Advanced Asymmetric Synthesis of Trifluoromethyl Spiro Oxindoles for Pharmaceutical Innovation
The pharmaceutical industry is constantly seeking novel scaffolds that can offer improved metabolic stability and biological activity, and the patent CN113173931A addresses this critical need by disclosing a new class of trifluoromethyl-containing tetrahydrobenzothiophene pyrrole spiro oxoindole compounds. These complex molecules are characterized by a unique structural framework that integrates a spiro-oxindole core with a tetrahydrobenzothiophene moiety, further functionalized with a trifluoromethyl group. The presence of the trifluoromethyl group is particularly strategic in modern medicinal chemistry, as it is known to enhance the lipophilicity and metabolic resistance of drug candidates without significantly increasing steric bulk. This patent represents a significant breakthrough in organic synthesis, providing a robust method to access these highly functionalized architectures which serve as valuable building blocks for new drug discovery programs. The compounds possess four continuous chiral centers, presenting a formidable synthetic challenge that this new methodology overcomes with remarkable efficiency. 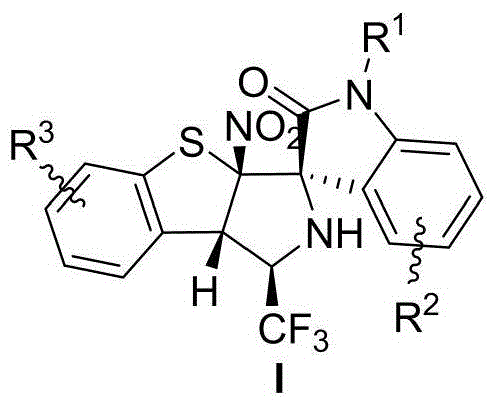
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of spiro-oxindole frameworks has heavily relied on the asymmetric dearomatization of electron-rich aromatic systems, such as indoles and naphthols, which utilize the inherent nucleophilicity of these substrates. While effective for certain applications, these conventional approaches often struggle when applied to electron-deficient heteroaromatic compounds, limiting the diversity of accessible chemical space. Specifically, the asymmetric dearomatization of 2-nitrobenzothiophene has been largely underexplored due to the difficulty in controlling the stereochemistry and reactivity of such electron-poor systems. Previous attempts to functionalize similar scaffolds often required harsh reaction conditions, expensive transition metal catalysts, or resulted in poor diastereoselectivity and low yields. Furthermore, the introduction of trifluoromethyl groups into these complex polycyclic systems via traditional routes often involves multi-step sequences that are not atom-economical and generate significant waste, posing challenges for both cost and environmental compliance in large-scale manufacturing.
The Novel Approach
The methodology described in the patent introduces a transformative [3+2] cycloaddition strategy that utilizes the asymmetric dearomatization of 2-nitrobenzothiophene, effectively bypassing the limitations of prior art. By reacting trifluoromethyl isatin imines with 2-nitrobenzothiophenes in the presence of a specialized organocatalyst, the process achieves the simultaneous formation of multiple bonds and stereocenters in a single operational step. This approach is distinguished by its mild reaction conditions, typically proceeding at temperatures ranging from -10°C to 50°C, with optimal results observed at 0°C. The reaction demonstrates exceptional compatibility with a wide range of solvents, although dichloromethane is preferred for maximizing both yield and stereoselectivity. ![General reaction scheme showing the [3+2] cycloaddition between trifluoromethyl isatin imine and 2-nitrobenzothiophene](/insights/img/trifluoromethyl-spiro-oxindole-synthesis-pharma-supplier-20260304221813-04.webp) The result is a highly efficient synthesis that delivers the target spiro compounds with excellent diastereomeric ratios (>20:1 dr) and enantiomeric excesses (up to 99% ee), providing a direct route to high-purity intermediates suitable for rigorous pharmaceutical screening.
