Advanced Synthesis of 1-Hydroxycyclopropanecarboxylic Acid for Commercial Scale-Up
Advanced Synthesis of 1-Hydroxycyclopropanecarboxylic Acid for Commercial Scale-Up
The development of efficient synthetic routes for cyclopropane-containing building blocks remains a critical challenge in modern medicinal chemistry, particularly for the production of enzyme inhibitors and bioactive peptides. Patent CN110862311A introduces a groundbreaking methodology for the synthesis of 1-hydroxycyclopropanecarboxylic acid and its esters, addressing long-standing issues regarding safety, yield, and scalability. This novel approach leverages a diazotization strategy starting from readily available 1-aminocyclopropyl methyl formate, bypassing the hazardous reagents and complex multi-step sequences associated with traditional methodologies. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process chemistry, offering a pathway to high-purity products with drastically reduced environmental impact and operational risk.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-hydroxycyclopropanecarboxylic acid derivatives has relied on cumbersome and dangerous chemical transformations that pose severe risks for industrial scale-up. As illustrated in the reaction scheme below, conventional routes often involve the use of metallic sodium as a strong base and liquid bromine as an oxidant to facilitate ring contraction from four-membered precursors. These reagents are classified as controlled chemicals due to their extreme reactivity; metallic sodium is pyrophoric and requires stringent handling protocols, while liquid bromine is highly corrosive and toxic. Furthermore, the traditional three-step sequence typically results in abysmal total yields ranging from only 10% to 20%, generating substantial volumes of waste solvent and wastewater that complicate downstream purification and environmental compliance.
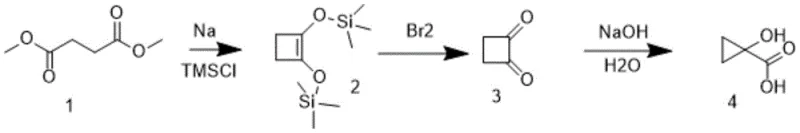
The Novel Approach
In stark contrast, the innovative process disclosed in the patent utilizes a direct diazotization of 1-aminocyclopropyl methyl formate, followed by a mild hydrolysis step, to achieve the target molecule in just two steps. This strategy eliminates the need for hazardous halogenation and metal-mediated ring contractions, replacing them with aqueous acid chemistry and standard base hydrolysis. By optimizing reaction parameters such as acid concentration and temperature control, the inventors have achieved a total yield of 60-70%, representing a three-to-four-fold increase in efficiency compared to prior art. This shift not only enhances the economic viability of the process but also aligns with green chemistry principles by reducing the E-factor and simplifying the post-treatment workflow to basic extraction and concentration.
Mechanistic Insights into Diazotization and Hydrolysis
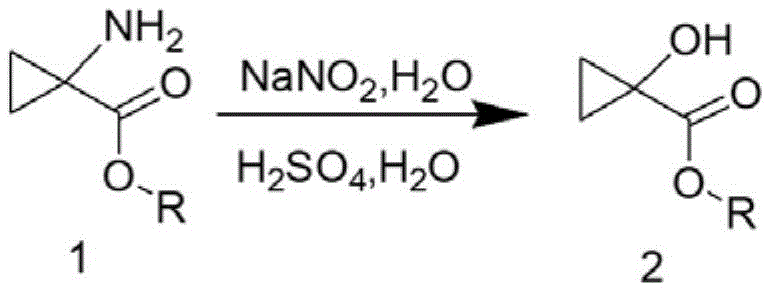
The core of this synthetic breakthrough lies in the precise control of the diazotization reaction, where the primary amine group is converted into a diazonium species which subsequently undergoes nucleophilic substitution by water to form the hydroxyl group. The first step involves dissolving the amino-ester in a specific concentration of sulfuric acid and reacting it with sodium nitrite at low temperatures (0-5°C) to generate the diazonium intermediate safely. Crucially, the patent specifies that sulfuric acid must be used instead of hydrochloric or nitric acid, as the latter can promote the unwanted opening of the strained cyclopropane ring, leading to linear byproducts. The reaction mixture is then carefully added dropwise into a refluxing sulfuric acid solution, a technique designed to minimize the residence time of the reactive intermediate in the system, thereby preventing thermal degradation and ensuring high conversion to the 1-hydroxycyclopropyl formate.
