Advanced Synthesis of Trimeric Indenyl BODIPY-Fullerene Star Compounds for High-Efficiency Optoelectronic Applications
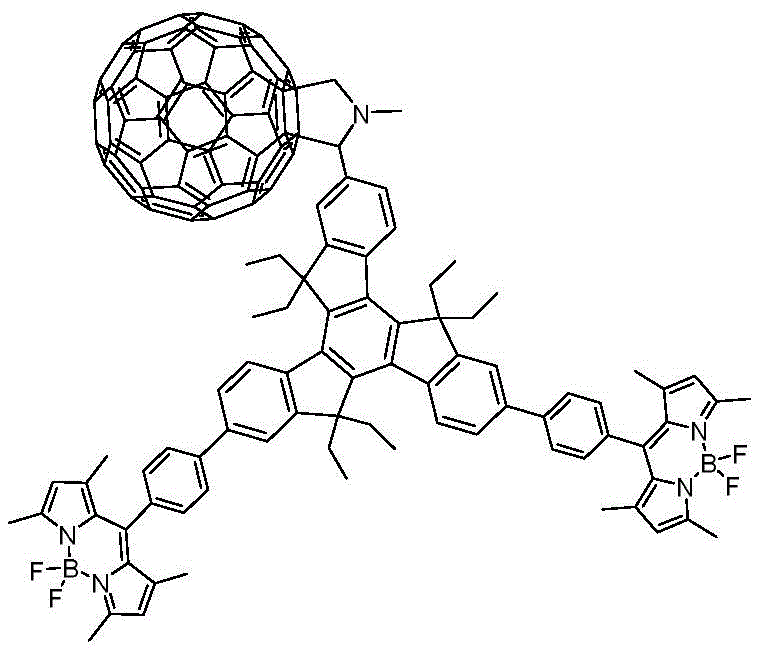
The rapid evolution of organic photovoltaics and molecular electronics demands sophisticated architectures capable of efficient charge separation and energy transfer. Patent CN108997391B introduces a groundbreaking methodology for synthesizing a trimeric indenyl BODIPY-fullerene star compound, designated as Formula (I). This unique molecular design integrates the rigid, highly conjugated triindenyl core with photoactive BODIPY donors and fullerene acceptors, creating a three-dimensional star-shaped topology that significantly enhances solubility and optoelectronic performance. For R&D directors and procurement specialists in the electronic materials sector, this patent represents a critical advancement in creating reliable electronic chemical supplier pathways for next-generation solar cell materials. The synthesis leverages robust transition metal catalysis and cycloaddition chemistry to achieve a complex donor-acceptor system under relatively mild conditions, addressing the historical challenges of low yields and structural complexity associated with star-shaped molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of star-shaped donor-acceptor systems has been plagued by synthetic inefficiencies that hinder commercial scalability. Traditional routes often involve multi-step sequences with harsh reaction conditions, leading to significant decomposition of sensitive photoactive units like BODIPY or fullerene derivatives. Conventional methods frequently suffer from poor regioselectivity during the functionalization of the central core, resulting in difficult-to-separate isomeric mixtures that compromise the purity required for high-performance electronic devices. Furthermore, the incorporation of fullerene cages typically requires aggressive conditions that can damage the delicate conjugated linkers, reducing the overall quantum yield of the final material. These limitations translate directly into increased manufacturing costs and extended lead times, creating bottlenecks for supply chain heads seeking consistent quality in display and optoelectronic materials manufacturing.
The Novel Approach
The methodology outlined in CN108997391B overcomes these barriers through a streamlined two-step strategy that prioritizes modularity and yield optimization. By utilizing a Suzuki-Miyaura cross-coupling reaction as the initial assembly step, the process ensures high fidelity in connecting the BODIPY arms to the triindenyl aldehyde core with excellent tolerance for functional groups. This is followed by a 1,3-dipolar cycloaddition (Prato reaction) to attach the fullerene units, a transformation known for its reliability and ability to proceed under thermal conditions without degrading the sensitive fluorophore. This novel approach not only simplifies the purification workflow through standard silica gel chromatography but also preserves the integrity of the electron-donating and accepting moieties, ensuring the final star compound exhibits the intended strong light absorption and efficient intramolecular energy transfer properties essential for advanced photonic applications.
Mechanistic Insights into Suzuki Coupling and 1,3-Dipolar Cycloaddition
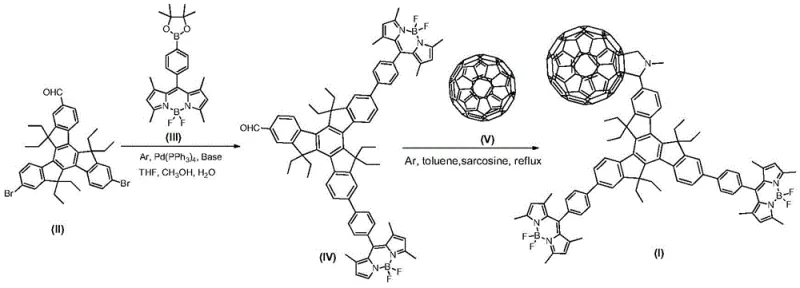
The core of this synthesis relies on the precise orchestration of palladium-catalyzed cross-coupling mechanisms to build the molecular scaffold. In the first stage, the 7,12-dibromo triindenal derivative undergoes oxidative addition with the tetrakis(triphenylphosphine)palladium(0) catalyst, activating the carbon-bromine bonds for subsequent transmetallation with the meso-phenylboronate BODIPY derivative. The presence of a carbonate base facilitates the formation of the reactive boronate species, driving the equilibrium towards the coupled product while minimizing homocoupling side reactions. This step is critical for establishing the rigid planar geometry of the triindenyl core, which serves as the central hub for the star architecture, and the reaction parameters—specifically temperatures between 65-85°C and durations of 20-26 hours—are optimized to balance reaction kinetics with the thermal stability of the BODIPY units.
