Advanced Organocatalytic Route for Chiral Benzoaromatic Heterocyclic Dihydropyranones Manufacturing
Advanced Organocatalytic Route for Chiral Benzoaromatic Heterocyclic Dihydropyranones Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex chiral scaffolds with high precision. Patent CN113801137A introduces a groundbreaking approach for the synthesis of chiral benzo-aromatic heterocyclic dihydropyrone compounds, a structural motif prevalent in numerous bioactive natural products and drug candidates. This technology leverages a tandem [4+2] Michael/ester exchange reaction to efficiently build hexahydro lactone skeletons featuring two contiguous stereocenters. By utilizing mild reaction conditions and highly selective organocatalysts, this method addresses critical challenges in asymmetric synthesis, providing a reliable pathway for generating high-purity pharmaceutical intermediates. The ability to access these complex architectures with such fidelity opens new avenues for lead compound optimization and drug discovery programs globally.
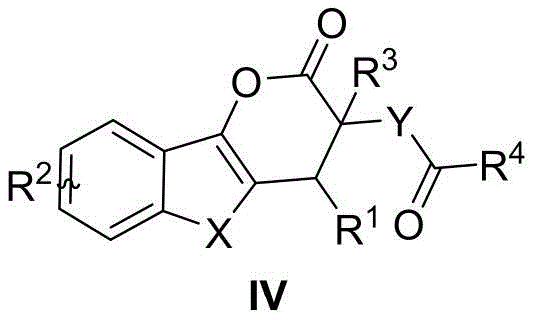
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of chiral benzo-aromatic heterocyclic dihydropyrones has relied on methods that often suffer from significant drawbacks regarding efficiency and environmental impact. Conventional routes frequently necessitate the use of stoichiometric chiral auxiliaries or expensive transition metal catalysts, which introduce complications in downstream processing due to the need for rigorous metal removal to meet regulatory standards. Furthermore, many existing protocols operate under harsh conditions that can compromise sensitive functional groups, leading to lower yields and difficult purification processes. The lack of general methods capable of simultaneously controlling multiple stereocenters with high diastereo- and enantioselectivity has historically limited the rapid exploration of chemical space for this important class of compounds, creating a bottleneck in the development of novel therapeutic agents.
The Novel Approach
In stark contrast to traditional limitations, the methodology disclosed in CN113801137A utilizes a sophisticated organocatalytic cascade reaction that streamlines the synthesis process while maximizing stereochemical control. This novel approach employs readily available benzo-aromatic heterocyclic 2-alkene-3-ketones reacting with butenolides or azlactones in the presence of a chiral bifunctional catalyst. The reaction proceeds smoothly at temperatures ranging from 0°C to 60°C, eliminating the need for extreme thermal conditions. As illustrated in the general reaction scheme below, this tandem process constructs the target dihydropyrone framework in a single operational step, significantly reducing the number of synthetic operations required compared to stepwise conventional methods.
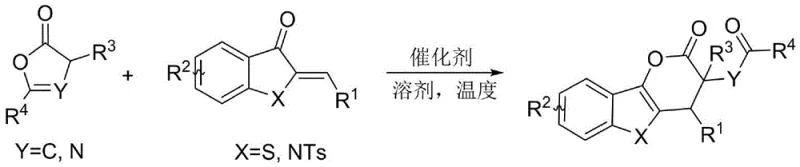
Mechanistic Insights into Bifunctional Squaramide Catalysis
The success of this transformation lies in the precise mechanistic action of the chiral catalyst, which acts as a molecular template to organize the reactants in a specific spatial arrangement. The catalyst, typically a cinchona alkaloid-derived squaramide, functions through a dual activation mode where it simultaneously activates both the nucleophile and the electrophile via hydrogen bonding networks. This bifunctional activation lowers the energy barrier for the initial Michael addition while rigidly defining the trajectory of the attack, thereby ensuring high enantioselectivity. Following the Michael addition, an intramolecular ester exchange or cyclization occurs rapidly to lock in the stereochemistry, resulting in the formation of the stable dihydropyrone ring system with two contiguous chiral centers. The structural diversity of the catalysts, as shown in the figure below, allows for fine-tuning of the steric environment to accommodate various substrate combinations effectively.
