Advanced Synthesis of 4-Iodophenyl Carborane Derivatives for High-Performance Optoelectronic Materials
The landscape of advanced optoelectronic materials is undergoing a significant transformation, driven by the demand for highly stable and reactive building blocks capable of supporting next-generation organic light-emitting diodes (OLEDs) and neutron capture therapy agents. Patent CN112079856A introduces a groundbreaking methodology for the synthesis of 4-iodophenyl substituted carborane derivatives, addressing critical bottlenecks in traditional carborane chemistry. This innovation leverages a unique triazene-mediated strategy to efficiently introduce iodine atoms into the alkyne skeleton, subsequently facilitating the construction of the robust icosahedral carborane cage. Unlike conventional approaches that rely on unstable bromo-precursors and harsh cryogenic conditions, this novel route utilizes commercially available 4-iodoaniline and decaboric acid diacetonitrile complexes to achieve superior yields and purity. The strategic incorporation of the triazene moiety not only enhances the solubility of intermediate species but also streamlines the purification process, marking a pivotal advancement for manufacturers seeking reliable electronic chemical suppliers capable of delivering high-performance materials.
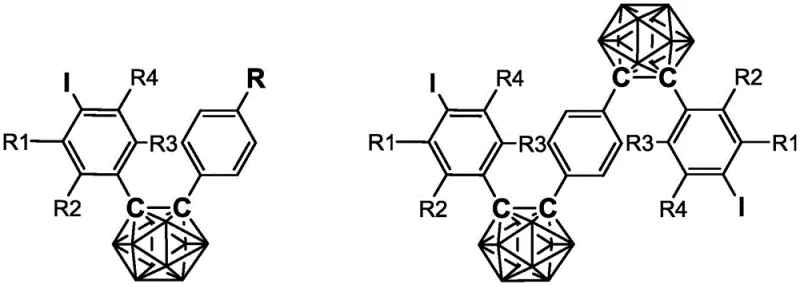
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carborane-based optoelectronic functional materials has been hindered by the reliance on bromine-substituted phenylcarborane precursors, which exhibit inherently poor reactivity in transition metal-catalyzed coupling reactions. The traditional pathway to access more reactive iodine-substituted analogs typically involves a multi-step sequence starting from bromo-alkyne raw materials, necessitating the use of strong bases such as butyllithium for debromination at extremely low temperatures, often around -78°C. This cryogenic requirement imposes severe constraints on process scalability and operational safety, particularly when handling large volumes of pyrophoric reagents. Furthermore, the solubility of conjugated alkynes at these low temperatures is notoriously poor, leading to incomplete reactions, heterogeneous mixtures, and significant resource wastage due to low overall yields. These technical barriers have long prevented the widespread commercial adoption of high-purity carborane derivatives in cost-sensitive industrial applications, creating a pressing need for a more robust and thermally tolerant synthetic methodology.
The Novel Approach
The methodology disclosed in patent CN112079856A represents a paradigm shift by circumventing the need for cryogenic debromination entirely through the innovative use of triazene intermediates. This approach initiates with a Sonogashira coupling reaction between a triazene-functionalized iodobenzene and various substituted alkynes, generating soluble alkyne intermediates that remain stable under ambient conditions. The subsequent iodine exchange step is performed thermally at 110-120°C using methyl iodide, a process that is far more operationally simple and safer than handling organolithium reagents. By maintaining the reaction in a homogeneous phase with improved solubility profiles, this novel route ensures higher conversion rates and facilitates easier downstream purification via standard column chromatography. The result is a series of 4-iodophenyl carborane derivatives with diverse substituent groups, synthesized with high efficiency and reproducibility, positioning this technology as a cornerstone for cost reduction in electronic chemical manufacturing.
