Advanced Palladium-Catalyzed Synthesis of Fluoroalkyl Pyrrolo[1,2-a]indoles for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those containing fluorine atoms which often enhance metabolic stability and bioavailability. Patent CN108069977B introduces a groundbreaking synthetic methodology for producing fluoroalkyl-substituted pyrrole[1,2-a]indoles, a privileged structural motif found in numerous bioactive natural products and drug candidates. This innovation represents a significant leap forward from traditional multi-step syntheses by employing a transition metal-catalyzed radical tandem cyclization strategy. By utilizing readily available N-3-butene indoles and inexpensive fluorine-containing halides as starting materials, this process achieves the construction of complex polycyclic skeletons in a single operational step. The method operates under relatively mild thermal conditions without the need for specialized additives or extreme temperatures, marking a substantial improvement in process greenness and economic efficiency for the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole[1,2-a]indole frameworks has relied heavily on classical organic transformations that present significant challenges for industrial application. Traditional routes often involve nucleophilic additions using highly reactive and hazardous lithium reagents, which require strict anhydrous conditions and cryogenic temperatures to control selectivity and prevent side reactions. Furthermore, many existing protocols depend on Lewis acid catalysis or thermal rearrangements that suffer from poor atom economy, generating substantial amounts of chemical waste and requiring complex purification sequences to remove toxic metal residues. These conventional methods frequently necessitate pre-functionalized starting materials, adding extra synthetic steps that increase both the overall cost and the lead time for material delivery. The harsh reaction environments associated with these older technologies also pose safety risks and limit the scope of functional groups that can be tolerated, thereby restricting the diversity of derivatives that can be practically accessed for drug discovery programs.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108069977B offers a streamlined, one-pot solution that dramatically simplifies the manufacturing landscape. This novel approach leverages a palladium-catalyzed radical cascade reaction to directly couple N-3-butene indoles with fluorinated halides, effectively building the target heterocyclic core and installing the fluoroalkyl group simultaneously. The reaction proceeds efficiently at moderate temperatures ranging from 50°C to 100°C, eliminating the energy-intensive requirements for deep cooling or high-pressure equipment. By employing simple and commercially available ligands and bases, the process minimizes the complexity of the reaction mixture, facilitating easier downstream processing. This strategic shift not only enhances the overall yield and purity of the final product but also aligns perfectly with modern principles of green chemistry by reducing solvent usage and waste generation, making it an ideal candidate for cost reduction in API manufacturing.
![General reaction scheme showing the synthesis of fluoroalkyl-substituted pyrrole[1,2-a]indoles from N-3-butene indole and RfX](/insights/img/fluoroalkyl-pyrroloindole-synthesis-pharma-supplier-20260302203207-01.png)
Mechanistic Insights into Pd-Catalyzed Radical Tandem Cyclization
The core of this technological advancement lies in the sophisticated yet robust mechanism of the palladium-catalyzed radical tandem cyclization. The reaction initiates with the oxidative addition of the fluorine-containing halide (RfX) to the low-valent palladium catalyst, generating a reactive fluoroalkyl-palladium species. This intermediate subsequently undergoes homolytic cleavage or single-electron transfer processes to release a fluoroalkyl radical, which then adds regioselectively to the terminal alkene of the N-3-butene indole substrate. The resulting carbon-centered radical is positioned perfectly for an intramolecular cyclization onto the electron-rich indole ring, forming the new C-C bond that closes the pyrrole ring system. This cascade sequence is highly efficient because it avoids the isolation of unstable intermediates, driving the reaction forward through thermodynamic stability of the aromatic product. The choice of ligand plays a critical role in stabilizing the palladium center and modulating its redox potential, ensuring that the catalytic cycle turns over rapidly without deactivating into palladium black.
From an impurity control perspective, this mechanism offers distinct advantages over ionic pathways. Because the reaction proceeds via radical intermediates rather than highly basic or acidic ionic species, there is significantly less risk of hydrolysis or degradation of sensitive functional groups present on the substrate. The mild reaction conditions prevent the formation of polymeric by-products that often plague high-temperature thermal rearrangements. Furthermore, the use of stoichiometric inorganic bases like sodium carbonate helps to neutralize any acidic by-products generated during the cycle without promoting unwanted elimination reactions. This high level of chemoselectivity ensures that the crude reaction mixture contains fewer structurally related impurities, simplifying the purification burden and allowing for the production of high-purity fluoroalkyl substituted pyrrole[1,2-a]indole suitable for stringent pharmaceutical applications without extensive recrystallization steps.
