Advanced Rhodium-Catalyzed Synthesis of 2-(2-Indolyl)-Acetate Derivatives for Commercial Scale-Up
Introduction to Next-Generation Indole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct privileged scaffolds, and the indole nucleus remains one of the most ubiquitous structures in bioactive molecules. Patent CN111285846B introduces a groundbreaking synthetic methodology for producing 2-(2-indolyl)-acetate derivatives, utilizing a rhodium-catalyzed C-H bond activation strategy that fundamentally shifts the paradigm from traditional stoichiometric approaches. This innovation leverages easily prepared N-phenyl-2-aminopyridine as a starting material, enabling direct ortho-position C-H activation without the need for cumbersome pre-functionalization steps. The significance of this technology lies in its ability to generate a diverse library of indole derivatives under mild conditions, offering a robust platform for the development of new active pharmaceutical ingredients (APIs) and agrochemical intermediates. By streamlining the construction of the indole core, this method addresses critical bottlenecks in process chemistry, providing a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates.
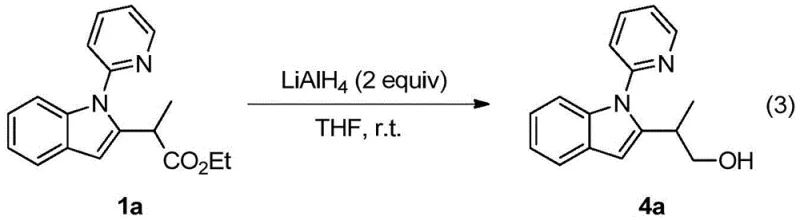
Furthermore, the versatility of the resulting products is exemplified by their capacity for downstream functionalization, such as the reduction of the ester moiety to primary alcohols, thereby expanding the chemical space accessible to medicinal chemists. This patent represents a significant leap forward in atom-economical synthesis, aligning perfectly with modern green chemistry principles while maintaining high regioselectivity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indole ring systems has relied heavily on classical named reactions such as the Fischer, Madelung, and Bischler indole syntheses, which, despite their utility, suffer from inherent limitations that hinder modern process efficiency. These traditional methods often demand harsh reaction conditions, including high temperatures and strongly acidic or basic environments, which can be detrimental to sensitive functional groups present on the substrate. Moreover, conventional routes frequently require pre-functionalized starting materials, necessitating additional synthetic steps to install leaving groups or activating moieties prior to cyclization, thereby increasing the overall step count and reducing the overall atom economy of the process. The requirement for specific substrate geometries in these older methods also limits the structural diversity of the accessible indole library, often leading to poor yields when bulky or electron-deficient substituents are present. Consequently, the reliance on these legacy technologies can result in higher production costs, increased waste generation, and longer lead times for high-purity indole building blocks, creating significant challenges for supply chain managers aiming to optimize manufacturing workflows.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a transition metal-catalyzed C-H activation strategy that bypasses the need for pre-functionalization entirely, offering a direct and streamlined route to the target indole scaffold. By employing a rhodium catalyst system, the method activates the inert ortho-C-H bond of the N-phenyl-2-aminopyridine substrate directly, facilitating a coupling reaction with allenoic acid esters to form the indole ring in a single operational step. This catalytic cycle operates under remarkably mild conditions, typically between 40°C and 60°C, which preserves the integrity of sensitive functional groups and allows for a broader substrate scope compared to thermal cyclization methods. The use of allenoates as coupling partners introduces unique structural features that are difficult to access via traditional alkyne-based annulations, thereby expanding the chemical diversity available to researchers. This modern synthetic tactic not only simplifies the operational procedure but also significantly enhances the sustainability profile of the manufacturing process by reducing waste and energy consumption.