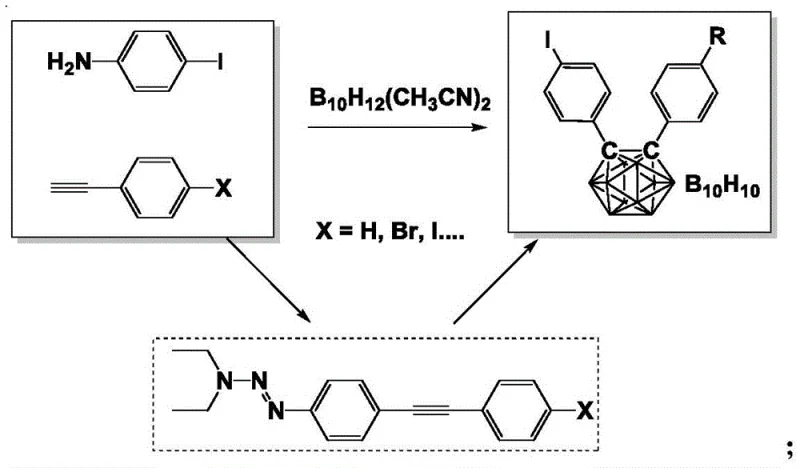
Mechanistic Insights into Triazene-Mediated Iodine Exchange and Cage Closure
The core mechanistic advantage of this synthesis lies in the dual functionality of the triazene group, which serves as both a solubility enhancer and a latent leaving group for iodine installation. In the initial Sonogashira coupling phase, the triazene moiety prevents the precipitation of large conjugated systems that typically plague traditional syntheses, ensuring that the palladium and copper catalysts maintain optimal contact with the substrates throughout the reflux period in tetrahydrofuran and triethylamine. Following the formation of the alkyne backbone, the thermal treatment with excess methyl iodide triggers a clean exchange reaction where the triazene unit is displaced by an iodine atom. This transformation is critical because it installs the highly reactive aryl iodide handle directly onto the rigid alkyne scaffold without compromising the integrity of other sensitive functional groups. The final step involves the reaction of these iodo-alkyne intermediates with the decaboric acid diacetonitrile complex in anhydrous toluene, where the alkyne triple bond undergoes insertion into the boron cluster to form the characteristic closo-carborane cage structure.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions associated with radical pathways or incomplete halogen-metal exchanges. The use of basic alumina for column chromatography in the final purification step further ensures the removal of any residual boron species or unreacted starting materials, resulting in products with exceptional purity profiles suitable for sensitive optoelectronic applications. The stability of the triazene intermediate allows for precise stoichiometric control, reducing the formation of homocoupling byproducts that are common in copper-catalyzed alkyne chemistry. Consequently, the overall impurity spectrum is significantly cleaner compared to routes involving cryogenic lithiation, where quenching errors can lead to complex mixtures. This level of chemical precision is essential for R&D directors focused on optimizing the performance and longevity of organic electronic devices, where trace impurities can act as quenching sites for excitons.
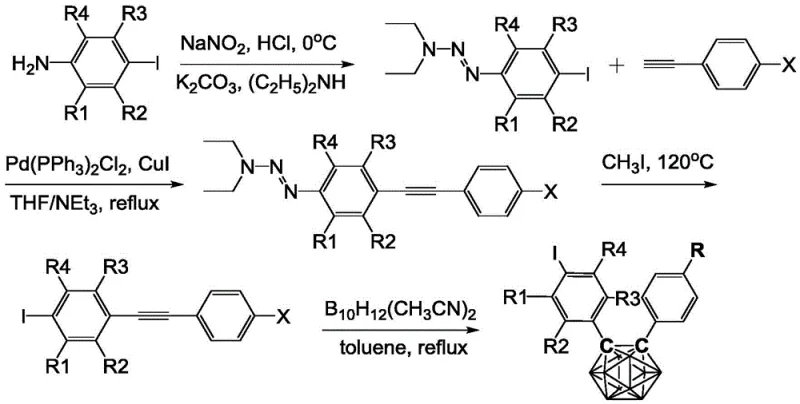
How to Synthesize 4-Iodophenyl Carborane Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing these valuable derivatives with high consistency. The process begins with the preparation of the triazene precursor via diazotization of 4-iodoaniline, followed by coupling with diethylamine, establishing the foundation for the subsequent carbon-carbon bond formation. The detailed standardized synthesis steps involve precise control of reaction temperatures, solvent ratios, and catalyst loading to maximize yield while maintaining safety standards. For organizations looking to implement this technology, adherence to the specified degassing procedures and inert atmosphere conditions is paramount to prevent catalyst deactivation. The following guide summarizes the critical operational parameters required to replicate the high yields reported in the intellectual property documentation.
- Perform Sonogashira coupling between 3,3-diethyl-1-(4-iodophenyl)-1-triazene and substituted alkynes using Pd/Cu catalysts in THF/TEA to form triazene-containing alkyne intermediates.
- Conduct iodine exchange by reacting the triazene intermediate with excess methyl iodide (CH3I) at 110-120°C in a sealed pressure vessel to generate iodo-alkyne intermediates.
