide compounds has relied heavily on the oxidative coupling of mercaptans (thiols). While chemically straightforward, this approach suffers from severe practical drawbacks that hinder large-scale manufacturing. Mercaptans are notorious for their intense, unpleasant odor, posing significant health and safety challenges for plant operators and surrounding communities. Moreover, the oxidation step typically requires stoichiometric amounts of harsh oxidizing agents such as bromine, iodine, or metal oxides like manganese dioxide. These reagents not only increase raw material costs but also generate substantial quantities of hazardous waste, complicating environmental compliance and waste disposal logistics. Alternative routes involving Bunte salts or phase-transfer catalysis with halides often involve multi-step sequences with cumbersome isolation procedures, leading to lower overall throughput and higher capital expenditure for equipment.
The Novel Approach
In stark contrast, the selenium-catalyzed method described in the patent offers a direct, atom-economical pathway that bypasses the need for pre-formed thiols. By reacting halogenated hydrocarbons directly with sulfur powder in the presence of carbon monoxide and water, the process constructs the disulfide bond in a single operational step. The use of carbon monoxide serves as a reducing equivalent, facilitating the transformation under neutral to mildly basic conditions. This eliminates the requirement for strong, corrosive oxidants and mitigates the safety risks associated with handling volatile thiols. Additionally, the ability to operate at atmospheric pressure removes the need for specialized high-pressure autoclaves, significantly lowering the barrier to entry for contract manufacturing organizations. This novel approach effectively transforms a traditionally hazardous and waste-intensive process into a cleaner, safer, and more economically viable operation suitable for modern green chemistry standards.
Mechanistic Insights into Selenium-Catalyzed Coupling
To fully appreciate the technical superiority of this route, one must examine the underlying catalytic cycle. The reaction initiates with the activation of elemental sulfur by the selenium catalyst within the organic solvent matrix. Under the flow of carbon monoxide and in the presence of water, the selenium species likely forms an active selenocarbonyl or similar intermediate that interacts with the halogenated hydrocarbon substrate. This interaction promotes the nucleophilic attack of sulfur species on the alkyl or aryl halide, leading to the formation of the carbon-sulfur bond. The presence of a base, whether organic like triethylamine or inorganic like sodium carbonate, aids in neutralizing the hydrogen halide byproduct, driving the equilibrium towards the desired disulfide product. The versatility of the system allows for a wide range of substrates, including benzyl halides, allyl halides, and even heteroaryl derivatives, demonstrating broad functional group tolerance.
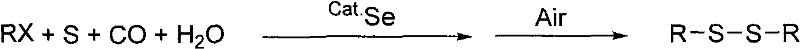
A particularly fascinating aspect of this mechanism is the so-called "phase transfer function" of the selenium catalyst. Initially, the selenium powder is insoluble in the reaction medium. However, upon heating and exposure to the reaction conditions, it transforms into a soluble active species that participates in the homogeneous catalytic cycle. This ensures efficient contact between the catalyst and the substrates, maximizing reaction rates. Upon completion of the reaction, switching the gas feed from carbon monoxide to air or oxygen triggers the re-precipitation of the selenium as a solid. This reversible solubility behavior combines the kinetic advantages of homogeneous catalysis with the separation benefits of heterogeneous catalysis. For quality control teams, this means that the final product stream is inherently free from dissolved selenium contaminants, simplifying the purification process and ensuring high purity specifications are met with minimal effort.
How to Synthesize Disulfide Compounds Efficiently
Implementing this synthesis route in a pilot or production setting requires careful attention to reaction parameters to maximize yield and purity. The patent provides extensive experimental data indicating that the molar ratio of halogenated hydrocarbon to sulfur should be maintained between 2:1 and 2:1.5 to ensure complete conversion while minimizing excess reagent waste. The amount of water introduced into the system is also critical, with optimal results observed when the molar ratio of halide to water ranges from 1:1 to 1:100, depending on the specific substrate solubility. Solvent selection plays a pivotal role, with polar aprotic solvents like DMF and DMSO showing superior performance compared to non-polar options, although toluene and benzene can also be utilized effectively. Detailed standardized synthetic steps see the guide below.
