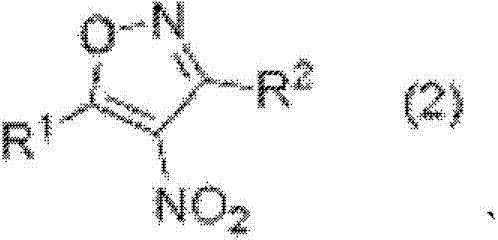
Scalable Production of 5-Trifluoromethyl-4-Nitro-2-Isoxazoline Derivatives for Advanced Material Synthesis
The chemical landscape for functional material synthesis is constantly evolving, driven by the need for more efficient and direct methodologies to access complex heterocyclic scaffolds. Patent CN103180303A introduces a groundbreaking approach for the manufacture of 5-trifluoromethyl-4-nitro-2-isoxazoline compounds, addressing a long-standing gap in synthetic organic chemistry where direct trifluoromethylation of the isoxazoline core was previously unreported. This technology leverages a direct trifluoromethylation strategy using Ruppert's reagent, allowing for the precise installation of the trifluoromethyl group at the 5-position of 4-nitro-2-isoxazole derivatives. The significance of this innovation extends beyond academic interest, offering tangible benefits for the industrial production of high-value intermediates used in agrochemicals and pharmaceuticals. By bypassing traditional multi-step building block strategies, this method provides a streamlined pathway that enhances both atom economy and process efficiency. For R&D teams and procurement specialists alike, understanding the mechanistic nuances and commercial implications of this patent is crucial for securing a competitive edge in the supply of specialized fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-trifluoromethyl-2-isoxazoline compounds has relied heavily on building block methods that necessitate the use of pre-functionalized starting materials containing the trifluoromethyl group. Conventional routes typically involve the cyclization of hydroxamic acid halides with trifluoromethyl-substituted olefins or the reaction of beta-trifluoromethyl-alpha,beta-unsaturated carbonyl compounds with hydroxylamine. These traditional approaches suffer from inherent limitations, including the requirement for specialized and often expensive trifluoromethylated precursors that may not be readily available on a large scale. Furthermore, the multi-step nature of constructing the isoxazoline ring from these specific building blocks can lead to cumulative yield losses and increased waste generation. The lack of flexibility in modifying the substitution pattern post-cyclization also restricts the structural diversity accessible through these older methods. Consequently, the industry has faced challenges in scaling up production while maintaining cost-effectiveness and supply chain stability for these critical intermediates.
The Novel Approach
In stark contrast to the legacy techniques, the method disclosed in CN103180303A utilizes a direct conjugate addition strategy that fundamentally simplifies the synthetic architecture. This novel approach employs 4-nitro-2-isoxazole compounds as the reaction substrate, capitalizing on the electron-withdrawing nature of the nitro group at the 4-position to activate the 5-position for nucleophilic attack. By reacting these substrates with (trifluoromethyl)trimethylsilane in the presence of a phase transfer catalyst and a base, the trifluoromethyl anion is generated in situ and adds directly to the isoxazoline ring. This eliminates the need for pre-installed trifluoromethyl groups on the olefinic precursors, thereby broadening the scope of accessible starting materials and reducing raw material costs. The process is robust, operating effectively under mild conditions, and allows for the subsequent acid treatment to isolate specific diastereomers, enhancing the purity profile of the final product.
Mechanistic Insights into Direct Trifluoromethylation
The core of this technological advancement lies in the generation and reactivity of the trifluoromethyl anion species derived from Ruppert's reagent, (trifluoromethyl)trimethylsilane. In the presence of a fluoride source or a suitable base system enhanced by a phase transfer catalyst, the silicon-carbon bond is activated to release the nucleophilic CF3 species. This anion then undergoes a conjugate addition (Michael-type addition) to the electron-deficient double bond of the 4-nitro-2-isoxazole ring. The nitro group at the 4-position plays a pivotal role by significantly lowering the LUMO energy of the adjacent carbon, making it highly susceptible to nucleophilic attack. This mechanistic pathway ensures high regioselectivity, directing the trifluoromethyl group exclusively to the 5-position of the heterocycle. The use of phase transfer catalysts, such as cetyltrimethylammonium bromide, facilitates the interaction between the ionic reagents and the organic substrate in the solution phase, ensuring homogeneous reaction kinetics and consistent conversion rates across different batches.
Following the initial addition, the reaction mixture undergoes a critical acid treatment step which serves multiple purposes regarding impurity control and stereochemical definition. The acid workup protonates the intermediate enolate species, stabilizing the newly formed C-C bond and yielding the saturated 2-isoxazoline ring system. Importantly, this step has been found to influence the diastereoselectivity of the product, allowing for the isolation of specific stereoisomers such as the (4S,5S) or (4R,5R) configurations depending on the reaction conditions and substrate structure. This level of control over the stereochemical outcome is vital for pharmaceutical applications where biological activity is often enantiomer-dependent. By minimizing the formation of unwanted by-products and simplifying the purification process through crystallization or standard chromatography, this mechanism supports the production of high-purity intermediates required for sensitive downstream applications in drug discovery and agrochemical formulation.