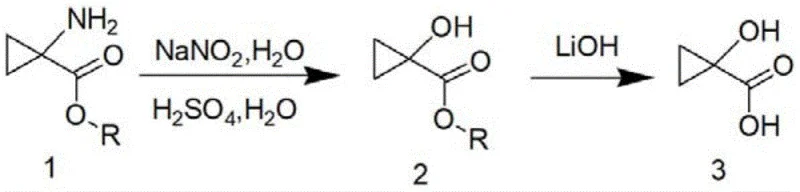
Following the formation of the hydroxy ester, the second stage involves the removal of the ester protecting group to yield the free carboxylic acid. This step requires careful selection of the base to avoid ring-opening side reactions that are common with highly strained cyclopropane systems under strongly basic conditions. The patent recommends using lithium hydroxide, sodium hydroxide, or potassium hydroxide in a mixed solvent system of tetrahydrofuran and water at moderate temperatures (20-40°C). This mild hydrolysis condition ensures that the cyclopropane ring remains intact while efficiently cleaving the ester bond. The final product is isolated through a straightforward workup involving pH adjustment, extraction with ethyl acetate, and drying, resulting in a high-purity white solid suitable for direct use in subsequent coupling reactions for API synthesis.
How to Synthesize 1-Hydroxycyclopropanecarboxylic Acid Efficiently
Implementing this synthesis route requires strict adherence to the optimized stoichiometry and temperature profiles outlined in the patent to maximize yield and safety. The process begins with the preparation of specific sulfuric acid solutions to manage the exothermic nature of the diazotization, followed by a controlled hydrolysis step that balances reaction rate with structural integrity. Operators must ensure that the molar ratios of reagents, particularly the slight excess of sodium nitrite and the precise acid equivalents, are maintained to drive the reaction to completion without generating excessive nitrous acid byproducts. The detailed standardized synthetic steps, including specific solvent volumes and workup procedures, are provided in the guide below to facilitate technology transfer and pilot plant trials.
- Dissolve 1-aminocyclopropyl methyl formate in aqueous sulfuric acid and react with sodium nitrite at controlled temperatures to form the hydroxy ester intermediate.
- Dropwise add the reaction mixture into refluxing sulfuric acid to minimize residence time and prevent cyclopropane ring opening.
- Hydrolyze the resulting ester using lithium hydroxide in a THF-water mixture to obtain the final 1-hydroxycyclopropanecarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound advantages in terms of cost stability and operational reliability. By eliminating the dependency on volatile and tightly regulated raw materials like liquid bromine and metallic sodium, manufacturers can significantly reduce the costs associated with hazardous material storage, specialized containment infrastructure, and waste disposal. The simplified two-step process also shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. Furthermore, the use of commodity chemicals such as sulfuric acid and sodium nitrite ensures a robust and resilient supply chain that is less susceptible to the geopolitical and logistical disruptions often seen with specialty reagents.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents directly translates to lower raw material costs and reduced overhead for safety compliance. The drastic improvement in yield from roughly 10% to over 60% means that significantly less starting material is required to produce the same amount of final product, effectively lowering the cost per kilogram. Additionally, the simplified workup procedure reduces solvent consumption and energy usage during concentration and purification, contributing to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Sourcing 1-aminocyclopropyl methyl formate is far more straightforward than managing the logistics of controlled substances like liquid bromine. This accessibility ensures a continuous supply of raw materials, minimizing the risk of production stoppages due to regulatory delays or supplier shortages. The mild reaction conditions also allow the process to be executed in a wider range of standard chemical reactors, increasing the number of qualified contract manufacturing organizations (CMOs) capable of producing this intermediate.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing aqueous media and avoiding the generation of heavy metal waste or halogenated byproducts. This aligns perfectly with increasingly stringent global environmental regulations, reducing the burden on wastewater treatment facilities and lowering the carbon footprint of the manufacturing process. The ability to run the reaction at near-ambient pressures and moderate temperatures further enhances the safety profile, making it an ideal candidate for multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific pipeline projects and for procurement teams assessing the long-term viability of the supply source.
Q: Why is sulfuric acid preferred over hydrochloric acid in this synthesis?
A: Sulfuric acid is critical because hydrochloric or nitric acid can induce the opening of the strained cyclopropane ring, leading to significant impurity formation and reduced yield.
Q: What is the yield advantage of this new method compared to conventional routes?
A: This optimized diazotization route achieves a total yield of 60-70%, which is a substantial improvement over the 10-20% yield typical of older methods involving metallic sodium and bromine.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (0-30°C), avoids hazardous reagents like liquid bromine, and employs simple extraction workups, making it highly scalable and safe for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Hydroxycyclopropanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality cyclopropane intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering 1-hydroxycyclopropanecarboxylic acid with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay every batch. Our facility is equipped to handle the specific safety requirements of diazotization chemistry, guaranteeing a safe and compliant production environment for our partners.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their drug discovery programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, helping you optimize your budget without compromising on quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your project timelines and enhance your supply chain resilience.