The result is a highly efficient synthesis that delivers the target spiro compounds with excellent diastereomeric ratios (>20:1 dr) and enantiomeric excesses (up to 99% ee), providing a direct route to high-purity intermediates suitable for rigorous pharmaceutical screening.
Mechanistic Insights into Chiral Tertiary Amine-Thiourea Catalysis
The success of this asymmetric transformation hinges on the precise design of the chiral catalyst, which acts as a bifunctional activator to orchestrate the reaction between the electrophilic isatin imine and the nucleophilic nitrobenzothiophene. The patent highlights the use of chiral tertiary amine-thiourea catalysts, specifically structures designated as A, B, and C, which are engineered to facilitate dual hydrogen-bonding interactions. In this mechanistic model, the thiourea moiety activates the nitro group of the benzothiophene through hydrogen bonding, thereby increasing its nucleophilicity, while the tertiary amine component likely interacts with the imine proton or stabilizes the transition state through non-covalent interactions. 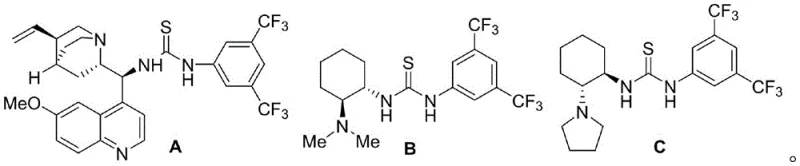 This cooperative activation lowers the energy barrier for the [3+2] cycloaddition and rigidifies the transition state, ensuring that the new carbon-carbon bonds are formed with strict stereochemical control. The ability of these organocatalysts to induce high levels of stereoselectivity without the need for toxic heavy metals is a critical advantage, simplifying the downstream purification process and ensuring the final product meets stringent regulatory standards for residual impurities.
This cooperative activation lowers the energy barrier for the [3+2] cycloaddition and rigidifies the transition state, ensuring that the new carbon-carbon bonds are formed with strict stereochemical control. The ability of these organocatalysts to induce high levels of stereoselectivity without the need for toxic heavy metals is a critical advantage, simplifying the downstream purification process and ensuring the final product meets stringent regulatory standards for residual impurities.
Furthermore, the mechanism allows for the generation of four contiguous chiral centers with defined absolute configurations, as confirmed by single-crystal X-ray diffraction analysis of the prototype compound I-a. The spatial arrangement of the substituents is dictated by the chiral pocket of the catalyst, which shields one face of the reacting species while exposing the other. This level of precision is vital for pharmaceutical applications, where the biological activity is often confined to a single enantiomer. The robustness of this catalytic system is evidenced by its tolerance to various substituents on both the isatin imine and the benzothiophene rings, including halogens, alkyl groups, and electron-withdrawing nitro groups, suggesting a versatile platform for generating diverse libraries of spiro-oxindole derivatives for structure-activity relationship (SAR) studies.
How to Synthesize Trifluoromethyl Spiro Oxindoles Efficiently
The practical implementation of this synthesis is designed to be straightforward and scalable, making it highly attractive for process chemistry teams looking to rapidly access novel chemical matter. The standard protocol involves dissolving the trifluoromethyl isatin imine and the 2-nitrobenzothiophene substrate in a dry organic solvent, followed by the addition of activated 4Å molecular sieves to maintain anhydrous conditions which are crucial for catalyst performance. A catalytic amount of the chiral thiourea organocatalyst is then introduced, and the mixture is stirred at a controlled temperature, typically around 0°C, for a period of 48 to 72 hours to ensure complete conversion. Upon completion, the reaction mixture is subjected to standard workup procedures, primarily involving filtration to remove the molecular sieves and concentration, followed by purification via silica gel column chromatography to isolate the pure spiro compound.
- Dissolve trifluoromethyl isatin imine and 2-nitrobenzothiophene in an organic solvent such as dichloromethane.
- Add molecular sieves and a chiral tertiary amine-thiourea bifunctional catalyst to the reaction mixture.
