Advanced Nickel-Catalyzed Synthesis of Fluorinated Cyclopentenindanones for Pharmaceutical Applications
Advanced Nickel-Catalyzed Synthesis of Fluorinated Cyclopentenindanones for Pharmaceutical Applications
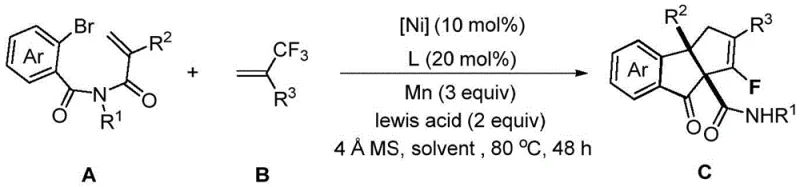
The rapid evolution of medicinal chemistry demands increasingly sophisticated synthetic methodologies to access complex fluorinated scaffolds efficiently. Patent CN114890912B discloses a groundbreaking preparation method for fluorocyclopentenindanone compounds, addressing a critical gap in the current landscape of organic synthesis. This technology leverages a robust nickel-catalyzed tandem reaction sequence involving 2-bromo-N-acryloyl benzamides and trifluoromethyl olefins to construct highly functionalized indanone cores. The significance of this innovation lies in its ability to introduce fluorine atoms, which are known to drastically improve the metabolic stability and bioavailability of drug candidates, directly into rigid cyclic systems. By utilizing earth-abundant nickel catalysts rather than expensive precious metals, this process offers a sustainable and economically viable pathway for generating high-value pharmaceutical intermediates. The methodology represents a substantial leap forward for reliable pharmaceutical intermediate supplier networks seeking to optimize their catalog of fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fluorinated cyclopentenindanone skeletons has been plagued by significant synthetic challenges that hinder widespread adoption in industrial settings. Traditional routes often rely on multi-step sequences involving harsh reaction conditions, such as strong acids or high temperatures, which can lead to the decomposition of sensitive functional groups. Furthermore, many existing protocols depend on stoichiometric amounts of expensive transition metal catalysts or specialized fluorinating reagents that are difficult to source and handle safely on a large scale. The lack of atom economy in these conventional methods results in substantial waste generation, creating environmental burdens and increasing the overall cost of goods sold for the final active pharmaceutical ingredient. Additionally, the regioselectivity and stereoselectivity control in older methods are frequently poor, necessitating tedious purification steps that further erode profit margins and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a streamlined nickel-catalyzed cascade reaction that elegantly solves these longstanding issues. By employing a catalytic system composed of nickel acetylacetonate and a terpyridine ligand, the reaction proceeds through a sophisticated mechanism involving cyclization, cross-coupling, and beta-fluorine elimination in a single operational step. This tandem process not only simplifies the synthetic route but also ensures high levels of chemo- and regioselectivity, minimizing the formation of unwanted byproducts. The reaction conditions are remarkably mild, typically operating at 80°C in a mixed solvent system of DMSO and THF, which preserves the integrity of delicate substituents on the aromatic rings. This method effectively transforms simple, commercially available starting materials into complex fluorinated architectures with impressive efficiency, setting a new standard for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nickel-Catalyzed Tandem Cyclization
The core of this transformative technology lies in the intricate catalytic cycle driven by the nickel complex, which orchestrates a series of bond-forming events with precision. The mechanism initiates with the oxidative addition of the nickel(0) species into the carbon-bromine bond of the 2-bromo-N-acryloyl benzamide substrate, generating a reactive organonickel intermediate. This species subsequently undergoes migratory insertion into the pendant alkene moiety, facilitating the formation of the initial cyclic structure. Crucially, the presence of the trifluoromethyl olefin allows for a cross-coupling event where the nickel center interacts with the electron-deficient double bond, leading to the incorporation of the fluorinated fragment. The subsequent beta-fluorine elimination step is particularly noteworthy, as it restores the unsaturation in the cyclopentene ring while retaining a specific fluorine atom, thereby establishing the unique fluorocyclopentenindanone motif. Understanding this mechanistic pathway is vital for R&D teams aiming to further optimize reaction parameters or adapt the chemistry to novel substrate classes.
From an impurity control perspective, the choice of ligands and additives plays a pivotal role in ensuring the purity of the final product. The use of terpyridine as a ligand stabilizes the nickel center throughout the catalytic cycle, preventing the formation of inactive nickel black or off-cycle species that could lead to side reactions. Moreover, the inclusion of manganese powder as a reducing agent ensures the continuous regeneration of the active nickel(0) catalyst from nickel(II) species formed during the cycle, maintaining high turnover numbers. The addition of Lewis acids like manganese bromide further enhances the electrophilicity of certain intermediates, promoting the desired nucleophilic attack and ring contraction steps. This careful balancing of redox potentials and coordination environments minimizes the generation of structural isomers or defluorinated byproducts, resulting in a cleaner crude reaction mixture that requires less intensive downstream processing to meet stringent purity specifications.