How to Synthesize 5-Trifluoromethyl-4-Nitro-2-Isoxazoline Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation within standard chemical manufacturing facilities. The process begins with the dissolution of the 4-nitro-2-isoxazole substrate in a polar aprotic solvent, with dimethylformamide (DMF) identified as the optimal medium for solubility and reaction rate. To this solution, a phase transfer catalyst and a mild inorganic base, preferably sodium acetate, are added to create the reactive environment necessary for trifluoromethylation. The careful addition of (trifluoromethyl)trimethylsilane at room temperature initiates the reaction, which proceeds smoothly over a period of 3 to 9 hours without the need for extreme temperatures or high-pressure equipment. Detailed standardized synthesis steps follow below to guide process engineers in replicating these results.
- Dissolve the 4-nitro-2-isoxazole substrate, a phase transfer catalyst such as cetyltrimethylammonium bromide, and a base like sodium acetate in a polar aprotic solvent such as dimethylformamide.
- Add (trifluoromethyl)trimethylsilane (Ruppert's reagent) to the reaction mixture at room temperature and stir for 3 to 9 hours to facilitate conjugate addition.
- Perform an acid workup using hydrochloric acid, followed by organic extraction, drying, and purification via column chromatography or recrystallization to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this direct trifluoromethylation technology presents a compelling value proposition centered on cost optimization and supply security. By shifting away from complex building block strategies that rely on scarce trifluoromethylated olefins, manufacturers can source more common and affordable 4-nitro-2-isoxazole precursors. This fundamental change in the bill of materials reduces the dependency on specialized suppliers and mitigates the risk of raw material shortages that often plague niche chemical markets. Furthermore, the simplified reaction workflow reduces the number of unit operations required, leading to lower utility consumption and reduced waste disposal costs associated with multi-step syntheses. The ability to produce these high-value intermediates more efficiently translates directly into improved margins and more competitive pricing structures for the final active ingredients.
- Cost Reduction in Manufacturing: The elimination of expensive pre-functionalized building blocks significantly lowers the direct material costs associated with producing 5-trifluoromethyl-4-nitro-2-isoxazoline derivatives. Since the trifluoromethyl group is introduced directly using a widely available reagent, the overall cost of goods sold is substantially reduced compared to traditional cyclization methods. Additionally, the use of mild reaction conditions minimizes energy expenditure, as there is no need for cryogenic cooling or high-temperature heating, further contributing to operational savings. The streamlined purification process also reduces solvent usage and chromatography media costs, enhancing the overall economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as sodium acetate and standard solvents like DMF ensures a robust and resilient supply chain that is less vulnerable to market fluctuations. The reliance on Ruppert's reagent, which is commercially produced at scale, guarantees a steady supply of the key trifluoromethylating agent without the lead time issues associated with custom-synthesized intermediates. This stability allows for better production planning and inventory management, ensuring that downstream customers in the pharmaceutical and agrochemical sectors receive their orders on time. The scalability of the process from gram to kilogram scales without significant re-optimization further strengthens supply continuity for long-term commercial contracts.
- Scalability and Environmental Compliance: The reaction operates under atmospheric pressure and near-room temperature conditions, which simplifies the engineering requirements for scale-up and reduces the safety risks associated with high-pressure reactors. The straightforward workup procedure involving acid treatment and extraction generates less hazardous waste compared to processes requiring heavy metal catalysts or harsh reagents. This aligns well with modern environmental regulations and sustainability goals, making it easier to obtain necessary permits for commercial production. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the environmental burden of solvent-intensive purification steps and supporting greener manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel isoxazoline compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or new product development pipelines.
Q: What is the primary advantage of this direct trifluoromethylation method over conventional building block approaches?
A: The primary advantage is the ability to directly introduce the trifluoromethyl group into the 5-position of the isoxazoline ring without needing pre-functionalized trifluoromethylated starting materials, significantly simplifying the synthetic route and reducing raw material costs.
Q: Which reagents are critical for achieving high yields in this transformation?
A: The process relies on (trifluoromethyl)trimethylsilane as the trifluoromethyl source, combined with a phase transfer catalyst like cetyltrimethylammonium bromide and a mild base such as sodium acetate to activate the nucleophile effectively.
Q: What are the potential industrial applications for these 5-trifluoromethyl-4-nitro-2-isoxazoline compounds?
A: These compounds serve as versatile intermediates for the production of functional materials, including pest control agents (agrochemicals), pharmaceutical active ingredients, and electronic materials, due to their unique structural properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-4-Nitro-2-Isoxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this direct trifluoromethylation technology for the next generation of pharmaceutical and agrochemical products. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reagent handling requirements of this process, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality intermediates. We are committed to delivering consistent batch-to-batch reproducibility that meets the exacting standards of global regulatory bodies.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can accelerate your time-to-market while reducing overall production costs.