- Stir the reaction at mild temperatures (preferably 0°C) for 48-72 hours, then separate and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement and supply chain management in the fine chemical sector. The reliance on organocatalysis rather than precious metal catalysis eliminates the need for expensive palladium, rhodium, or iridium complexes, which are subject to volatile market pricing and supply chain disruptions. Moreover, the absence of heavy metals significantly reduces the cost and complexity associated with metal scavenging and removal steps during purification, leading to a more streamlined manufacturing process. The use of readily available starting materials, such as substituted isatins and nitrobenzothiophenes, ensures a stable supply chain, reducing the risk of production delays caused by raw material shortages. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme heating or cryogenic cooling, contributing to overall operational cost reductions.
- Cost Reduction in Manufacturing: The process utilizes low catalyst loadings, potentially as low as 1 mol%, which drastically reduces the cost of goods sold (COGS) related to chiral reagents. Since the catalyst is metal-free, there is no requirement for costly metal recovery systems or extensive testing for heavy metal residues, which are mandatory for API production. The high stereoselectivity achieved (>20:1 dr and up to 99% ee) minimizes the loss of material during chiral separation or recrystallization, maximizing the overall yield of the desired active isomer. Additionally, the simplicity of the workup procedure, which primarily relies on column chromatography, avoids the need for complex distillation or extraction protocols, further lowering labor and utility costs in a production environment.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are structurally simple and can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction across different solvents and temperatures provides flexibility in manufacturing; if a specific solvent becomes restricted or expensive, the process can be adapted to alternative green solvents without compromising the outcome. This adaptability ensures continuity of supply even in the face of regulatory changes regarding solvent usage. Furthermore, the stability of the intermediates and the final product facilitates easier storage and transportation, reducing the logistical complexities often associated with sensitive pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions are inherently safe and scalable, avoiding the use of hazardous reagents or explosive intermediates that often limit batch sizes in traditional synthesis. The high atom economy of the [3+2] cycloaddition means that most of the mass of the starting materials ends up in the final product, minimizing waste generation and aligning with green chemistry principles. This reduced waste profile simplifies effluent treatment and lowers the environmental footprint of the manufacturing site. The ability to run the reaction at near-ambient temperatures (0°C to 25°C) reduces the energy load on plant infrastructure, making it easier to scale from kilogram to multi-ton production without requiring specialized cryogenic reactors or high-pressure vessels.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these novel spiro-oxindole compounds, based on the detailed experimental data provided in the patent documentation. Understanding these nuances is essential for R&D teams evaluating this technology for their own drug discovery pipelines.
Q: What is the primary advantage of using 2-nitrobenzothiophene in this synthesis?
A: The use of 2-nitrobenzothiophene allows for the asymmetric dearomatization of electron-deficient heteroaromatic compounds, creating four continuous chiral centers which are difficult to achieve with traditional electron-rich substrates.
Q: How does the trifluoromethyl group benefit the resulting drug candidates?
A: The incorporation of a trifluoromethyl group significantly improves the metabolic stability of the drug molecule while altering its physical and chemical properties, potentially enhancing biological activity.
Q: What type of catalyst is required for high stereoselectivity in this process?
A: A chiral tertiary amine-thiourea bifunctional catalyst is essential, specifically structures similar to catalysts A, B, or C, which enable dual activation of the substrates for excellent enantioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Spiro Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of these trifluoromethyl-containing spiro oxindoles as key intermediates for next-generation therapeutics. As a leading CDMO partner, we possess the technical expertise to translate this innovative academic research into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle complex asymmetric syntheses, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is paramount in pharmaceutical supply, which is why our rigorous QC labs enforce stringent purity specifications to ensure every batch meets the highest international standards. Our team is dedicated to optimizing the reaction parameters to further enhance yield and reduce cycle times, ensuring a reliable supply of high-purity pharmaceutical intermediates for your critical development programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can reduce your overall development costs. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that combines cutting-edge chemistry with unwavering reliability, accelerating your path from discovery to market.