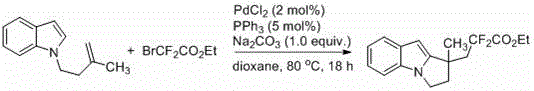
How to Synthesize Fluoroalkyl Pyrrolo[1,2-a]indoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and atmospheric control to maximize the efficiency of the palladium catalyst. The protocol is designed to be operationally simple, typically involving the charging of all reagents into a single vessel under an inert nitrogen atmosphere to prevent oxidation of the catalyst or radical quenching by oxygen. The reaction mixture is then heated to the specified temperature range and monitored until conversion is complete, after which the product can be isolated through standard filtration and purification techniques. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining N-3-butene indole, fluorine-containing halide (RfX), transition metal catalyst (e.g., PdCl2), ligand (e.g., PPh3), base, and organic solvent under inert atmosphere.
- Heat the reaction mixture to a temperature between 50°C and 100°C and maintain stirring for 16 to 24 hours to facilitate the radical tandem cyclization.
- Upon completion, filter the reaction mixture, dry the resulting solid, and purify the crude product via column chromatography to obtain the target fluoroalkyl-substituted pyrrole[1,2-a]indole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthetic route translates into tangible strategic benefits that extend far beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the supply chain for raw materials; instead of sourcing expensive, pre-functionalized building blocks or hazardous organometallic reagents, manufacturers can rely on commodity chemicals like N-3-butene indoles and industrial-grade fluorinated halides. This shift significantly reduces the vulnerability of the supply chain to fluctuations in the availability of niche reagents and lowers the overall inventory carrying costs. Additionally, the elimination of cryogenic cooling requirements means that the process can be run in standard glass-lined reactors without the need for specialized low-temperature infrastructure, thereby reducing capital expenditure and energy consumption during production runs.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the use of earth-abundant palladium catalysts at low loadings and inexpensive phosphine ligands. By removing the need for stoichiometric amounts of costly lithium reagents or exotic Lewis acids, the direct material cost per kilogram of product is substantially decreased. Furthermore, the simplified workup procedure, which often involves mere filtration and drying followed by standard chromatography, reduces the labor hours and solvent volumes required for purification. This streamlined workflow minimizes waste disposal costs and shortens the batch cycle time, allowing for higher throughput and better utilization of manufacturing assets without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures a more reliable supply of critical intermediates for downstream drug synthesis. Since the reaction tolerates a wide range of substituents and functional groups, it provides a flexible platform for generating diverse libraries of analogues without needing to re-optimize the entire process for each new derivative. This flexibility allows suppliers to respond quickly to changing R&D demands and scale up production of specific candidates as they advance through clinical trials. The use of stable, non-hazardous solvents like 1,4-dioxane or toluene further simplifies logistics and storage requirements, reducing the regulatory burden associated with transporting dangerous goods and ensuring continuous availability of materials for global partners.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage quantities is straightforward due to the absence of exothermic hazards associated with rapid lithiation or high-pressure hydrogenation steps. The mild thermal profile allows for safe heat management in large reactors, mitigating the risk of thermal runaway incidents. From an environmental standpoint, the high atom economy of the tandem cyclization means less chemical waste is generated per unit of product, facilitating compliance with increasingly strict environmental regulations. The ability to recycle the solvent and potentially recover the palladium catalyst adds another layer of sustainability, appealing to corporate social responsibility goals and reducing the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluoroalkylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production pipelines or for procurement specialists assessing the long-term viability of the supply source.
Q: What are the primary advantages of this Pd-catalyzed method over traditional lithiation routes?
A: This novel method eliminates the need for harsh conditions such as cryogenic temperatures or strong lithium reagents. It utilizes cheap, industrially available raw materials and operates under mild thermal conditions (50-100°C), significantly improving safety and atom economy while reducing waste generation.
Q: Which catalysts and ligands are compatible with this synthesis protocol?
A: The process is robust and compatible with various palladium sources including palladium acetate, palladium dichloride, and tetrakis(triphenylphosphine)palladium. Effective ligands include triphenylphosphine, 2,2'-bipyridine, and 1,10-phenanthroline, allowing for flexibility in optimizing cost and performance.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the protocol is highly scalable due to its one-pot nature and simple workup procedure involving filtration and standard purification. The use of common organic solvents like 1,4-dioxane and stable reagents ensures that the process can be safely transferred from laboratory to pilot and commercial plant scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl-substituted Pyrrole[1,2-a]indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality heterocyclic intermediates for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous QC labs that enforce stringent purity specifications to guarantee the integrity of every batch we deliver. We are committed to supporting your R&D efforts by providing materials that meet the highest industry standards for purity and consistency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects, ensuring a seamless transition from laboratory discovery to commercial success.
