Advanced Pillar[5]arene N-Heterocyclic Carbene Catalyst for Efficient Organic Synthesis
Advanced Pillar[5]arene N-Heterocyclic Carbene Catalyst for Efficient Organic Synthesis
The landscape of modern organic synthesis is constantly evolving, driven by the demand for more sustainable, efficient, and selective catalytic systems. A significant breakthrough in this domain is documented in patent CN114751872A, which introduces a novel class of Pillar[5]arene N-heterocyclic carbene (NHC) catalysts. This innovation represents a convergence of supramolecular chemistry and organocatalysis, offering a robust solution for complex bond formations. As a leading manufacturer, we recognize the immense potential of this technology to streamline the production of high-value pharmaceutical intermediates and fine chemicals. The patent outlines a streamlined two-step synthesis that avoids the pitfalls of traditional transition metal catalysis, providing a pathway to high-purity products with reduced environmental impact.
For R&D directors and process chemists, the structural integrity of the Pillar[5]arene scaffold offers unique advantages. The rigid framework and electron-rich cavity allow for precise guest molecule complexation, enhancing catalytic selectivity. Furthermore, the ten derivatization sites on the skeleton provide ample opportunity for functional tuning, making this catalyst platform highly versatile. By integrating this advanced catalytic system into your synthetic routes, organizations can achieve superior control over reaction outcomes while adhering to increasingly stringent regulatory standards regarding metal residues in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional N-heterocyclic carbene catalysts, while powerful, often suffer from significant limitations that hinder their widespread industrial adoption. Many existing protocols require stoichiometric amounts of the catalyst or excessively high loadings to drive reactions to completion, which drastically increases raw material costs. Moreover, conventional methods frequently rely on transition metals or harsh reaction conditions that necessitate rigorous purification steps to remove toxic metal traces. These downstream processing requirements not only extend production timelines but also generate substantial chemical waste, posing challenges for environmental compliance and cost management in large-scale manufacturing facilities.
The Novel Approach
The methodology described in patent CN114751872A offers a transformative alternative by leveraging the unique properties of Pillar[5]arenes. The synthesis begins with the construction of the macrocyclic skeleton followed by a straightforward quaternization step to install the active NHC moiety. This route is characterized by high operability and mild reaction conditions, typically proceeding at room temperature or simple reflux. The resulting catalyst exhibits remarkable efficiency, functioning effectively at low dosages to promote challenging transformations like the Stetter reaction. This shift from metal-dependent to metal-free organocatalysis simplifies the workflow and enhances the overall sustainability profile of the chemical process.
![Overall synthesis route of Pillar[5]arene NHC catalyst showing two main steps](/insights/img/pillar-5-arene-nhc-catalyst-synthesis-supplier-20260305095143-02.webp)
As illustrated in the reaction scheme above, the process flows logically from simple aromatic precursors to the complex supramolecular catalyst. The use of common reagents such as paraformaldehyde and 1,4-dimethoxybenzene ensures that the supply chain remains robust and cost-effective. The final product, a bromide salt of the thiazolium-functionalized Pillar[5]arene, is obtained in good yield after standard purification, ready for immediate application in catalytic cycles without the need for specialized handling equipment.
Mechanistic Insights into Pillar[5]arene-NHC Catalyzed Stetter Reaction
The core value of this catalyst lies in its ability to facilitate Umpolung chemistry, specifically the Stetter reaction, which forms carbon-carbon bonds between aldehydes and Michael acceptors. The N-heterocyclic carbene generated in situ acts as a nucleophilic catalyst, reversing the natural polarity of the aldehyde carbon to create an acyl anion equivalent. The Pillar[5]arene backbone likely plays a crucial role in stabilizing transition states through non-covalent interactions within its cavity, potentially enhancing both the rate and stereoselectivity of the reaction. This mechanistic advantage allows for the efficient synthesis of 1,4-dicarbonyl compounds, which are vital scaffolds in the construction of heterocycles found in many bioactive molecules.
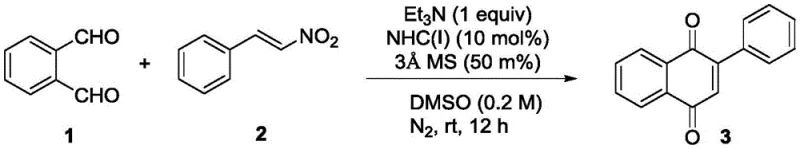
In the specific application detailed in the patent, the catalyst successfully mediates the coupling of o-phthalaldehyde and nitrostyrene to yield the corresponding naphthoquinone derivative with an impressive 88% yield. The reaction proceeds smoothly in DMSO at room temperature using triethylamine as a base, demonstrating the mildness of the protocol. For procurement and supply chain teams, this high efficiency translates directly into better atom economy and reduced solvent consumption. The ability to run such complex transformations under ambient conditions significantly lowers energy consumption compared to traditional high-temperature processes, aligning with global initiatives for greener chemical manufacturing.