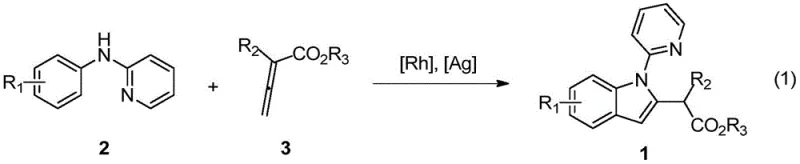
The general reaction scheme illustrates the elegance of this transformation, where simple mixing of the amine and allenoate components in the presence of the catalyst system leads directly to the desired 2-(2-indolyl)-acetate derivative with high efficiency.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The core of this technological advancement lies in the sophisticated rhodium catalytic cycle that drives the C-H bond cleavage and subsequent cyclization. The reaction initiates with the activation of the precatalyst, typically [RhCp*Cl2]2, by a silver salt additive such as silver hexafluoroantimonate (AgSbF6), which generates the active cationic rhodium(III) species in situ. This electrophilic metal center then coordinates with the nitrogen atom of the N-phenyl-2-aminopyridine directing group, positioning the metal in close proximity to the ortho-C-H bond of the phenyl ring. Through a concerted metalation-deprotonation (CMD) mechanism or electrophilic substitution pathway, the rhodium center inserts into the C-H bond, forming a stable five-membered rhodacycle intermediate. This organometallic species is the key to the reaction's regioselectivity, ensuring that functionalization occurs exclusively at the ortho-position relative to the nitrogen directing group. The subsequent coordination and insertion of the allenoic acid ester into the Rh-C bond sets the stage for the formation of the new carbon-carbon bonds required for ring closure.
Following the migratory insertion step, the catalytic cycle proceeds through a reductive elimination or protonolysis event that releases the indole product and regenerates the active rhodium species, ready to enter another turnover cycle. The presence of an oxidant, such as silver acetate (AgOAc), is crucial for maintaining the catalytic activity, particularly if the cycle involves changes in the oxidation state of the metal or requires the removal of hydride equivalents. Additionally, additives like zinc acetate play a pivotal role in facilitating the C-H activation step, potentially acting as a base to assist in the deprotonation of the C-H bond or stabilizing the transition state. The choice of solvent, specifically 1,2-dichloroethane (DCE), is optimized to solubilize both the organic substrates and the ionic catalyst components while providing a thermal environment conducive to the reaction kinetics. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, ensuring consistent quality and minimizing the formation of side products or impurities that could complicate downstream purification.
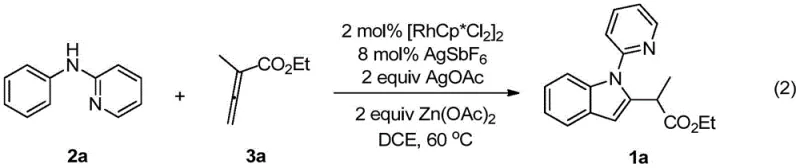
The specific example reaction conditions highlight the practical application of this mechanism, demonstrating how precise control over catalyst loading, temperature, and stoichiometry leads to reproducible high-yielding outcomes suitable for industrial replication.
How to Synthesize 2-(2-Indolyl)-Acetate Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized protocol established in the patent to ensure maximum yield and purity. The process begins with the careful weighing of N-phenyl-2-aminopyridine and the rhodium catalyst complex within an inert atmosphere glove box to prevent moisture or oxygen from deactivating the sensitive metal species. Once the reagents are charged into the reaction vessel along with the necessary silver and zinc additives, the solvent and allenoate coupling partner are introduced, and the mixture is heated to the optimal temperature range. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-value transformation.
- Weigh N-phenyl-2-aminopyridine, rhodium catalyst [RhCp*Cl2]2, silver hexafluoroantimonate, silver acetate, and zinc acetate into a reaction vessel under nitrogen atmosphere.
- Add 1,2-dichloroethane solvent and diacrylate ester, then heat the mixture to 60°C and stir for 24 hours to facilitate the cross-coupling reaction.