- React the iodo-alkyne intermediate with decaboric acid diacetonitrile complex (B10H12(CH3CN)2) in anhydrous toluene at 100-120°C for 48 hours to form the final carborane derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this triazene-mediated synthesis route offers tangible benefits that extend beyond mere chemical novelty, directly impacting the bottom line through process simplification and risk mitigation. By eliminating the dependency on cryogenic cooling systems and hazardous organolithium reagents, the operational expenditure associated with specialized equipment maintenance and safety compliance is drastically reduced. The improved solubility of intermediates translates to higher throughput in reactor vessels, as there is no need to dilute reactions excessively to manage precipitation, thereby increasing the effective capacity of existing manufacturing infrastructure. Furthermore, the robustness of the triazene intermediates allows for potential storage and transport between synthesis stages, providing greater flexibility in production scheduling and inventory management. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of the global electronics market.
- Cost Reduction in Manufacturing: The elimination of butyllithium and the associated cryogenic infrastructure removes a significant cost center from the production budget, as these reagents are expensive and require specialized handling protocols. The thermal iodine exchange step operates at elevated temperatures using commodity chemicals like methyl iodide, which are far more cost-effective and easier to source in bulk quantities than specialty organometallics. Additionally, the higher yields achieved with iodine precursors compared to bromine analogs mean that less raw material is wasted per unit of finished product, optimizing the overall material efficiency of the process. This streamlined approach reduces the number of purification cycles required, lowering solvent consumption and waste disposal costs, which are increasingly significant regulatory burdens in the fine chemical industry.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials such as 4-iodoaniline and decaboric acid diacetonitrile complexes ensures a stable and diversified supply base, reducing the risk of bottlenecks associated with proprietary or scarce reagents. The thermal stability of the intermediates minimizes the risk of batch failures due to temperature excursions or handling errors, leading to more predictable production timelines and consistent delivery schedules. This reliability is crucial for downstream customers in the OLED and semiconductor sectors, where supply continuity is often linked to long-term contractual agreements and just-in-time manufacturing models. By adopting a synthesis route that is less sensitive to environmental variables, suppliers can offer greater assurance of product availability even during periods of market volatility.
- Scalability and Environmental Compliance: The transition from batch processes requiring extreme cold to thermal reflux conditions significantly eases the engineering challenges associated with scaling up from laboratory to pilot and commercial production. The absence of pyrophoric reagents simplifies the safety case for large-scale reactors, reducing the need for complex containment systems and emergency response measures. Moreover, the improved atom economy and reduced solvent usage align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating 4-iodophenyl carborane derivatives into their existing product portfolios or research pipelines.
Q: Why is the triazene group critical in this synthesis route?
A: The triazene group significantly improves the solubility of conjugated alkyne intermediates, which is a major bottleneck in traditional low-temperature syntheses. It also acts as a stable protecting group that facilitates efficient iodine exchange under thermal conditions, bypassing the need for harsh cryogenic reagents like butyllithium.
Q: How does the reactivity of these iodo-derivatives compare to bromo-precursors?
A: According to the patent data, the 4-iodophenylcarborane derivatives exhibit significantly higher reactivity in subsequent coupling reactions (Sonogashira and Suzuki) compared to bromo-analogs. Yields for iodine-based precursors generally exceed 80%, whereas bromo-precursors typically achieve only 40-60% under similar conditions.
Q: Is this method suitable for large-scale production?
A: Yes, the method is designed for scalability. By eliminating the need for -78°C cryogenic conditions and utilizing thermally stable triazene intermediates, the process avoids the solubility limitations and safety hazards associated with large-scale butyllithium handling, making it ideal for pilot and commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Iodophenyl Carborane Supplier
As the demand for advanced boron-cluster materials continues to surge across the pharmaceutical and electronic sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis capabilities and rigorous quality standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging deep expertise in organometallic chemistry and cluster synthesis to optimize yield and purity. We understand that the successful deployment of these materials depends on stringent purity specifications and rigorous QC labs capable of detecting trace impurities that could compromise device performance. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of the triazene-mediated route, ensuring that every batch meets the exacting standards required by global innovators.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific application needs, whether for neutron capture therapy agents or next-generation OLED emitters. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this iodine-based platform. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the superior reactivity and scalability of our 4-iodophenyl carborane derivatives before committing to large-scale procurement. Let us collaborate to accelerate the development of your high-performance materials.