- Charge reactor with halogenated hydrocarbon, sulfur powder, selenium catalyst, base, and organic solvent under inert atmosphere.
- Continuously feed carbon monoxide gas and heat the mixture to 20-100°C for 1-24 hours to facilitate the coupling reaction.
- Switch gas flow to air or oxygen to precipitate unreacted selenium, filter, and add water to the filtrate to isolate the pure disulfide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this selenium-catalyzed technology translates into tangible strategic advantages beyond mere chemical efficiency. The primary benefit is the drastic simplification of the supply chain for raw materials. By eliminating the need for malodorous and hazardous thiols, facilities can reduce their inventory of dangerous goods, lower insurance premiums, and simplify regulatory reporting. Furthermore, the reliance on commodity chemicals like sulfur powder and common halides ensures a stable and resilient supply base, immune to the volatility often seen in specialized reagent markets. The atmospheric pressure operation also implies that existing glass-lined or stainless steel reactors can be utilized without costly modifications for high-pressure containment, accelerating the timeline from process development to commercial production.
- Cost Reduction in Manufacturing: The economic impact of replacing precious metal catalysts with non-metallic selenium cannot be overstated. Selenium is significantly cheaper than palladium, platinum, or rhodium, and its unique precipitation behavior allows for potential recovery and reuse, further driving down the cost per kilogram of the active ingredient. Additionally, the elimination of stoichiometric oxidants and the reduction in solvent usage during workup contribute to substantial savings in raw material and waste disposal costs. The simplified downstream processing, which avoids complex chromatography or heavy metal scavenging steps, reduces labor hours and utility consumption, resulting in a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions contributes to higher batch success rates and consistent product quality. Because the process tolerates industrial-grade carbon monoxide containing impurities like nitrogen and carbon dioxide, manufacturers are not held hostage by the availability of ultra-high-purity gases. This flexibility allows for sourcing from local industrial gas suppliers, reducing logistics lead times and transportation costs. Moreover, the short reaction times, typically ranging from 1 to 24 hours depending on temperature, enable faster turnaround times for custom synthesis projects, allowing suppliers to respond more agilely to fluctuating market demands and urgent customer requirements.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with modern sustainability goals. The process generates minimal waste, primarily consisting of recoverable selenium and aqueous salt solutions, avoiding the generation of heavy metal sludge associated with traditional oxidation methods. The absence of volatile thiols improves workplace air quality and reduces the risk of community odor complaints, which is a critical factor for maintaining social license to operate in densely populated industrial zones. This green profile facilitates easier permitting for capacity expansion and ensures long-term compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this disulfide synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using Selenium over transition metals for disulfide synthesis?
A: Selenium offers a cost-effective alternative to expensive transition metals like palladium or platinum. Crucially, the patent describes a unique phase-transfer behavior where selenium precipitates out as a solid post-reaction, allowing for easy filtration and potential recycling, thereby eliminating complex heavy metal removal steps required in traditional catalysis.
Q: Can this process utilize industrial grade Carbon Monoxide?
A: Yes, the technology is robust enough to handle industrial carbon monoxide tail gas containing impurities such as nitrogen, carbon dioxide, or water vapor, provided the air content remains below 10%. This flexibility significantly reduces raw material costs compared to processes requiring ultra-high purity gases.
Q: How does this method address the environmental issues associated with thiol oxidation?
A: Traditional thiol oxidation often generates malodorous waste and requires stoichiometric oxidants. This selenium-catalyzed route starts from stable halides and elemental sulfur, operating under atmospheric pressure with minimal waste generation. The catalyst recovery via precipitation further enhances the green chemistry profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disulfide Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the selenium-catalyzed synthesis route for producing high-value disulfide intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology from the laboratory bench to full-scale commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of reaction parameters—such as gas flow rates, temperature gradients, and precipitation kinetics—is maintained consistently across large batches. We operate state-of-the-art rigorous QC labs equipped to verify stringent purity specifications, guaranteeing that every shipment meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you are looking to optimize an existing legacy process or develop a new supply chain for complex sulfur-containing molecules, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how we can enhance your supply chain resilience and profitability.
