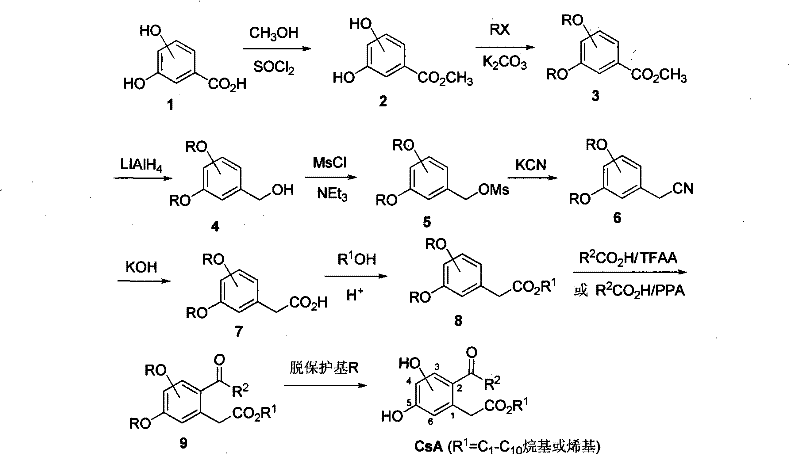
Scalable Synthesis of Bishydroxy-2-acylphenylacetate Intermediates for Commercial Drug Development
The pharmaceutical industry continuously seeks robust synthetic pathways for novel antitumor agents, and patent CN101402573B presents a significant advancement in the preparation of bishydroxy-2-acylphenylacetate derivatives. This intellectual property discloses a comprehensive chemical strategy to synthesize compounds that were previously only accessible through limited natural extraction from fungal metabolites. By establishing a fully synthetic route, the technology addresses critical supply chain vulnerabilities associated with biologically sourced active pharmaceutical ingredients. The methodology leverages standard organic transformations such as esterification, protection, reduction, and acylation to construct the complex molecular architecture required for high-potency drug candidates. For R&D directors and procurement specialists, this patent represents a viable pathway to secure reliable access to these specialized pharmaceutical intermediates without relying on unpredictable biological fermentation processes. The ability to manufacture these structures synthetically ensures batch-to-batch consistency and facilitates the rigorous quality control standards demanded by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, compounds like Cytosporone B and its analogues were isolated exclusively from the metabolites of endophytic fungi found in specific mangrove ecosystems. This reliance on natural sources introduces severe constraints regarding scalability, reproducibility, and environmental sustainability. The extraction yield from fungal cultures is inherently low, making it economically unfeasible to produce the quantities required for clinical trials or commercial drug manufacturing. Furthermore, biological variability leads to inconsistent impurity profiles, complicating the purification process and risking regulatory non-compliance. Seasonal changes and ecological factors can disrupt the supply of the raw fungal biomass, creating unacceptable risks for supply chain heads who require continuous production lines. The inability to structurally modify the natural product easily also limits the scope for structure-activity relationship studies, hindering the optimization of therapeutic efficacy. Consequently, the pharmaceutical industry has long needed a chemical synthesis solution to overcome these bottlenecks and enable the widespread application of these promising antitumor agents.
The Novel Approach
The synthetic methodology outlined in the patent data offers a transformative solution by utilizing readily available chemical starting materials such as dihydroxybenzoic acid. This approach decouples production from biological constraints, allowing for precise control over reaction conditions and stoichiometry. The route is designed to be modular, enabling the introduction of diverse alkyl and alkenyl chains at specific positions on the benzene ring to tailor biological activity. By employing common reagents like thionyl chloride, lithium aluminum hydride, and potassium cyanide, the process remains compatible with existing infrastructure in fine chemical manufacturing plants. The strategic use of protecting groups ensures that reactive hydroxyl functionalities are preserved during harsh transformation steps, thereby maximizing overall yield and minimizing side reactions. This chemical precision translates directly into commercial advantages, including reduced waste generation and simplified downstream processing. The result is a scalable, cost-effective manufacturing process that can support the demands of modern drug development pipelines.
Mechanistic Insights into TFAA-Mediated Acylation and Deprotection
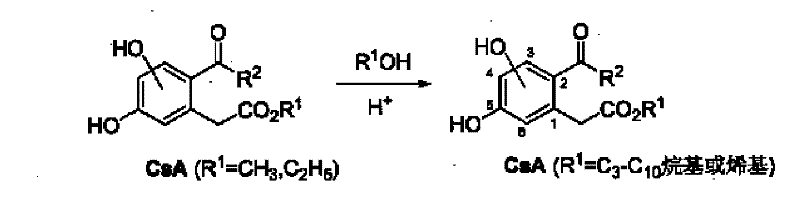
A critical component of this synthesis is the acylation step, where the phenylacetate intermediate reacts with carboxylic acids under the influence of trifluoroacetic anhydride or polyphosphoric acid. This activation mechanism generates a highly reactive acylating species in situ, facilitating the electrophilic aromatic substitution necessary to install the acyl group at the ortho position relative to the ester side chain. The choice of activator allows for flexibility in processing; trifluoroacetic anhydride offers mild conditions suitable for sensitive substrates, while polyphosphoric acid provides a robust alternative for larger scale operations. Following acylation, the removal of protecting groups is executed with high selectivity. For benzyl-protected intermediates, catalytic hydrogenolysis using palladium on carbon effectively cleaves the ether bonds without affecting the ester or ketone functionalities. Alternatively, Lewis acids like aluminum chloride can be employed for methyl ether deprotection. This dual deprotection strategy provides process chemists with options to optimize based on equipment availability and safety protocols, ensuring that the final deprotection step does not become a bottleneck in the overall production schedule.