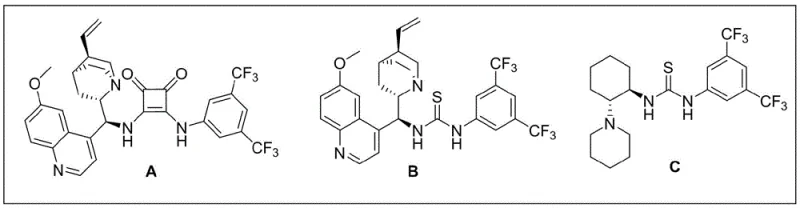
Impurity control is inherently managed by the high selectivity of the organocatalytic system, which minimizes the formation of side products commonly associated with non-selective background reactions. The mild nature of the reaction conditions prevents the degradation of sensitive functional groups on the aromatic rings or the lactone moiety, ensuring a clean reaction profile. Moreover, the use of organocatalysts avoids the introduction of heavy metal contaminants, a critical advantage for pharmaceutical manufacturing where residual metal limits are strictly enforced. The robustness of the catalytic cycle ensures that the reaction maintains high fidelity even on larger scales, making it an attractive option for industrial applications where consistency and purity are paramount concerns for regulatory compliance.
How to Synthesize Chiral Benzoaromatic Heterocyclic Dihydropyranones Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to specific protocols regarding solvent selection and catalyst loading to achieve optimal results. The process begins with the dissolution of the starting materials in an appropriate organic solvent, followed by the addition of the chiral catalyst under controlled atmospheric conditions. Detailed standard operating procedures regarding mixing rates, temperature ramping, and work-up techniques are essential to replicate the high yields and selectivity reported in the patent literature. For a comprehensive guide on the standardized synthesis steps, please refer to the section below.
- Dissolve benzo-aromatic heterocyclic 2-alkene-3-ketone and butenolide or azlactone substrates in an organic solvent such as toluene or dichloromethane.
- Add a chiral bifunctional catalyst, preferably a cinchona alkaloid-squaramide derivative, at a loading of approximately 20 mol%.
- Stir the reaction mixture at temperatures between 0°C and 60°C for 12 to 72 hours, then purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this organocatalytic technology offers substantial benefits that directly address the pain points of modern supply chain management and cost engineering in the fine chemical sector. The shift towards metal-free synthesis significantly simplifies the purification workflow, as there is no need for expensive scavenging resins or complex extraction protocols to remove trace metals. This reduction in processing steps translates to shorter production cycles and lower operational expenditures, enhancing the overall economic viability of producing these high-value intermediates. Additionally, the mild reaction conditions contribute to improved safety profiles and reduced energy consumption, aligning with global sustainability goals and green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a major cost driver associated with traditional asymmetric synthesis, while the high atom economy of the cascade reaction minimizes waste generation. By avoiding the use of stoichiometric chiral reagents, the material costs are drastically lowered, allowing for more competitive pricing structures in the final API supply chain. The simplified downstream processing further reduces solvent usage and labor hours, contributing to a leaner and more cost-effective manufacturing model that maximizes return on investment for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable organocatalysts ensures a consistent supply of critical reagents, mitigating the risks associated with the volatility of the precious metal market. The broad substrate scope of this methodology means that a single platform technology can be adapted to produce a wide variety of analogues, providing flexibility to respond quickly to changing project requirements or clinical trial demands. This versatility strengthens the supply chain resilience, ensuring that production timelines are met without the delays often caused by specialized reagent sourcing or complex catalyst preparation.
- Scalability and Environmental Compliance: The robust nature of the reaction conditions facilitates seamless scale-up from gram to kilogram and tonne quantities without significant loss in performance or selectivity. The absence of toxic heavy metals simplifies waste treatment and disposal procedures, ensuring full compliance with stringent environmental regulations across different jurisdictions. This eco-friendly profile not only reduces the environmental footprint of the manufacturing process but also enhances the corporate social responsibility standing of the supply chain partners involved in the production of these critical pharmaceutical building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and capabilities of this synthesis technology, based on the detailed experimental data provided in the patent documentation. Understanding these nuances is crucial for R&D teams evaluating this route for their specific pipeline projects. The answers reflect the proven performance metrics and operational parameters established during the development of this novel methodology.
Q: What level of stereoselectivity can be achieved with this synthesis method?
A: The patented method demonstrates exceptional stereocontrol, achieving diastereomeric ratios (dr) greater than 20:1 and enantiomeric excess (ee) values up to 99% across various substrates.
Q: What type of catalysts are utilized in this asymmetric transformation?
A: The process employs bifunctional organocatalysts, specifically cinchona alkaloid-derived squaramides, which facilitate dual activation of the substrates through hydrogen bonding interactions.
Q: Does this method support a broad range of substrate variations?
A: Yes, the protocol exhibits excellent substrate universality, successfully accommodating various substituted benzothiophenes and indoles, as well as different butenolide and azlactone derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Benzoaromatic Heterocyclic Dihydropyranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in accelerating the development of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of chiral benzoaromatic heterocyclic dihydropyranones meets the highest industry standards for potency and optical purity.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this metal-free methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a successful and efficient partnership.