- Remove solvent via rotary evaporation and purify the crude residue using silica gel column chromatography with petroleum ether and ethyl acetate to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed methodology presents a compelling value proposition centered around cost efficiency and operational reliability. By eliminating the need for pre-functionalized substrates, the process removes entire synthetic steps from the supply chain, which directly translates to reduced raw material consumption and lower overall manufacturing costs. The use of commercially available and relatively inexpensive starting materials, such as N-phenyl-2-aminopyridine and simple allenoates, ensures a stable and secure supply base that is less susceptible to market volatility compared to exotic or highly specialized reagents. Furthermore, the mild reaction conditions reduce the energy burden on the manufacturing facility, as there is no requirement for extreme heating or cryogenic cooling, contributing to a lower carbon footprint and reduced utility expenses. The robustness of the reaction also implies fewer batch failures and less downtime for equipment cleaning, enhancing the overall throughput of the production line.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps significantly reduces the number of unit operations required, leading to substantial cost savings in labor, solvents, and waste disposal. Since the reaction proceeds with high atom economy, the mass intensity of the process is improved, meaning less waste is generated per kilogram of product, which lowers the environmental compliance costs associated with effluent treatment. Additionally, the high selectivity of the rhodium catalyst minimizes the formation of difficult-to-separate isomers, simplifying the purification process and reducing the loss of valuable material during chromatography or crystallization. These factors combine to create a leaner, more cost-effective manufacturing process that improves the margin profile for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals for the starting materials ensures that the supply chain is resilient against disruptions that often plague the sourcing of custom-synthesized building blocks. The simplicity of the operational procedure means that the technology can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without requiring specialized equipment or highly niche expertise. This flexibility allows for multi-sourcing strategies, reducing the risk of single-supplier dependency and ensuring continuous availability of critical intermediates for downstream drug production. The stability of the reagents and the tolerance of the reaction to various functional groups also mean that inventory management can be optimized, with raw materials having long shelf lives and minimal storage requirements.
- Scalability and Environmental Compliance: The reaction conditions described, specifically the moderate temperatures and standard solvent systems, are inherently scalable from gram-scale laboratory experiments to multi-ton commercial production without significant re-engineering. The use of 1,2-dichloroethane, while requiring proper handling, is a well-understood solvent in the industry with established recovery and recycling protocols, facilitating compliance with environmental regulations. The high yield and selectivity of the process reduce the volume of chemical waste generated, aligning with increasingly stringent global environmental standards and corporate sustainability goals. This scalability ensures that as demand for the final API grows, the supply of the intermediate can be ramped up quickly and efficiently to meet market needs without compromising on quality or safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are designed to clarify the operational benefits and technical feasibility for stakeholders evaluating this route for their own supply chains. Understanding these details is crucial for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of this Rh-catalyzed method over traditional indole synthesis?
A: Unlike traditional methods such as Fischer or Madelung synthesis which often require harsh conditions and pre-functionalized substrates, this Rh-catalyzed C-H activation approach offers superior atom economy, mild reaction temperatures (40-60°C), and eliminates the need for pre-functionalization, significantly simplifying the synthetic route.
Q: What is the optimal catalyst and oxidant system for this transformation?
A: The patent identifies [RhCp*Cl2]2 as the optimal catalyst precursor when used in conjunction with silver hexafluoroantimonate (AgSbF6) as an additive and silver acetate (AgOAc) as the oxidant. Zinc acetate is also preferred as an additive to enhance reaction efficiency and yield.
Q: Can the resulting indole derivatives be further functionalized?
A: Yes, the synthesized 2-(2-indolyl)-acetate derivatives possess versatile functional groups that allow for further chemical transformations. For instance, the ester group can be reduced to an alcohol using lithium aluminum hydride, demonstrating the utility of these compounds as flexible building blocks for complex molecule synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Indolyl)-Acetate Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modernizing the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111285846B are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2-(2-indolyl)-acetate derivatives meets the exacting standards required by global regulatory bodies. We are committed to delivering high-purity indole derivatives that empower our clients to accelerate their drug discovery and development timelines with confidence.
We invite you to engage with our technical procurement team to discuss how this cutting-edge synthesis route can be integrated into your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this catalytic method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition to a more efficient and sustainable supply chain.