Following the formation of the trimeric indenyl BODIPY intermediate, the mechanism shifts to a thermal 1,3-dipolar cycloaddition to install the fullerene acceptors. In this process, sarcosine reacts with the aldehyde groups on the intermediate to generate an azomethine ylide in situ, a highly reactive 1,3-dipole. This dipole immediately attacks the electron-deficient 6,6-junctions of the C60 fullerene cage in a concerted [3+2] cycloaddition, forming a stable pyrrolidine ring linkage. This mechanistic pathway is advantageous because it occurs under reflux in toluene at 110-120°C, avoiding the need for photochemical activation or extreme pressures. The result is a robust covalent bond that effectively bridges the donor and acceptor domains, facilitating the rapid charge separation observed in the final material's photophysical profile.
How to Synthesize Trimeric Indenyl BODIPY-Fullerene Efficiently
Executing this synthesis requires strict adherence to inert atmosphere techniques and precise stoichiometric control to maximize the yield of the complex star-shaped architecture. The process begins with the rigorous exclusion of oxygen to prevent catalyst deactivation during the Suzuki coupling, followed by careful temperature ramping to initiate the cycloaddition without inducing thermal degradation. Operators must utilize high-purity solvents, particularly freshly dried toluene for the second step, to prevent hydrolysis of the reactive intermediates. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and high purity suitable for electronic grade applications.
- Perform Suzuki coupling between 7,12-dibromo triindenal derivative and meso-phenylboronate BODIPY derivative using Pd catalyst at 65-85°C.
- Purify the resulting trimeric indenyl BODIPY intermediate via silica gel column chromatography using petroleum ether/dichloromethane.
- React the intermediate with Fullerene C60 and sarcosine in toluene at 110-120°C via 1,3-dipolar cycloaddition to form the final star compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The reliance on commercially available starting materials, such as standard fullerene C60 and accessible boronic acid derivatives, mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the elimination of exotic reagents or cryogenic conditions simplifies the infrastructure requirements for production, allowing for cost reduction in electronic chemical manufacturing by utilizing standard reactor setups found in most fine chemical facilities. The robustness of the reaction conditions also implies a lower rate of batch failure, enhancing overall supply reliability.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal removal steps often associated with more complex cross-coupling variants, as the palladium loading is kept relatively low and the workup is straightforward. By avoiding harsh acidic or basic conditions that require specialized corrosion-resistant equipment, the process significantly lowers capital expenditure and maintenance costs. Additionally, the high selectivity of the reactions reduces the burden on downstream purification, minimizing solvent consumption and waste disposal fees associated with separating complex byproduct mixtures.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the independent sourcing of the BODIPY arms and the triindenyl core, creating a resilient supply network that is less vulnerable to single-point failures. Since the reaction tolerances are broad regarding temperature and time windows within the specified ranges, production scheduling becomes more flexible, reducing lead time for high-purity optoelectronic intermediates. This flexibility ensures that manufacturing partners can scale up production rapidly in response to market demand fluctuations without compromising product quality.
- Scalability and Environmental Compliance: The use of common organic solvents like toluene, THF, and dichloromethane aligns with established solvent recovery protocols, facilitating easier compliance with environmental regulations compared to processes requiring chlorinated or highly toxic solvents. The mild thermal conditions reduce energy consumption per kilogram of product, contributing to a lower carbon footprint for the manufacturing process. Moreover, the simplicity of the isolation steps, primarily involving filtration and evaporation, supports seamless scale-up from laboratory grams to commercial tonnage without the need for complex continuous flow reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this advanced star-shaped compound. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this material for integration into their device architectures. Understanding these nuances is essential for assessing the feasibility of adopting this technology in large-scale manufacturing environments.
Q: What are the key reaction conditions for the Suzuki coupling step?
A: The Suzuki coupling requires an argon atmosphere, temperatures between 65-85°C, and a reaction time of 20-26 hours using a palladium catalyst and carbonate base.
Q: How is the final star-shaped compound purified?
A: The final product is purified using silica gel column chromatography with a dichloromethane and petroleum ether mixture as the eluent to ensure high purity.
Q: What applications benefit from this BODIPY-fullerene architecture?
A: This architecture is ideal for solar cells, molecular absorption antennas, and artificial photosynthesis due to its efficient intramolecular energy transfer capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimeric Indenyl BODIPY-Fullerene Supplier
As the global demand for high-efficiency organic photovoltaics and molecular sensors accelerates, securing a partner with deep expertise in complex organic synthesis is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless. Our stringent purity specifications and rigorous QC labs guarantee that every batch of trimeric indenyl BODIPY-fullerene meets the exacting standards required for high-performance electronic applications, minimizing variability in device performance.
We invite forward-thinking organizations to collaborate with us to leverage this innovative technology for their next-generation products. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