How to Synthesize Pillar[5]arene NHC Catalyst Efficiently
The synthesis of this advanced catalyst is designed for reproducibility and scalability, utilizing standard laboratory glassware and commercially available reagents. The process is divided into two distinct stages: the formation of the Pillar[5]arene precursor (S-I) and the subsequent functionalization to the active catalyst (I). Detailed operational parameters, including molar ratios and solvent systems, are optimized to maximize yield and purity. For technical teams looking to implement this route, the following guide outlines the critical steps derived from the patent examples to ensure successful replication.
- Prepare Pillar[5]arene S-I by reacting 1,1-dibromo-p-phenylene diethyl ether, 1,4-dimethoxybenzene, and paraformaldehyde with BF3·OEt in DCE at room temperature.
- React the intermediate S-I with 4-methyl-5-thiazole ethanol in acetonitrile under reflux conditions overnight to form the final catalyst.
- Purify the final product using flash column chromatography with dichloromethane and methanol to obtain the high-purity catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel catalytic technology offers substantial strategic benefits for chemical manufacturers aiming to optimize their production costs and supply chain resilience. The shift towards metal-free organocatalysis eliminates the dependency on volatile precious metal markets, stabilizing raw material costs over the long term. Furthermore, the simplified purification process reduces the consumption of expensive scavenging resins and silica gel, leading to direct savings in operational expenditures. These factors combined create a more predictable and economical manufacturing environment for high-value intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metals from the catalytic cycle removes the necessity for expensive and time-consuming metal removal steps, such as treatment with activated carbon or specialized scavengers. This streamlining of the downstream process significantly reduces the overall cost of goods sold (COGS) by minimizing material usage and waste disposal fees. Additionally, the high yield of the catalyst synthesis itself ensures that the input costs for the catalyst production remain low, further enhancing the economic viability of the entire process.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including paraformaldehyde, 1,4-dimethoxybenzene, and common halogenated ethers, are commodity chemicals with stable and widespread global supply chains. This reduces the risk of production delays caused by the scarcity of exotic reagents often associated with specialized ligand synthesis. By relying on bulk-available feedstocks, manufacturers can secure long-term supply contracts and maintain consistent production schedules even during market fluctuations.
- Scalability and Environmental Compliance: The reaction conditions described, primarily involving room temperature stirring and standard reflux, are inherently safer and easier to scale than high-pressure or cryogenic processes. The use of standard solvents like dichloroethane and acetonitrile allows for established recovery and recycling protocols, minimizing environmental discharge. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals without compromising on output quality or volume.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of new catalytic systems is essential for making informed procurement and development decisions. The following questions address common inquiries regarding the performance, stability, and application scope of the Pillar[5]arene NHC catalyst. These insights are derived directly from the experimental data and technical specifications provided in the source patent documentation.
Q: What are the primary advantages of this Pillar[5]arene NHC catalyst over traditional methods?
A: Unlike traditional N-heterocyclic carbene catalysts that often require high loadings or equivalent amounts, this novel Pillar[5]arene-based catalyst operates effectively at low dosages under mild conditions. Its rigid supramolecular framework enhances stability and selectivity, while the metal-free nature eliminates the need for costly and complex heavy metal removal steps.
Q: What specific reactions can this catalyst facilitate?
A: The catalyst is specifically designed and validated for the Stetter reaction, demonstrating high efficiency in coupling o-phthalaldehyde with nitrostyrene. It achieves excellent yields (up to 88%) at room temperature, showcasing its potential for synthesizing complex 1,4-dicarbonyl compounds essential in pharmaceutical intermediates.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation method utilizes readily available starting materials like paraformaldehyde and common solvents such as 1,2-dichloroethane and acetonitrile. The reaction conditions are mild (room temperature for step 1, reflux for step 2) and the work-up involves standard filtration and chromatography, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pillar[5]arene NHC Catalyst Supplier
At NINGBO INNO PHARMCHEM, we are committed to bridging the gap between academic innovation and industrial application. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results translate into reliable manufacturing realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst meets the exacting standards required for pharmaceutical and fine chemical synthesis. Our infrastructure is designed to support the complex needs of modern organocatalysis, providing a secure foundation for your supply chain.
We invite you to explore how this advanced Pillar[5]arene technology can enhance your current synthetic routes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to help you evaluate the potential integration of this high-performance catalyst into your portfolio, ensuring a partnership built on transparency, quality, and mutual growth.