How to Synthesize Fluorocyclopentenindanone Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield and reproducibility. The process begins with the rigorous exclusion of oxygen and moisture, typically achieved by conducting the reaction under an inert argon atmosphere within sealed vessels. The precise stoichiometry of reagents is critical, with the patent specifying optimal molar ratios of substrate to catalyst to reducing agent to ensure complete conversion. Solvent selection is equally important, with a 1:1 mixture of DMSO and THF providing the ideal polarity and solubility profile for the reactants and intermediates. While the general protocol is robust, slight adjustments in temperature or reaction time may be necessary depending on the electronic nature of the specific substrates employed. For a detailed breakdown of the standardized synthesis steps, please refer to the guide below.
- Prepare the reaction mixture by combining nickel acetylacetonate, terpyridine ligand, manganese powder, manganese bromide, and 4 Angstrom molecular sieves in a reaction vessel under argon atmosphere.
- Add the substrate 2-bromo-N-acryloyl benzamide and the trifluoromethyl olefin coupling partner to the mixture containing DMSO and THF solvent.
- Seal the reaction tube and heat the mixture to 80°C for 48 hours, followed by concentration and purification via flash column chromatography to isolate the product.

Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply security. The shift from precious metal catalysts to abundant nickel salts dramatically lowers the raw material costs associated with the synthesis, directly impacting the bottom line of the manufacturing budget. Furthermore, the simplicity of the reaction setup and workup reduces the requirement for specialized equipment and extensive labor, streamlining the overall production workflow. The high functional group tolerance of the reaction means that a wider variety of building blocks can be sourced from general chemical suppliers without needing custom synthesis, enhancing supply chain resilience. By integrating this technology, companies can achieve substantial cost savings while simultaneously shortening the timeline from process development to commercial launch.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with inexpensive nickel acetylacetonate and manganese powder significantly decreases the direct material costs per kilogram of product. This economic advantage is compounded by the high atom economy of the tandem reaction, which minimizes waste disposal fees and solvent consumption. Additionally, the mild reaction conditions reduce energy expenditures related to heating and cooling, contributing to a leaner and more profitable manufacturing process. The elimination of complex protection and deprotection steps further simplifies the synthesis, reducing the total number of unit operations required.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-bromo-N-acryloyl benzamides and trifluoromethyl olefins, are structurally simple and can be synthesized from readily available commodity chemicals. This reduces dependency on single-source suppliers for exotic reagents, thereby mitigating the risk of supply disruptions. The robustness of the reaction conditions also implies a wider operating window, making the process less susceptible to minor fluctuations in utility supplies or environmental conditions. Consequently, manufacturers can maintain consistent production schedules and meet delivery commitments with greater confidence, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream clients.
- Scalability and Environmental Compliance: The protocol is inherently scalable, as demonstrated by its successful application across a broad range of substrates without significant loss in efficiency. The use of less toxic nickel compared to other heavy metals aligns better with modern green chemistry principles and regulatory standards regarding residual metals in drug substances. Simplified purification processes mean less solvent waste is generated, easing the burden on wastewater treatment facilities and lowering the environmental footprint of the production site. This sustainability profile is increasingly important for meeting corporate social responsibility goals and satisfying the rigorous audit requirements of global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They serve to clarify the operational parameters and strategic benefits for stakeholders evaluating this synthetic route. Understanding these details is essential for making informed decisions about process adoption and resource allocation.
Q: What are the key advantages of this nickel-catalyzed method over traditional synthesis routes?
A: This method utilizes inexpensive nickel catalysts instead of precious metals, operates under mild conditions (80°C), and exhibits excellent functional group tolerance, significantly reducing production costs and complexity.
Q: What types of substrates are compatible with this fluorination protocol?
A: The protocol demonstrates broad substrate scope, accommodating various substituted 2-bromo-N-acryloyl benzamides including those with halogen, methoxy, or methyl groups, as well as diverse trifluoromethyl olefins with electron-withdrawing groups.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of cheap reagents like manganese powder and nickel salts, combined with simple workup procedures and high atom economy, makes this route highly amenable to commercial scale-up for API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorocyclopentenindanone Supplier
As the demand for fluorinated drug candidates continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative for success. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We understand the complexities of handling fluorinated intermediates and have optimized our processes to deliver consistent quality and reliability.
We invite you to engage with our technical procurement team to discuss how this innovative nickel-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your R&D and supply chain teams to make data-driven decisions for your next-generation pharmaceutical programs.