Controlling the impurity profile during these mechanistic steps is paramount for meeting the stringent requirements of pharmaceutical intermediates. The use of column chromatography at multiple stages, as described in the patent examples, serves to remove unreacted starting materials and by-products such as over-acylated species or isomeric impurities. The hydrolysis of the nitrile intermediate to the corresponding carboxylic acid is another critical control point, where pH regulation ensures complete conversion while preventing ester hydrolysis. By maintaining strict control over reaction temperatures and molar ratios, particularly during the mesylation and cyanation steps, the formation of toxic by-products is minimized. This attention to detail in the reaction mechanism directly supports the goal of producing high-purity intermediates suitable for subsequent coupling reactions in drug synthesis. The robustness of these mechanistic steps ensures that the process can be transferred from laboratory scale to pilot plant operations with minimal deviation in product quality.
How to Synthesize Bishydroxy-2-acylphenylacetate Efficiently
Implementing this synthesis requires a systematic approach to unit operations, beginning with the preparation of the protected benzoic acid ester. The process flows through reduction and chain extension before culminating in the final acylation and deprotection sequence. Operators must adhere to strict anhydrous conditions during the reduction and mesylation steps to prevent reagent degradation and ensure high yields. The subsequent hydrolysis and esterification steps require careful pH monitoring to drive equilibrium towards the desired product. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-value pathway.
- Esterification of dihydroxybenzoic acid followed by hydroxyl protection using alkyl halides.
- Reduction of the ester group to alcohol, followed by mesylation and cyanation to extend the carbon chain.
- Hydrolysis of nitrile to acid, esterification, acylation with carboxylic acids, and final deprotection to yield the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial benefits by utilizing commodity chemicals that are widely available in the global market. Unlike specialized biological extracts, reagents such as methanol, thionyl chloride, and common carboxylic acids are sourced from established supply chains with stable pricing structures. This reduces the risk of raw material shortages and protects against volatile cost fluctuations that often plague niche natural product sourcing. The elimination of fermentation tanks and biological containment requirements further simplifies the facility setup, lowering capital expenditure for manufacturing partners. Additionally, the synthetic nature of the process allows for continuous improvement and optimization, leading to gradual cost reductions over the lifecycle of the product. For supply chain managers, the predictability of chemical synthesis timelines ensures more accurate forecasting and inventory management, reducing the need for excessive safety stock.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive extraction solvents and large-scale fermentation infrastructure, significantly lowering the overhead costs associated with production. By using catalytic amounts of acids and recoverable catalysts like palladium on carbon, the consumption of high-value reagents is minimized. The high yields reported in the patent examples indicate efficient atom economy, which reduces the volume of waste disposal and associated environmental compliance costs. Furthermore, the ability to synthesize various analogues from a common intermediate allows for economies of scale, spreading fixed costs across multiple product variants. These factors combine to create a highly competitive cost structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Synthetic manufacturing is not subject to the seasonal or ecological variables that affect natural sourcing, guaranteeing a consistent year-round supply. The use of standard chemical equipment means that production can be easily shifted between different qualified manufacturing sites if necessary, enhancing supply chain resilience. The stability of the intermediates allows for stockpiling at strategic points in the synthesis, providing a buffer against unexpected demand surges. This reliability is crucial for maintaining uninterrupted clinical trial supplies and commercial drug production schedules. Partners can rely on predictable lead times and consistent quality, fostering long-term strategic relationships.
- Scalability and Environmental Compliance: The reaction conditions described, such as reflux temperatures and atmospheric pressure hydrogenation, are inherently scalable from gram to tonne quantities without requiring exotic high-pressure equipment. Waste streams are primarily organic solvents and salts, which can be managed through standard distillation and treatment protocols familiar to fine chemical plants. The avoidance of heavy metal catalysts in the main chain construction, reserving them only for the final deprotection where recovery is feasible, aligns with green chemistry principles. This simplifies the regulatory approval process for the manufacturing site and reduces the environmental footprint of the operation. Scalability ensures that the technology can grow with the commercial success of the downstream drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bishydroxy-2-acylphenylacetate intermediates. The answers are derived from the specific technical disclosures within the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into their existing portfolios.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: This synthetic method overcomes the limited availability and seasonal dependency associated with isolating compounds from fungal metabolites, ensuring consistent supply and quality control.
Q: Is the acylation step compatible with various carboxylic acid substrates?
A: Yes, the process utilizes trifluoroacetic anhydride or polyphosphoric acid to facilitate acylation with a wide range of aliphatic carboxylic acids, allowing for structural diversity.
Q: How is the purity of the final intermediate ensured during scale-up?
A: The route incorporates multiple purification stages, including column chromatography and recrystallization, along with controlled deprotection steps to minimize impurities and ensure high purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bishydroxy-2-acylphenylacetate Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to support your drug development initiatives. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from research to market. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the complex impurity profiles associated with these intermediates. We understand the critical nature of antitumor drug supply chains and are committed to delivering materials that meet the highest international standards. Our technical team is proficient in handling the specific reagents and conditions required for this synthesis, including moisture-sensitive reductions and catalytic hydrogenations.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain insights into how optimizing this route can benefit your overall project budget. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify our capabilities. Let us collaborate to bring these promising antitumor candidates to patients faster and more efficiently through our shared commitment to chemical excellence and supply chain integrity.
